Organic Chemistry I For Dummies, 2nd Edition (2014)
Part II. Hydrocarbons
Chapter 11. It Takes Alkynes: The Carbon-Carbon Triple Bond
IN THIS CHAPTER
Seeing the carbon-carbon triple bond
Naming alkynes
Seeing alkyne-making reactions
Reacting alkynes
As a functional group, alkynes are somewhat less common than alkenes, but they still have some very interesting features and reactivities that are unique to them.
Alkynes are molecules that contain carbon-carbon triple bonds. As you may expect, the reactivities and properties of alkynes are very similar to those of alkenes, although a few interesting differences between the two types of molecules also exist. In this chapter, I discuss the properties of alkynes, show you how to name alkynes, and cover the fundamental reactions that form alkynes and convert them into other functional groups.
Naming Alkynes
Alkynes are named under the systematic nomenclature scheme in the same way that alkenes are (refer to Chapter 9), except that instead of the name ending with the suffix –ene, the names of alkynes end with the suffix –yne. As with alkenes, a number in the prefix is used to indicate the position of the alkyne in the molecule, as demonstrated in Figure 11-1.
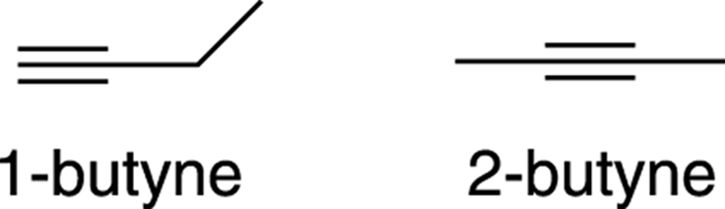
FIGURE 11-1: The names of two alkynes.
Alkynes are often known by their common names; these common names are derivatives of the simplest alkyne, acetylene. Under the common name system, the two R groups are named as substituents of acetylene (see Figure 11-2 for some examples). An alkyne with two methyl groups would be dimethyl acetylene, an alkyne with two isopropyl groups, diisopropyl acetylene, and so on.
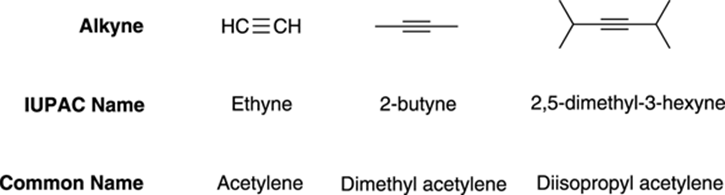
FIGURE 11-2: The common names of some alkynes.
Seeing Alkyne Orbitals
Alkyne carbons, by having only two substituents, are sp hybridized, and so have linear bonds that are oriented at 180 degrees to each other. (See Chapter 2 for more on hybridization.) The triple bond in an alkyne is made up of one sigma bond and two pi bonds. The two pi bonds result from the side-by-side overlap of the two p orbitals on each carbon atom (see Figure 11-3). As in the case with alkenes, the pi bonds are the reactive bonds in alkynes.
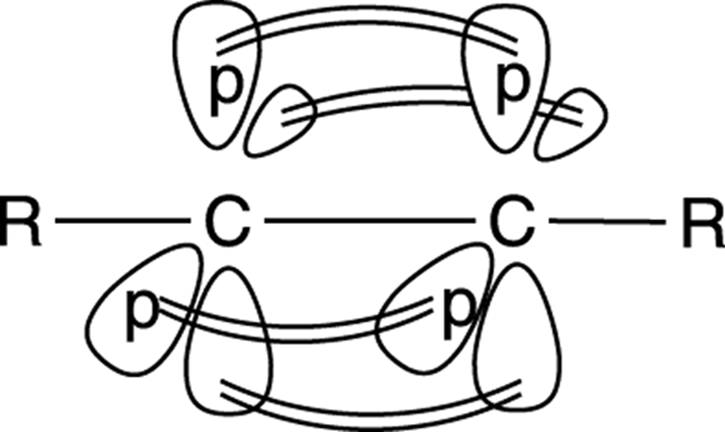
FIGURE 11-3: The pi bonds in an alkyne.
The overlapping p orbitals that make up the two pi bonds keep the alkyne in a linear geometry. Because alkynes have linear geometries, they’re drawn in a straight line, as shown in Figure 11-4.

