The Handy Chemistry Answer Book (2014)
MACROSCOPIC PROPERTIES: THE WORLD WE SEE

PHASES OF MATTER AND INTENSIVE PROPERTIES
What are the different phases of matter?
There are three phases, or states, of matter that you come across every day—solids, liquids, and gases. There is a fourth phase of matter, plasma, which is only naturally found in stars and elsewhere in outer space. The distinctions between the first three phases are usually made using bulk properties. Solids have a defined shape and volume, while liquids easily change their shape but not their volume. Gases have neither a defined shape nor volume.
What is plasma?
Plasma, the fourth state of matter, is a gas where some amount of the particles have been ionized. As little as 1% ionization leads to very different properties for plasmas than gases, including increased conductivity (like lightning) and magnetization.
Are there plasmas in our daily lives?
Plasmas are found in fluorescent lights and neon lights. If you’ve ever seen a Tesla coil at a science museum, the arcs of light that they produce are plasma, as is lightning. Plasma TVs and plasma lamps are correctly named—both generate light using plasma, similar to fluorescent lights.
What is a phase diagram?
A phase diagram shows the phases of a particular substance as a function of temperature and pressure. An example for a single component phase diagram (as in not a mixture) is shown above. Phase diagrams also exist for mixtures, but these get very complicated very quickly.
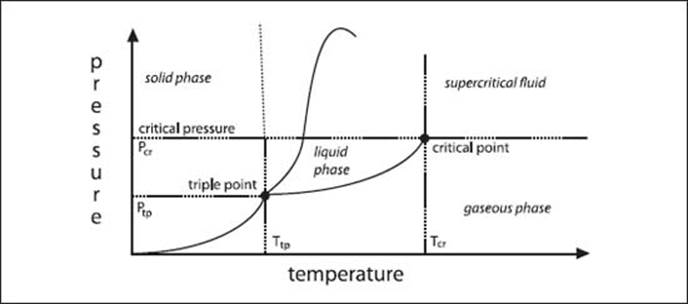
A phase diagram shows the phases of a particular substance as a function of temperature and pressure.
What is a triple point?
The temperature and pressure at which three phases of a substance are in equilibrium is known as a triple point. The three phases can be solid, liquid, and gas, but can also be two solid phases (different arrangements of the molecules in the solid) and a liquid phase.
What is a critical point?
A critical point is a combination of temperature and pressure values above which a phase boundary no longer exists. There are liquid-liquid critical points above which the two liquid phases become miscible, and also liquid-gas critical points above which the boundary between the liquid and gas phases disappears and the substance becomes supercritical.
What is a supercritical fluid?
Above its critical point, a given temperature and pressure combination, a substance behaves like both a liquid and a gas and is called supercritical. Supercritical fluids are very good solvents, like liquids, and as a result many modern chemical processes use them.
How is coffee decaffeinated?
Consider the chemistry required to decaffeinate coffee. Most techniques use an extraction process to remove the caffeine from the green coffee beans before they are roasted. One method starts by steaming the green beans and then rinsing them with an organic solvent (usually dichloromethane) to pull the caffeine molecules out of the beans. The other method uses supercritical carbon dioxide to extract caffeine. The latter obviously avoids the use of toxic solvents, but is energy-intensive. With all extraction techniques, it is challenging to only remove the caffeine and not the flavor compounds that we want to taste in our coffee.
How many phases can be in coexistence?
A rule called the Gibbs phase rule tells us how many phases can be in coexistence for a given substance or mixture. The rule arises from the fact that, to be in coexistence, a constraint exists that the chemical potentials of each component in each phase must be equal. After some math, one can find a relationship between the number of components of a system, the number of free variables (such as temperature, pressure, or the fraction of a given component present in a mixture), and the number of phases that can be in coexistence. This relationship is:
F = C – P + 2
where F is the number of degrees of freedom, C is the number of independent components, and P is the number of phases.
What is the difference between a homogeneous and heterogeneous mixture?
A homogeneous mixture is one that is uniformly mixed and has the same proportions of components throughout the mixture. An example is a transparent solution of sugar dissolved in water (specifically one in which there is no undissolved sugar floating around). A heterogeneous mixture is one that is not consistent or uniform throughout, such as a glass of sugar water with some chunks of undissolved sugar also floating around.
