Organic Chemistry I For Dummies, 2nd Edition (2014)
Part III. Functional Groups
Chapter 13. Getting Drunk on Organic Molecules: The Alcohols
IN THIS CHAPTER
Seeing and classifying alcohols
Naming alcohols
Seeing reactions that form alcohols
Seeing reactions that convert alcohols into other groups
When most people think of alcohol, they think of ethyl alcohol, the alcohol found in beer and wine. Many people, though, don’t realize that there are many thousands of different alcohols. Ethylene glycol is an alcohol found in antifreeze; methanol is a wood alcohol that can cause blindness if ingested; isopropyl alcohol is an alcohol used for sterilizing cuts. Alcohols, like the alkenes, are an extremely important functional group, not just because they’re found in so many valuable products (and not just beers and wines), but also because they’re so versatile: They’re easily formed and converted into many other functional groups.
In this chapter, I introduce you to the alcohol functional group, show you how to name and classify alcohols, and show you reactions that both make alcohols and convert alcohols into other functional groups.
Classifying Alcohols
Alcohols are molecules that contain a hydroxyl (OH) group, and they’re typically classified by the carbon to which the hydroxyl group is attached. If the carbon bonded to the OH is attached to one other alkyl group, the alcohol is classified as primary (1°); if the carbon is attached to two other alkyl groups, the alcohol is classified as secondary (2°); if the carbon is attached to three alkyl groups, the alcohol is classified as tertiary (3°), as shown in Figure 13-1.
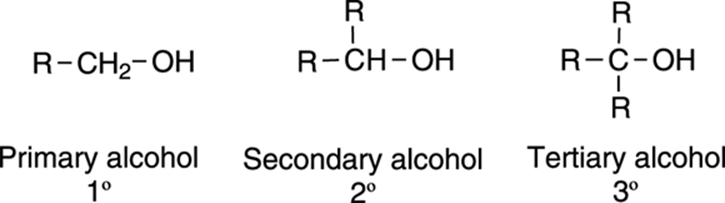
FIGURE 13-1: Alcohol classification.
An Alcohol by Any Other Name: Naming Alcohols
You can name alcohols just by extending the nomenclature rules used for alkanes (refer to Chapter 7 for an alkane refresher). To name an alcohol you follow these five steps:
1. Determine the parent name of the alcohol by looking for the longest chain that includes the alcohol.
Snip the e off the suffix for the alkane and replace it with the suffix –ol, which stands for alcohol. For example, a two-carbon alcohol would not be ethane but ethanol.
2. Number the parent chain. Start numbering from the side closer to the hydroxyl group.
3. Identify all the substituents of the parent chain and name them.
4. Order the substituents alphabetically in front of the parent name.
5. Identify the location of the hydroxyl group by placing a number in front of the parent name.
Now try naming the alcohol shown in Figure 13-2.
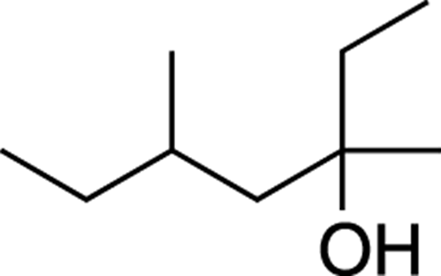
FIGURE 13-2: An alcohol.
First, find the parent chain, as shown in Figure 13-3. The parent chain is the longest chain of carbons that contains the hydroxyl group. In this case, the parent chain is seven carbons long, so this is a heptanol.
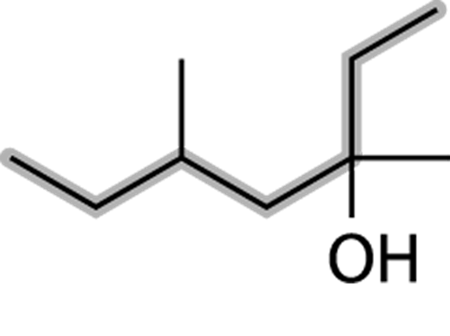
FIGURE 13-3: The parent group.
Then number the parent chain, starting from the end that reaches the hydroxyl group sooner. In this case, that’s from right to left (see Figure 13-4).
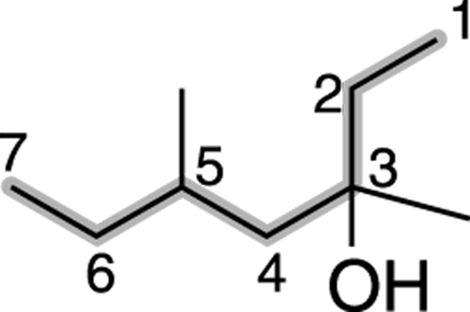
FIGURE 13-4: Numbering the chain.
