Organic Chemistry I For Dummies, 2nd Edition (2014)
Part IV. Spectroscopy and Structure Determination
Chapter 20. Following the Clues: Solving Problems in NMR
IN THIS CHAPTER
Using clues to determine structure
Tackling NMR problems
Avoiding common NMR problem-solving mistakes
Irealize that spectroscopy problems are intimidating, particularly when you’re first approaching them. A spectrum presents so much information, so many peaks and lines and squiggles that trying to interpret one for the first time can make you feel like a child trying to read Moby-Dick.The amount of information to absorb makes it hard to even know where to begin.
The trick is not to get intimidated or discouraged. You may groan when you hear this now, but after you’ve worked enough problems to become good at them, solving spectroscopy problems can be really fun. Think of these problems as puzzles whose rules happen to be dictated by nature.
In this chapter, I present a general guide to figuring out the structures of molecules using NMR spectroscopy (for a description of how NMR works, see Chapter 19). Unfortunately, what I present isn’t a definitive guide, because chemists don’t have an indisputable “right way” to solve every NMR problem. Because of this, many textbooks don’t give a specific problem-solving strategy. Instead, they instruct you to “work lots of problems” and assure you that after a while and lots of hard work you’ll get the hang of it. Working many problems is, indeed, excellent advice, but it leaves you in the awkward position of not knowing where to begin. As a result, students often start in the wrong places (I talk about common mistakes a little later in this chapter), and many of them never grasp how to work these types of problems. Come exam time, they’re often ready to admit themselves to the local psychiatric ward.
Because of the difficulty so many students have with solving problems in NMR spectroscopy, I present the structure-solving strategy that works for me and that’s worked for others. I suggest that it might work for you too, at least as a foundation until you’re confident enough to alter it to fit your own learning style and adapt it to the particular problem at hand. Spectroscopy problems can be particularly intimidating because they can be solved in a number of different ways. But the way I like to determine the structure of a compound using NMR is systematically, by following the clues.
Follow the Clues
I like to think of solving spectroscopy problems as analogous to a detective in the dark with a flashlight, searching for “whodunit.” Methodical clue gathering is the best way to determine the culprit — the mischievous compound in this case — instead of blindly waving the flashlight (or pencil) about, hoping for some sort of chance epiphany (which never comes during an exam, but only after you’ve turned it in).
You’re likely to encounter many different kinds of NMR problems. Many problems will give you at least the molecular formula and the 1H NMR spectrum. Some problems will also provide the IR spectrum, mass spectrum, and 13C NMR spectrum as well. This clue-gathering technique I give you can be tailored to fit just about any of these problem types; simply ignore any clues that don’t apply to the particular problem that you’re working on. After I tell you about how to gather all the clues, I use two examples to show you how to apply this guide to real problems.
Here’s the rundown of how the clue gathering works. You start by determining the degrees of unsaturation from the molecular formula. This gives a quick indication of whether the compound contains rings, double bonds, or triple bonds, or whether the molecule is saturated. A more definitive indication of what kind of molecule you’re dealing with can be found by looking at the IR spectrum (if you’re given one in the problem) and also by a quick peek at the 1H NMR for peaks that scream out a particular functional group (specifically, carboxylic acids, aldehydes, and aromatic rings). Next, you use the integration (plus the clues from the degrees of unsaturation and the IR) to determine all the fragments of the molecule. With fragments in hand, you put the pieces together in a way that’s consistent with the chemical shift and the peak splitting. Finally, you double-check your proposed structure by making sure that everything is consistent with all the clues. In short, first you determine all the pieces of the molecule, and then you try to fit them together in the correct way.
That’s the big picture of how you solve a structure with this technique. Next, I cover the practical details of each of those steps. So, switch on your flashlight, detective — here’s how to gather those clues!
Clue 1: Determine the degrees of unsaturation from the molecular formula
For a reminder on degrees of unsaturation (DOU), mosey on over to Chapter 9.
 If you’re given the molecular formula, just plug the formula into the equation:
If you’re given the molecular formula, just plug the formula into the equation:
![]()
If you have a formula with more than just carbons and hydrogens, you also need the following conversions:
· Halogens (F, Cl, Br, I): Add one hydrogen to the molecular formula for each halogen present.