Can multiple liquid phases exist for a mixture?
Yes. One familiar example is a mixture of oil and water. The immiscible oil and water phases are two different phases of liquid matter.
Can there be more than one solid phase for a given substance?
Yes, solids can adopt different types of microscopic arrangements. If there is a repeating pattern to the atoms in a solid, it is called a crystal. If the ordered structure exists for the entire material, then the phase is known as a single crystal (think of a diamond). If a sample is a bunch of individual crystals, then we refer to the material as polycrystalline. Lots of solids also just have no pattern to the arrangement of their atoms, and this class is known as amorphous.
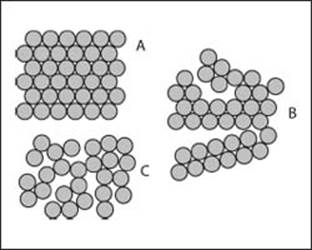
The arangement of atoms in a solid can take several form, depending on the arrangement of the atoms: A) crystalline, B) polycrystalline, or C) amorphous.
What is the density of a material?
Density is the mass (or weight) of a material divided by a unit of volume. Water, for example, has a density of 1.0 g/cm3 (1.0 gram per cubic centimeter).
What determines the density of a substance?
At the most basic level, density is determined by how close the atoms or molecules in a substance are packed, as well as the mass of those atoms. While it’s not quite as simple as assuming that the heaviest elements on the periodic table have the highest densities, having a high mass does help: heavy metals like iridium and osmium are the densest metals known to date. Remember that the density of a material does not depend on how much material you have; the density of 1 gram of lead is the same as the density of 1 kilogram of lead. Density is an intensive property, so changing the amount of a material you have does not affect its density.
Why does ice float?
Ice floats in water because it is less dense than water, though this is actually a very unusual case in terms of comparing the densities of the solid and liquid phases for a given substance. Most substances increase in density when moving from the liquid to the solid phase of matter, but H2O does the opposite. When water freezes, it forms a network of hydrogen bonds between H2O molecules, and because of the spacing of the molecules in this lattice, ice is less dense and floats in water.
What is temperature?
Temperature is a measure of the average kinetic energy of particles in a substance. What does that mean? “Average kinetic energy” is a precise way of saying how fast something is moving, in this case on a molecular level. The faster molecules vibrate, the hotter they feel because heat is being transferred from the object to your hands.
How are the Fahrenheit, Celsius, and Kelvin temperature scales related?
The Celsius and Kelvin scales use the same size degree (“incremental scaling” is the technical term), but set their zero values at different absolute numbers. Let’s explain that sentence a bit more: If you go up by one degree Celsius or one degree Kelvin, you’ve raised the temperature the same amount, but 0 °C (the temperature at which water freezes) is 273.15 K. Thus the two scales are offset from one another by 273.15.
Fahrenheit is completely different though. Water freezes at 32 °F, and a change of one degree on the Fahrenheit scale is equal to a change of 0.55 °C.

A comparison of the Fahrenheit, Celsius, and Kelvin scales.
What makes metals feel colder than air to the touch?
Metals feel colder than air when you touch them because they are good conductors of heat. The cold metal is able to conduct heat away from your hand through the entire object quickly, making it feel colder than the air around you.
What is a boiling point?
The technical definition of boiling point is the temperature at which the vapor pressure of the liquid phase equals the pressure of the surrounding gas (atmospheric pressure usually). This is a precise way of saying the temperature at which a liquid turns to vapor.
What molecular properties lead to higher boiling points?
There are a few factors that play important roles in boiling points of substances. The first is molecular weight: in general, heavier molecules have higher boiling points, which is pretty straightforward considering how we defined boiling point above (heavier molecules take more energy to move from the liquid to the gas phase).
All other properties that affect boiling point deal with intermolecular forces, or interactions between molecules. Think of this like the affinity of one molecule of a substance to be attracted to, or stick to, another molecule. Noncovalent bonds, like ionic or hydrogen bonds, significantly increase boiling point. Why? Because to move to the vapor phase the molecules typically have to break these interactions. Dipole interactions and Van der Waals forces have similar effects (see “Atoms and Molecules”), but these interactions are weaker, so the effect they have on boiling points is smaller. Finally, branching of the carbon backbone of a molecule is also frequently touted as a factor leading to lower boiling points; while this is true, it is really the weakening of Van der Waals forces at work here too.