FIGURE 11-4: The correct way to draw an alkyne.
Alkynes in Rings
Because alkynes prefer linear geometries, alkynes in small rings are highly unstable. Small- to medium-size cycloalkynes have bonds that must stretch out of their preferred geometries, and so inflict a good amount of ring strain on the atoms. Imagine grabbing your leg (which, like an alkyne, prefers to be linear) and touching your big toe to your nose, forming a ring with your body. The strain on your body is similar to the strain an alkyne feels. Just as you probably wouldn’t feel particularly stable forming a ring by touching your toe to your nose, alkynes in rings smaller than seven carbons are too unstable to be made or isolated. Cycloheptyne, the smallest cycloalkyne isolated, has been characterized, but it’s highly reactive (see Figure 11-5). Alkynes in rings containing more than seven carbons can be made and are fairly stable compounds.
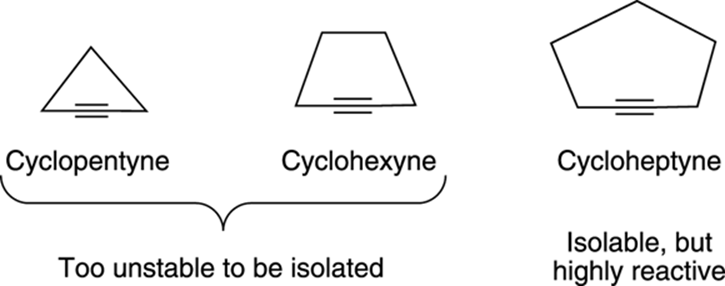
FIGURE 11-5: The relative stabilities of alkynes in small rings.
Making Alkynes
To make alkynes, two reactions are readily available, one that you may expect based on reactions that form alkenes, and another one that you may not expect based on what you know about the chemistry of alkenes.
Losing two: Dehydrohalogenation
To form alkenes (see Chapter 8), you can react one equivalent (unit) of base with an alkyl halide to eliminate one equivalent of acid and form the alkene. You may expect that to make an alkyne, you may treat a dibromide with two equivalents of base, eliminating two equivalents of acid to make the alkyne. This is, indeed, the case, as shown in Figure 11-6. Usually, the base that’s used is the strong base sodium amide (NaNH2, sometimes called sodamide), although sodium hydroxide (NaOH) can also be used.
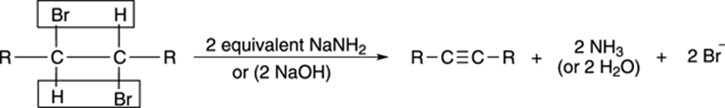
FIGURE 11-6: The double dehydrohalogenation of an alkyl dihalide.
 Recall that dibromides can be made by reacting an alkene with bromine (Br2). This is a convenient way to convert alkenes into alkynes: Brominate the alkene using Br2 to make the dibromide, and then treat the dibromide with two equivalents of base to make the alkyne.
Recall that dibromides can be made by reacting an alkene with bromine (Br2). This is a convenient way to convert alkenes into alkynes: Brominate the alkene using Br2 to make the dibromide, and then treat the dibromide with two equivalents of base to make the alkyne.
Coupling alkynes: Acetylide chemistry
The major difference between the reactivities of alkenes and alkynes is the result of the acidity of the protons attached directly to terminal alkynes. (Terminal alkynes are alkynes in which the triple bond is at the end of a chain; internal alkynes are alkynes whose triple bond is in the middle of a chain.) The pKa of a terminal alkyne proton is roughly 25, which means that the molecule is not terribly acidic, but it’s acidic enough for the hydrogen to be pulled off by a very strong base. (See Chapter 4 for a discussion of acids, bases, and pKa values.) The base that’s usually used to deprotonate terminal alkynes is sodium amide (NaNH2). Deprotonating the alkyne makes the acetylide anion, as shown in Figure 11-7.
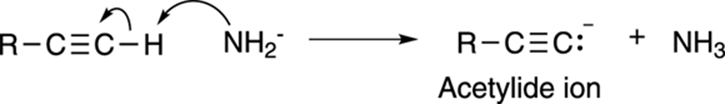
FIGURE 11-7: Making the acetylide anion.