· Nitrogen: Subtract one hydrogen for each nitrogen present.
· Oxygen or sulfur: Ignore.
The degree of unsaturation is a valuable clue because it tells you how many double bonds, triple bonds, or rings must be present in the unknown compound (although it doesn’t tell you which of these elements of unsaturation are present).
 Double bonds and rings count for one degree of unsaturation each, and triple bonds count for two degrees each. If the molecule has zero degrees of unsaturation, you know that there are no double bonds, triple bonds, or rings in the molecule.
Double bonds and rings count for one degree of unsaturation each, and triple bonds count for two degrees each. If the molecule has zero degrees of unsaturation, you know that there are no double bonds, triple bonds, or rings in the molecule.
Clue 2: Look at the IR spectrum to determine the major functional groups present in the unknown compound
To brush up on IR spectroscopy, see Chapter 18. If possible, confirm the presence of the major functional groups in the NMR spectrum. Easy ones to confirm include aldehydes, carboxylic acids, and benzene rings.
Clue 3: Determine the peak ratios by measuring the heights of the integration curves
Determine how many hydrogens each peak represents by comparing the integration ratios to the molecular formula. Especially if you’re just starting to learn how to solve 1H NMR problems, using a ruler can be indispensable for finding out the relative integration ratios. What you need to measure with the ruler is the height of the integration curve over a given peak (the integration curve is the squiggle over a peak that looks like a capital S). These heights are related to the areas underneath a peak, which in turn are related to the number of hydrogens that the particular peak represents. Determining how many hydrogens each peak represents will help you to find the fragments of the molecule in Clue 4.
Here’s an example of how to determine the integration using a ruler (see Figure 20-1; for clarity, the peaks underneath the integrations aren’t shown). First, you want to establish the relative ratio between the integrations. To do this, use a ruler to measure the difference between the top and the bottom of each of the integration curves. After you measure the height of each peak on the spectrum, divide each of the heights by the smallest height. This will give you the ratios.
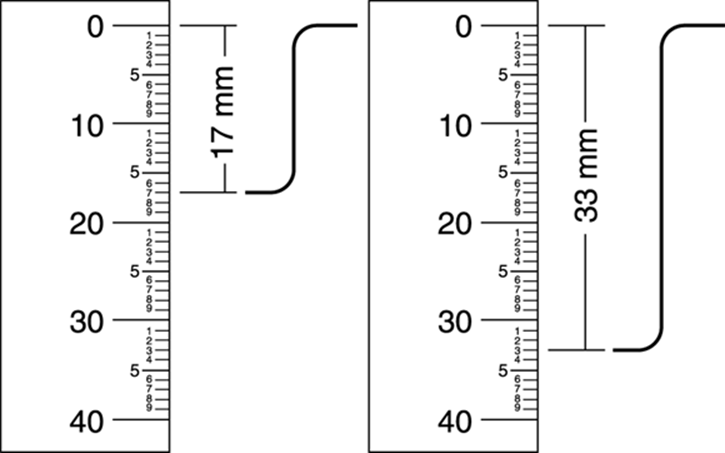
FIGURE 20-1: Using a ruler to measure integration peaks.
In this example, the smallest height is 17 mm, so you divide both heights by 17 mm. In this case, this gives an integration ratio of 1:1.94. Because you can never have a fraction of a hydrogen atom, you need to convert the relative ratios into whole numbers.
As is the case in this example, you often find that the integrations don’t work out to be perfect whole numbers. If the numbers are very close to whole numbers, the integrations are probably just in error by a bit, and you simply need to round them to the nearest whole number. Because 1.94 is very close to 2.0, in this case you would say that the integration is 1:2.
 These imperfect integrations can be attributed to noise in the baseline, a poorly calibrated integral, impurities in the sample, and errors made while measuring the heights of the curves with the ruler.
These imperfect integrations can be attributed to noise in the baseline, a poorly calibrated integral, impurities in the sample, and errors made while measuring the heights of the curves with the ruler.
For ratios that are not close to whole numbers, you need to multiply the ratio by the smallest number that makes both numbers in the ratios whole numbers. If the ratio were 1.0:1.5, for example, you would multiply both parts of the ratio by two to give a whole-number ratio of 2:3. If the ratio were 1.0:1.33, you would multiply both by three to give a whole-number ratio of 3:4, and so on.