Find and name the substituents. The molecule shown in Figure 13-5 has two methyl group substituents — one at the number-three carbon and one at the number-five carbon.
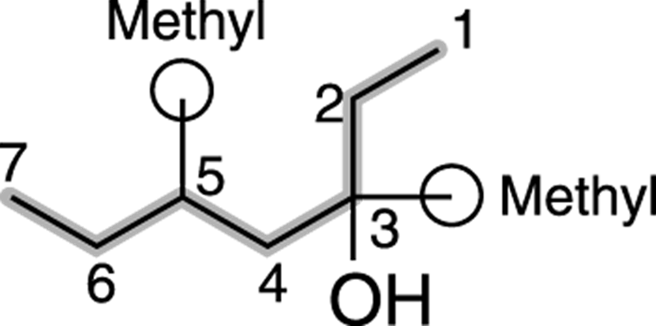
FIGURE 13-5: Identifying substituents.
Then place the substituents (in alphabetical order) in front of the parent group. Indicate the position of the hydroxyl group by placing a number in front of the parent name. The two methyl groups combine to make dimethyl, and the name for this alcohol is, therefore 3,5-dimethyl-3-heptanol.
Alcohol-Making Reactions
So, now, take a look at how you can make alcohols.
Adding water across double bonds
Chapter 10 shows you two reactions that produce alcohols from alkenes. The oxymercuration of alkenes makes the Markovnikov alcohol (the alcohol with the OH on the most highly substituted carbon), while hydroboration makes the anti-Markovnikov alcohol (the alcohol with the OH on the least-substituted carbon). This conversion is outlined in Figure 13-6.
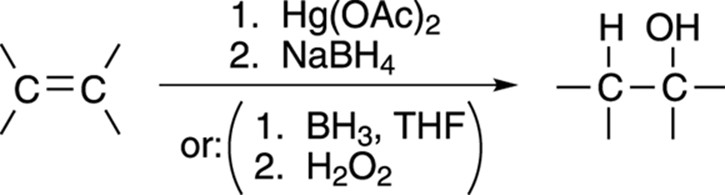
FIGURE 13-6: Adding water to an alkene.
Reduction of carbonyl compounds
Alcohols can also be formed by the reduction of carbonyl (C=O) compounds. Two reagents are widely used to reduce carbonyl compounds, sodium borohydride (NaBH4), a weaker reducing agent, and lithium aluminum hydride (LiAlH4), a stronger reducing reagent. In metaphorical terms, sodium borohydride acts as a reductive popgun, while lithium aluminum hydride acts as a reductive cannon, as illustrated in Figure 13-7.
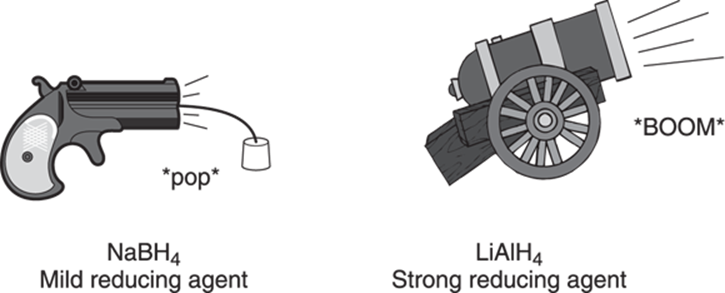
FIGURE 13-7: The relative strengths of reducing agents.
What do I mean by sodium borohydride being a weaker reducing agent than lithium aluminum hydride? Well, sodium borohydride can reduce carbonyl compounds that are easy to reduce to alcohols, like aldehydes and ketones. But this reagent is not strong enough to reduce the more stubborn carbonyl compounds like esters and carboxylic acids. For that job, you bring out the cannon reagent (lithium aluminum hydride), which is strong enough for the task.
Reducing aldehydes with sodium borohydride (or lithium aluminum hydride) is a very convenient way of making primary alcohols, as shown in Figure 13-8. Similarly, the reduction of ketones with sodium borohydride is a convenient way of preparing secondary alcohols. Tertiary alcohols cannot be prepared by reduction.