This acetylide ion is a very useful nucleophile (or “nucleus lover”), and will react with primary alkyl halides to form a new, internal alkyne, as shown in Figure 11-8. This reaction is highly useful because it forms a carbon-carbon bond where one did not exist before. Note, however, that only primary alkyl halides can be used in this reaction; that is, the halide must be attached to a methylene (CH2) unit. Secondary and tertiary alkyl halides will not react in this way.
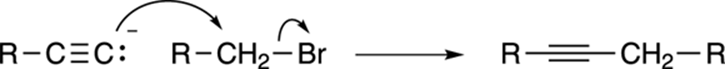
FIGURE 11-8: Acetylide addition.
Alkyne reactions are similar to those of the alkenes, and these reactions use reagents similar to those used in alkene reactions. Alkynes can form a variety of functional groups, as shown in Figure 11-9.
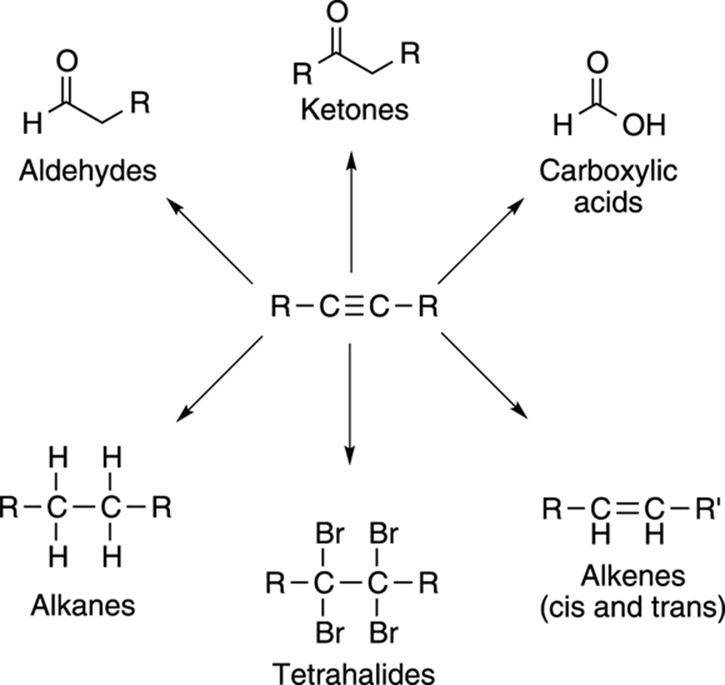
FIGURE 11-9: Some of the compounds that can be made from alkynes.
Brominating alkynes: Double the fun
Bromine reacts with the pi bond in alkynes to make the dibromide via the same mechanism as bromine addition to alkenes. (Do you recall the mechanism of the alkene reaction? Think “bromonium ion,” and see Chapter 8 if you need to refresh your memory.) Because there are two pi bonds in an alkyne, two equivalents (units) of bromine can be added to make a tetrabromide, as shown in Figure 11-10.
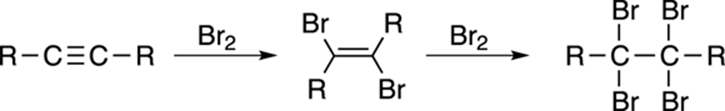
FIGURE 11-10: The bromination of an alkyne.
Saturating alkynes with hydrogen
Alkynes can also be reduced to alkanes by bubbling two equivalents of hydrogen gas (H2) over the alkyne in the presence of a metal catalyst as shown in Figure 11-11. This catalyst is usually palladium on carbon (Pd/C), but platinum (Pt) is also sometimes used.
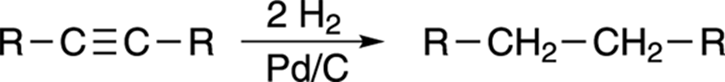
FIGURE 11-11: The saturation of an alkyne with hydrogen.
Adding one hydrogen molecule to alkynes
Stopping the reaction of alkynes with hydrogen at the alkene stage is possible because alkenes are somewhat less reactive than alkynes, but this reaction requires a special catalyst. To reduce an alkyne to the cis alkene (see Chapter 9 for more about cis and trans alkenes), you use Lindlar’s catalyst, which is a cocktail of palladium (Pd) powder made less reactive with added lead (Pb) and quinoline (C9H7N). (In reaction diagrams, instead of writing out all the components of the catalyst, chemists often write “Lindlar’s catalyst” over or under the arrow.) Lindlar’s catalyst is not as reactive as palladium on carbon (Pd/C) and generates the cis alkene, as shown in Figure 11-12.