So, for this example, you could say that your relative ratio of hydrogens is 1:2. Alternatively, using organic-speak, you could say that the peak on the left integrates for one and that the peak on the right integrates for two.
But does this then mean that the peak on the left represents one hydrogen, and the peak on the right represents two hydrogens? Not necessarily. Remember that this is the relative ratio of hydrogens, not the absolute ratio, which means that you know only that the larger peak has twice the number of hydrogens as the smaller peak; you don’t know the actual number of hydrogens. To find the absolute number of hydrogens, you must compare your ratio to the molecular formula.
If the sum of all the numbers in the relative ratio matches the number of hydrogens in the molecular formula — in this example, if there were only three hydrogens in the molecular formula — then you could say that the peak on the left represents one hydrogen and the peak on the right represents two hydrogens, because then all the hydrogens in the molecule would be accounted for. But if the molecular formula had six hydrogens in it, you would have to multiply the relative ratio by two to make the number in your spectrum match the number in your molecular formula — so in that case the peak that integrates for one would represent two hydrogens and the peak that integrates for two would represent four hydrogens. If there were nine hydrogens, you would multiply the relative integrations by three, and so forth.
 Remember that the sum of all the hydrogens (absolute number) that you derived from the integrations must exactly match the number of hydrogens in the molecular formula. If the numbers don’t match, then you need to reconsider the values of the integrals.
Remember that the sum of all the hydrogens (absolute number) that you derived from the integrations must exactly match the number of hydrogens in the molecular formula. If the numbers don’t match, then you need to reconsider the values of the integrals.
Here’s a summary of the steps to determine the number of hydrogens represented by each peak:
· Measure the heights of each peak using a ruler.
· Divide each of the heights by the smallest height measured.
· If one or more of the numbers are not whole numbers, convert them to whole numbers by rounding (if they are all very near a whole number) or by multiplying all the numbers in the ratio by a whole number. This gives you the relative ratio of hydrogens in the peaks.
· To determine the actual number of hydrogens each peak represents, add each of the numbers in your relative ratio and compare this number to the number of hydrogens in the molecular formula. If these numbers are not equal, determine what you need to multiply your relative ratio by in order to make the number of hydrogens in your spectrum match the number of hydrogens in the molecular formula. If they’re the same, you don’t need to multiply the ratio by anything.
Clue 4: Break the NMR peaks into fragments using the integration from Clue 3
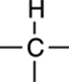
After you’ve determined the number of hydrogen atoms that each peak represents, you can assign each peak to a fragment of the molecule. Table 20-1 shows some common fragments.
TABLE 20-1 Common Fragments
|
Number of Hs |
Likely Fragment |
Notes |
|
1H |
|
Check the IR spectrum to make sure this is not an alcohol (OH), secondary amine (NH), aldehyde (CHO), or acid proton (COOH). |
|
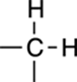
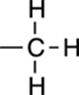
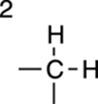
2H |
|
Rarely: This is two symmetric CH groups. |
|
3H |
|
Rarely: This is three symmetric CH groups. |
|
4H |
|
Usually two symmetric methylene (CH2) groups. |
|
6H |
|
Rarely: This is three symmetric CH2 groups. Often indicates an isopropyl group. |
|
9H |
|
Often indicative of a tertiary butyl group. |
As you determine the fragments in the molecule, write them all down on a sheet of scratch paper so they’ll be in front of you when you’re ready to solve the structure. For example, if you have three peaks — one that integrates for 1H, one that integrates for 2H, and one that integrates for 3H — you would write on your scrap paper CH, CH2, and CH3. After you determine all the fragments (including fragments you determined from the IR, if you’re given one), add up all the atoms in your fragments to make sure that they match the molecular formula and to be certain that you’re not missing any atoms.