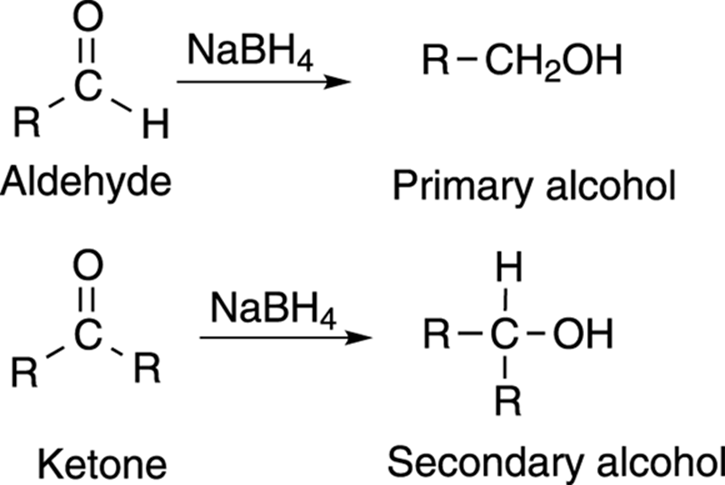
FIGURE 13-8: The formation of an alcohol by the reduction of an aldehyde or ketone.
Lithium aluminum hydride is strong enough to reduce both carboxylic acids and esters, as shown in Figure 13-9. Both carboxylic acids and esters are reduced to primary alcohols with this reagent.
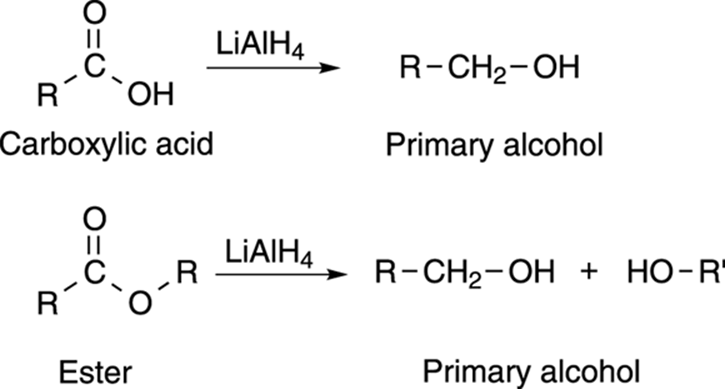
FIGURE 13-9: The formation of an alcohol via a lithium aluminum hydride reduction.
The Grignard reaction
Another extremely useful reaction for making alcohols is the Grignard reaction (pronounced grin-yard). To make alcohols using the Grignard reaction, you react a “Grignard reagent” with a carbonyl compound. Making a Grignard reagent is fairly simple: You simply add magnesium to an alkyl halide, as shown in Figure 13-10, which inserts the magnesium into the C-X bond to make the Grignard reagent.
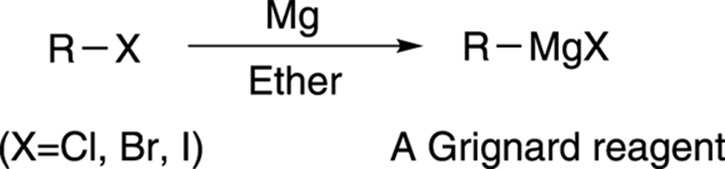
FIGURE 13-10: Making a Grignard reagent.
A Grignard reagent is an extremely powerful nucleophile (nucleus lover), and can react with electrophiles like carbonyl compounds. To determine the products made in a Grignard reaction, you can ignore the magnesium halide portion of the reagent (because this portion doesn’t get involved in the reaction) and think of the Grignard reagent as acting as a carbanion in disguise. Figure 13-11 illustrates this idea.
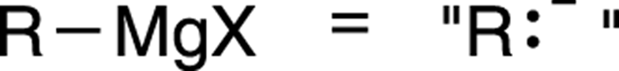
FIGURE 13-11: A Grignard reagent.
The mechanism for the addition of a Grignard reagent to a carbonyl is shown in Figure 13-12.
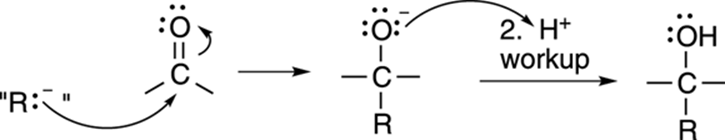
FIGURE 13-12: The Grignard reaction.
Although you can make only primary and secondary alcohols by reduction, you can make all kinds of alcohols using the Grignard reaction. If you react a Grignard reagent with formaldehyde, as shown in Figure 13-13, you can make primary alcohols. If you react it with an aldehyde, you get secondary alcohols. If you react it with ketones, you get tertiary alcohols.