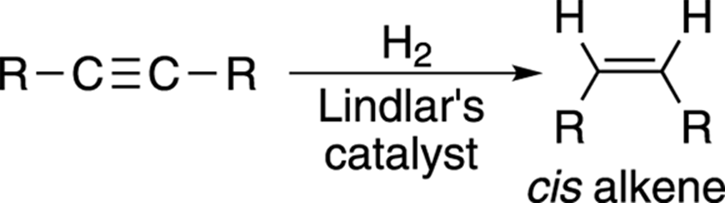
FIGURE 11-12: Using Lindlar’s catalyst to make cis alkene.
To convert an alkyne to the trans alkene, you use sodium metal (Na) in liquid ammonia (NH3), as shown in Figure 11-13.
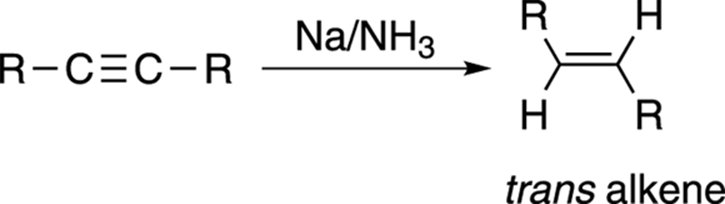
FIGURE 11-13: Making a trans alkene from an alkyne.
Oxymercuration of alkynes
Reacting alkynes with mercury, water, and acid, you may expect the reaction to make the alcohol on the double bond, just as mercuric acetate reacts with alkenes to make the Markovnikov alcohol (see Chapter 10 for a review of Markovnikov products and the oxymercuration of alkenes). Indeed, the Markovnikov enol (an alcohol on a double bond) is formed in which the alcohol group is placed on the more-substituted carbon. However, enols are unstable and rapidly convert into ketones, as shown in Figure 11-14.
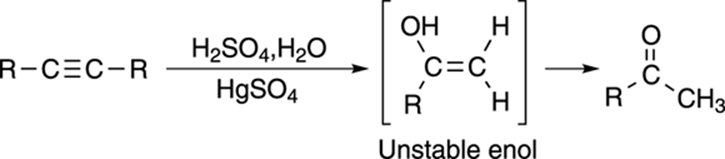
FIGURE 11-14: The oxymercuration of an alkyne.
 The term enol is a combination of the suffixes –en (from alkene) and –ol (from alcohol).
The term enol is a combination of the suffixes –en (from alkene) and –ol (from alcohol).
The reaction that converts the enol into the ketone is called a tautomerization reaction, and both the enol and the ketone are considered tautomers of each other. Tautomers are molecules that differ only in the placement of a double bond and a hydrogen.
Hydroboration of alkynes
The hydroboration reaction of alkynes works in the same way as the hydroboration reaction of alkenes, and forms the anti-Markovnikov product. In the case of the hydroboration of alkynes, the product is an enol with the alcohol group on the least-substituted carbon, as shown in Figure 11-15. As with the oxymercuration reaction, this enol is unstable and tautomerizes to the aldehyde.
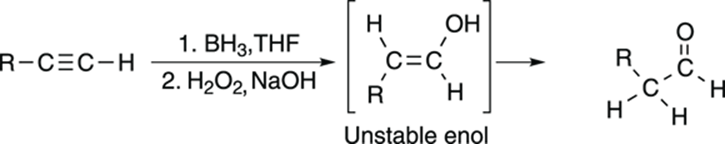
FIGURE 11-15: The hydroboration of an alkyne.
 Oxymercuration and hydroboration of carbon-carbon triple bonds are useful primarily with terminal alkynes because with terminal alkynes, you get only a single product (with hydroboration, you get the aldehyde; with oxymercuration, you get the ketone). With internal alkynes, both sides of the alkyne are equally substituted, so water can be added equally well to either side of the triple bond, so these reactions yield mixtures of two products.
Oxymercuration and hydroboration of carbon-carbon triple bonds are useful primarily with terminal alkynes because with terminal alkynes, you get only a single product (with hydroboration, you get the aldehyde; with oxymercuration, you get the ketone). With internal alkynes, both sides of the alkyne are equally substituted, so water can be added equally well to either side of the triple bond, so these reactions yield mixtures of two products.