Clue 5: Combine the fragments in a way that fits with the NMR peak splitting, the chemical shift, and the degrees of unsaturation
Often, when you’re starting, the best way to go from fragments to a possible structure is to simply brainstorm all the possible molecules that could include the fragments you listed, because in many cases there will be more than one way to put all the fragments together. After you’ve brainstormed all the possible structures, you systematically eliminate all the incorrect structures. You can eliminate the incorrect structures based on a few different factors. Most often, the incorrect structures will not produce the peak splitting that you see in your NMR spectrum, or will have symmetry when your NMR spectrum suggests that the molecule is not symmetrical. But sometimes you may need to use the chemical shift to eliminate incorrect structures. This procedure may seem tedious, but after working a few problems, you’ll develop an intuition about which structures are likely correct, and putting together the fragments to make the correct structure will take no time at all.
 After working several problems, you’ll notice some common patterns. A quartet that integrates for 2H and a triplet that integrates for 3H on a spectrum is often indicative of an ethyl group (–H2CH3). Likewise, a multiplet that integrates for 1H and a doublet that integrates for 6H usually suggest an isopropyl group (–CH(CH3)2). A singlet integrating for 9H is usually a t-butyl group (–C(CH3)3).
After working several problems, you’ll notice some common patterns. A quartet that integrates for 2H and a triplet that integrates for 3H on a spectrum is often indicative of an ethyl group (–H2CH3). Likewise, a multiplet that integrates for 1H and a doublet that integrates for 6H usually suggest an isopropyl group (–CH(CH3)2). A singlet integrating for 9H is usually a t-butyl group (–C(CH3)3).
Clue 6: Recheck your structure with the NMR and the IR spectrum to make sure it’s an exact match
For this clue, look only at your proposed compound, and then predict what the spectrum should look like, going carbon atom by carbon atom. Then compare this expected spectrum of the compound you proposed to the real spectrum to see if they match. Here is where you check to see if the chemical shift for each proton (H) makes sense. Remember: Protons near electronegative atoms (like N, O, Br, F, Cl, and so on) will have a higher chemical shift than protons that that are not near electronegative atoms.
So, what if you get to Clue 5 or Clue 6 and realize, “Oh no! My structure’s wrong!”?
In that case, there are two checks to spot the problem.
Check 1
The first thing to check is whether you combined the fragments in the correct way. Often, there is more than one way to put the structure together from the fragments. If you’ve tried all the ways and none of them checks out, then the problem is probably with the fragments themselves (see Check 2).
Check 2
A less likely possibility is that the fragments themselves are incorrect. The likely fragment in Table 20-1 will be correct in most cases — so it’s certainly a good place to begin. Unfortunately, this procedure is not 100 percent foolproof. When it does fail, it’s usually with molecules that have a high degree of symmetry. If the procedure fails, you’ll reach Clue 5 and be unable to figure out how to reasonably put the fragments back together again in a way that makes sense with the splitting and chemical shift. In that case, return to the table and rethink your choices of fragments, looking in the notes column to see alternative possibilities.
An example illustrating how the most likely fragment in the table would be incorrect is shown in Figure 20-2. Because of the symmetry in the molecule — that is, the left-hand side of the molecule is identical to the right-hand side of the molecule — the two CH3 groups are identical and will show a single peak (with an integration of 6H), and the two CHs will also show a single peak (with an integration of 2H).
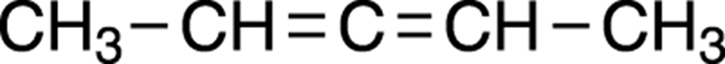
FIGURE 20-2: Dimethyl allene.
From the 6H row of Table 20-1, the two symmetrical CH3 fragments are predicted correctly in the “Likely Fragment” column; the 2H integration, however, is predicted to be a CH2 fragment rather than two identical CH fragments. Here, choosing the likely fragment (and not the rare case) from the table, you would reach Clue 5 and be unable to piece together a sensible structure. Going back to the “Notes” column in Table 20-1 and looking at the integration for 2H, you see that in rare cases this integration could also represent two symmetrical CH fragments. Making this substitution, then, would lead you down the road to the correct answer.
Working Problems
Here’s an example of a bare-bones problem — one where you’re given just the molecular formula and the 1H NMR and asked to solve the structure. When you’re doing problems on your own, write all the clues you find on a sheet of scratch paper, just as a detective would write down all the clues on a notepad, so that they’ll all be in front of you when you’re ready to solve the murder, er, the structure.