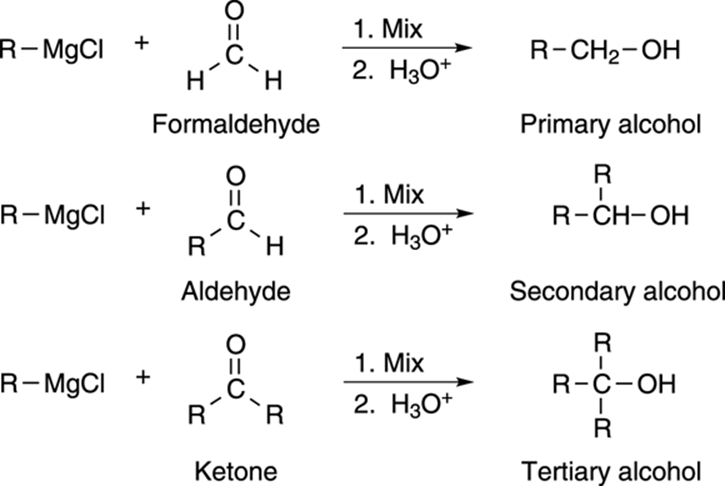
FIGURE 13-13: The formation of alcohols via the addition of Grignard reagents to carbonyl compounds.
Reactions of Alcohols
So, what can you do with alcohols? Now that you’ve seen reactions that make alcohols, I show you reactions that convert alcohols into other functional groups.
Losing water: Dehydration
One of the best reagents for converting alcohols into alkenes is phosphorous oxychloride (POCl3). This reagent causes the alcohol to lose water to make the alkene, as shown in Figure 13-14.
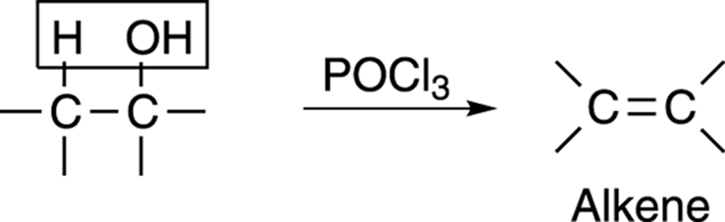
FIGURE 13-14: The dehydration of an alcohol.
Making ethers: Williamson ether synthesis
Ethers can also be made from alcohols. Reacting an alcohol with sodium metal forms an alkoxide, the alcohol’s conjugate base, as shown in Figure 13-15. Alkoxides are strong nucleophiles that can react with primary halides to make ethers in an SN2 reaction (refer to Chapter 12).
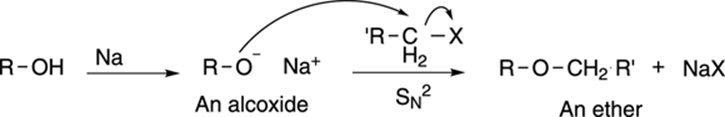
FIGURE 13-15: The Williamson ether synthesis.
Oxidation of alcohols
In the previous section, I showed how alcohols can be made by the reduction of carbonyl compounds. Running that reaction in reverse is also possible, and oxidizing alcohols into carbonyl compounds (ketones and aldehydes) can be accomplished with either of two different reagents — PCC and the Jones reagent. PCC (which stands for pyridinium chlorochromate) is a weaker oxidizing agent than the Jones reagent (a mix of chromium trioxide [CrO3] and acid). For example, PCC oxidizes primary alcohols into aldehydes. The Jones reagent, on the other hand, oxidizes the primary alcohol even further into the carboxylic acid (see Figure 13-16). Both reagents oxidize secondary alcohols into ketones.
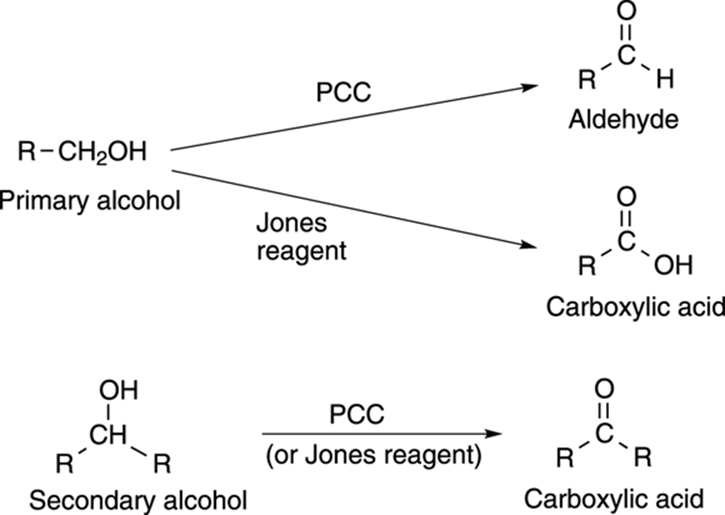
FIGURE 13-16: The oxidation of alcohols.
You cannot oxidize a tertiary alcohol.