What is a melting point?
The melting point is the temperature at which a substance changes from the solid state to the liquid state. At this exact temperature, the two phases are in equilibrium, so fractions of the sample are constantly moving between the two phases of matter. In practice, it is pretty difficult to observe the exact melting point of a substance.
What molecular properties lead to lower melting points?
Most of the trends we talked about for raising boiling points hold true for melting points for most of the same reasons. There is one major exception, though. The more branched, and therefore compact, a molecule is, the higher its melting point, because, in general, compact molecules will pack better in a crystal lattice. The better packed a lattice is, the more stable it is, and the more heat (energy) it takes to break up that lattice and melt the solid.
How do the boiling point and melting point of a substance change as a solute/impurity is added?
The addition of a solute typically raises the boiling point and lowers the melting point of a substance. These effects are appropriately named “boiling point elevation” and “melting point depression.”
Boiling points are raised when a nonvolatile solute (like NaCl) is added to a solution because the solute lowers the vapor pressure of the solution. That is a somewhat circular explanation though. It is important to know that this change in boiling point does not depend on what you add to the liquid, so there are no specific interactions going on here (like forming hydrogen bonds, etc.). As long as the solute has lower vapor pressure (remember we said it was nonvolatile, so its vapor pressure is essentially zero), this effect will be present. It is perfectly correct to think about it as just lowering the vapor pressure of the mixture (if you add something with very low vapor pressure, the average vapor pressure of whatever liquid you’re adding it to will go down).
Melting (or freezing) points are usually lowered when a solute is added to a liquid. The best explanation for this effect is based on entropy (see “Physical and Theoretical Chemistry”). When a molecule of solvent moves from the liquid to the solid phase (freezes), the amount of liquid solvent (i.e., its volume) is reduced. This means the same amount of solute is in a smaller space, which reduces their entropy (or raises their energy). This raising of energy means that you have to take even more energy out of the system for each molecule that joins the solid phase. Less energy means lower temperature, so adding a solute lowers the freezing point. An alternative way to look at this is that any impurity will disrupt the crystal lattice, raising its energy, relative to the liquid phase. This also contributes to lowering the freezing point as solutes are added.
How is the concentration of a solution defined?
The concentration of a substance is the amount of that substance in a solution divided by the volume of the solution. Chemists typically use molar concentration (moles of material/volume).
What properties influence solubility?
The most significant properties are intermolecular forces and temperature. If there are favorable interactions between the solute and the solvent, solubility will be higher. This is actually a balance of the interactions of the solute with the solvent and the stability of the solute in the solid phase. Temperature also influences solubility, and for most substances, solubility increases as the temperature of the solvent rises.
Why does putting salt on the road help to melt snow?
Like we discussed, when a solute is added to a solution, its freezing point is lowered. When salt is placed on snow, it begins to dissolve into any small amount of water on the ice with which it is in immediate contact. This lowers the freezing point of the surrounding water/ice, causing it to melt into the water. This process continues until the salt is completely dissolved.
What are the basic units of length?
Almost every length scale used in science is based on the meter. Chemistry frequently deals with very small lengths, so while you’re probably familiar with millimeters (10−3 m), it’s hard to have an intuitive sense about just how small a nanometer (10−9 m) is. There’s another length scale commonly used when talking about chemical bonds—the Ångström. An Ångström (Å) is one ten-billionth of a meter (10−10 m). The lengths of chemical bonds vary depending on the elements and other factors, but are usually around 1–2 Å.
How much space does an atom occupy?
The atomic radius of the smallest atom, hydrogen, is 53 × 10−12 meters, so it is about 10−10 meters in size. The atomic radius of the largest atom, cesium, is about 270 × 10−12 meters, or roughly five times that of hydrogen. These are all very, very small sizes!
How much space does the nucleus take up?
The nucleus of an atom takes up a very, very small fraction of the total space occupied by the atom. The diameter of a nucleus is on the order of 100,000 times smaller than that of a whole atom.
How long are chemical bonds?