Example 1: Using the molecular formula and NMR to deduce the structure of a molecule
Determine the structure of a compound whose molecular formula is C8H8O2, using its 1H NMR spectrum in Figure 20-3.
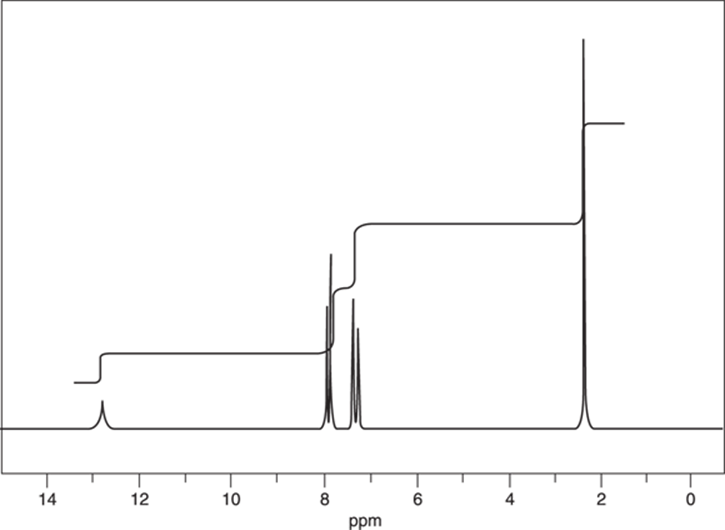
FIGURE 20-3: The NMR spectrum for a compound with molecular C8H8O2.
The way to tackle a problem like this is to work through it systematically, clue by clue.
Clue 1: Determine the degrees of unsaturation
Plugging the molecular formula, C8H8O2, into the equation gives you five degrees of unsaturation.
 Whenever you see four or more degrees of unsaturation in a molecule, the molecule likely contains a benzene ring. To confirm this, look for peaks around δ6.5 to δ8.5 ppm in the 1H NMR. Benzene rings account for four degrees of unsaturation (three for the three double bonds and one for the ring).
Whenever you see four or more degrees of unsaturation in a molecule, the molecule likely contains a benzene ring. To confirm this, look for peaks around δ6.5 to δ8.5 ppm in the 1H NMR. Benzene rings account for four degrees of unsaturation (three for the three double bonds and one for the ring).
Clue 2: Determine the major functional groups
In this problem, you’re not given an IR spectrum to determine the functional groups. However, you can still look at the NMR to see if any peaks indicative of functional groups are present. The three biggies to look for in the 1H NMR are
· Carboxylic acid protons around δ12 ppm (short, fat peaks)
· Aldehyde protons around δ10 ppm
· Aromatic protons between δ6.5 ppm and δ8.5 ppm
Only rarely (rarely, that is, in problems seen in an undergraduate class) do any other types of protons absorb in these regions.
In the example in Figure 20-3, you have both a carboxylic acid (the short, fat peak around δ13 ppm), and a benzene ring (the two sets of peaks between δ7 ppm and δ8 ppm), which you would have suspected earlier from the number of degrees of unsaturation. (For a list of chemical shifts in NMR produced by common functional groups, see Chapter 19.) Write down those two portions of the molecule on your scratch paper as shown in Figure 20-4.
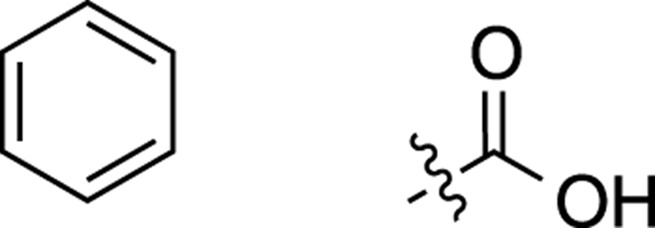
FIGURE 20-4: Carboxylic acid and a benzene ring.
 After you’ve identified from the NMR spectrum the peaks that belong to a particular functional group, put a check mark next to these peaks on the spectrum so that you don’t reassign them to fragments in Clue 4. In this example, you would put a check mark next to the benzene protons and the carboxylic acid proton, as they’ve already been assigned to fragments.