Chemical bonds are typically about 2 atomic radii in length, since they are formed from two atoms joined together. These distances are on the order of 10−10 meters.
What are the basic units of pressure?
Unlike units of length and temperature, pressure is reported in at least six common units. The pascal (abbreviated Pa) is the official standard unit, but bar, millimeters of mercury (mmHg), standard atmospheres (atm), torr, and pounds per square inch (psi) are all used in different areas.
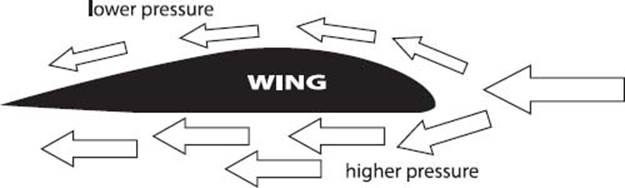
How do planes stay in the air?
Airplanes are very heavy, so the force required to balance gravity and keep them in the air must be large. The engines propel the airplane forward, but we need to understand what gives the upward push, or lift, necessary to keep the plane in the air. This lift comes from the shape of the wings, which are typically curved on the top and flat on the bottom. This design requires air to flow more rapidly over the top of the wings than over the bottom, which creates a lower air pressure above the wing than below. The lower air pressure above the wing is what lifts the plane off the ground and keeps it in the air. This is commonly referred to as the Bernoulli principle. If you blow across the top of a sheet of paper, you will see it lift into the air for the same reason.
What makes oil more slippery than water?
The purpose of lubricants, like motor oil, is to reduce the friction between surfaces so that parts last longer and less energy can be expended in the process of moving them. The key to a good lubricant is that the characteristic length scale for the formation of a thin film of the lubricant must be much smaller than the characteristic length scale of movement in the application. Basically, oils are good lubricants because they can form very thin films that persist even when the parts they serve to lubricate are constantly in motion. This ability to form thin films typically correlates with other properties that are easier to recognize. For example, good lubricants often have a high boiling point, low freezing point, high viscosity, and are stable toward chemical oxidation and changes in temperature.
What prevents all of the air from escaping Earth’s atmosphere?
Gravity! Every molecule on Earth is pulled toward the planet by gravity, even the very lightest gas molecules. To overcome a planet’s gravitational pull, an object, be it a space ship or a helium atom, must reach at least the escape velocity. Almost all of the molecules in the atmosphere are below this velocity because of the temperature of the planet Earth (higher temperature = faster molecules).
Almost all? Yes. Earth is very slowly losing its atmospheric gases. The lightest ones go first—about three kilograms of H2 escape the gravitational pull of Earth per year. A few particles can escape because kinetic energy of gases is a Boltzmann distribution, and in a Boltzmann distribution there are always very small probabilities of very high values.
What is the composition of air?
Earth’s atmosphere is made up of 78% nitrogen (N2) and 21% oxygen (O2), if you ignore water vapor, which shifts too much to include in averages for the entire planet. The last 1% is made up of mostly argon (Ar), followed by carbon dioxide (CO2) and other trace gases.
What gives a substance its color?
The color of a substance is the combination of the light that is reflected back at your eye. In other words, you’re seeing the light that is not absorbed by the substance. Certain frequencies can be absorbed because of the electronic structure of a substance, while others simply bounce off back into your eye.
What is a glass?
A glass is a noncrystalline solid—it lacks order in the solid state. In polymer chemistry, scientists are concerned with the glass transition temperature (Tg), which is the temperature at which a material changes from a hard to a rubbery state. What’s neat about this transition is that the material is not changing phases (i.e., from solid to liquid), but is changing from one type of solid to another.
What makes mercury so dangerous?
Mercury can be absorbed through the skin, making it particularly dangerous to handle. Organometallic mercury compounds, like dimethylmercury (CH3HgCH3), are particularly dangerous and have caused the deaths of a number of laboratory research chemists. Most research on this most toxic of mercury compounds has ceased. Be careful cleaning up that broken thermometer!
What is a vacuum?
A vacuum is space without matter. The word vacuum derives from a Latin word that means “empty.” A perfect vacuum, or one with absolutely no matter in a given space, is very hard to achieve, but through modern engineering scientists can get pretty close without having to go into deep space to run their experiments.
Can sound move through a vacuum?
No. Sound is a mechanical wave, which means that for the wave to move, actual molecules must bump into one another. In a vacuum, there is no matter, so in space, no one can hear you scream.
How does a vacuum cleaner work?
We commonly use the word vacuum to refer to any area that is of relatively low pressure. The air in areas of higher pressure will spontaneously move into areas of lower pressure, and this is the principle upon which a vacuum cleaner operates. A fan is used to push air out of the vacuum cleaner, creating an area of lower pressure behind the fan. Air from the outside then comes rushing in to reduce the gradient in pressure, carrying dust and dirt along with it. Since the fan is running continuously, the gradient in pressure is constantly being maintained, so the vacuum cleaner is able to keep running even though air is always flowing in.
Can light move through a vacuum?
Yes. Unlike sound, light is an electromagnetic wave, so no molecules are needed to propagate the wave. But you knew this—the Sun’s light crosses the vacuum of space to reach Earth each and every day.
What are some chemical reactions that we can observe with our naked eye?
There are lots of chemical reactions that we can easily detect by sight. Some examples are the formation of rust on metal, wood burning, fireworks exploding, silver becoming tarnished, or baking soda and vinegar reacting with one another.
What makes paper towels absorbent?
Paper towels are composed of cellulose fibers, which are polymers that contain many sugar monomers. These sugar monomers can interact strongly with water molecules, which is what makes paper towels so useful for cleaning up when something gets spilled.
What is an electric current?
Electric currents are the flow of electrons through a material. The electricity that comes out of the wall in your home or office is just the flow of electrons through a wire.

An explosion is a dramatic example of a chemical reaction we can see with our naked eyes.
What makes a material a good conductor of electricity?
Materials that are good conductors of electricity have many “free” electrons. Here we mean free in the sense that they are not bound strongly to a given atom or molecule. Metals are often good conductors of electricity. Whether or not a material has freely conducting electrons is related to the detailed electronic structure of the material, and a thorough description would be beyond the scope of this book. Basically, the more electrons that are free to move around in a material, the more easily it can carry current, and the higher its conductivity.
What makes a rubber band stretchy?
Rubber bands are made of long polymer molecules. These are all tangled together, and you could think of them as being similar to a bunch of interwoven springs. The polymers can be stretched to a more extended state, which is what allows a rubber band to stretch without breaking. More accessible configurations exist, however, when they are in a more contracted state, which means that the more contracted states have higher entropy (see “Physical and Theoretical Chemistry”) and for this reason are more favorable. This is what makes the rubber band want to contract and is what gives rise to its elasticity. (Check out “Polymer Chemistry” for more questions on polymers.)
How are soft drinks carbonated?
Soft drinks are carbonated using pressurized CO2 gas and a siphon that introduces the pressurized gas into the water or soda. The CO2 gas is forced into the liquid at a concentration beyond that which could exist under atmospheric conditions and then the container is sealed, preventing it from being released. That’s why if you leave a glass of soda sitting out, it will go flat; the CO2 escapes into the air.
Why might a soft drink freeze when you open it?
Remember that the freezing point of a solution is decreased by having anything dissolved in it. When a soft drink is opened and CO2 rushes out, less CO2 is present in the solution, causing the freezing point of the solution to increase. So at a constant temperature, the increase in freezing point causes the soda to freeze. If you haven’t seen this happen, you can try to make it happen by placing a soda in the freezer for a while, but be careful not to leave it in too long or it might explode!

Helium balloons float because the gas inside the balloon is much less dense than the surrounding atmosphere.
Why do helium balloons float?
Helium balloons float because helium is less dense than air, so gravity pulls down on air more than it does on the helium balloon. The difference in density is enough that the displaced air supports the weight of the balloon and makes it float…up, up, and away.
What is dry ice, and why does it “evaporate” from the solid phase?
Dry ice is solid carbon dioxide (CO2). At constant atmospheric pressure we see that an increase in temperature will bring it from the solid phase directly to the gas phase. This process is called sublimation, and it takes place at −78.5 °C.
How far away can one molecule “feel” another?
Molecules “feel” one another through the intermolecular forces they exert on one another. These typically span lengths that are a little longer than the length of chemical bonds, or something in the neighborhood of 5 × 10−10 meters.