After you’ve identified from the NMR spectrum the peaks that belong to a particular functional group, put a check mark next to these peaks on the spectrum so that you don’t reassign them to fragments in Clue 4. In this example, you would put a check mark next to the benzene protons and the carboxylic acid proton, as they’ve already been assigned to fragments.
Clue 3: Determine the peak ratios
For this step, you first want to figure out the height ratios for the four peaks on the spectrum. Going from left to right across the spectrum, the ratio is 1:2:2:3. If you can’t see that just by looking at the spectrum, break out a ruler and physically measure the size of the integration lines just as I did in the preceding example and prove to yourself that this ratio is correct.
Now that you have the relative ratio (1:2:2:3), you need to convert that ratio into the absolute number of hydrogens that each peak represents. In this case, the molecular formula (C8H8O2) tells you that eight hydrogens are in the molecule. Because the relative ratio adds up to 8 (1 + 2 + 2 + 3), you don’t have to multiply the ratio by anything; the relative ratio is also the absolute ratio. You now know that the first peak represents 1H, the second and third peaks represent 2H, and the fourth peak represents 3H.
Clue 4: Finding the fragments
You’ve already determined from Clue 2 that the peak at around δ12 ppm is from a carboxylic acid proton, and that the two peaks between δ7 ppm and δ8 ppm are from a benzene ring. So, you’ve accounted for all the peaks in the NMR except for the peak at δ2.3 ppm that integrates for 3H. From Table 20-1, you find that 3H represents a –CH3 fragment. Jot that down on your scratch paper.
Now, take a closer look at the peaks for the benzene ring. From the integration, you know that the benzene has four hydrogens. This tells you that the ring must be substituted exactly twice; that is, that two non-hydrogen groups are attached to it.
 If the aromatic region has five protons, then you have a benzene ring that’s substituted once. If it has four protons, it’s substituted twice (disubstituted). If it has three protons, then it’s substituted three times (trisubstituted). If you need to, draw the structures out to prove this to yourself.
If the aromatic region has five protons, then you have a benzene ring that’s substituted once. If it has four protons, it’s substituted twice (disubstituted). If it has three protons, then it’s substituted three times (trisubstituted). If you need to, draw the structures out to prove this to yourself.
If you have a disubstituted benzene, as in this case, you must determine the orientation of the substituents relative to each other. A disubstituted benzene ring can have three different orientations. The substituents can either be ortho, meta, or para to each other (see Figure 20-5; for a review of aromatic substitution patterns see Chapter 15).
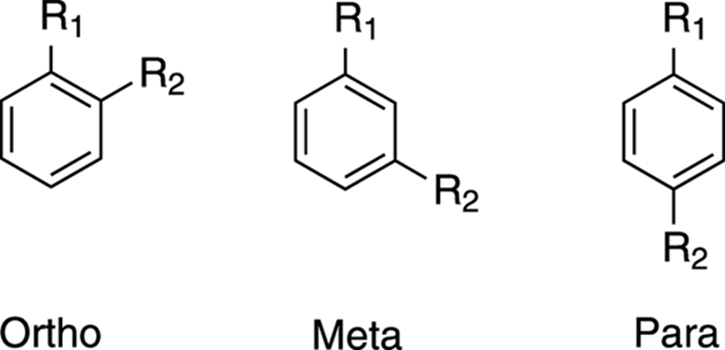
FIGURE 20-5: The three possible orientations for a disubstituted benzene ring.
Because the benzene ring in this compound contains four hydrogens, you might expect to see four peaks, one for each hydrogen. However, in the spectrum you can see only two peaks in the aromatic region. This suggests a plane of symmetry in the molecule. Looking back at Figure 20-4, only a para-substituted benzene has such a plane of symmetry — in this case, the left side of the ring is identical to the right side.
Because of this symmetry (see Figure 20-6), the two hydrogens neighboring R1 (labeled Ha) are in identical chemical environments (both are one carbon away from R1 and two carbons from R2), and the two hydrogens neighboring R2 (labeled Hb) are in identical chemical environments. Because of this symmetry, you would expect to see only two peaks in the 1H NMR that integrate for 2H each. This pattern is typical for para-substituted benzene rings.
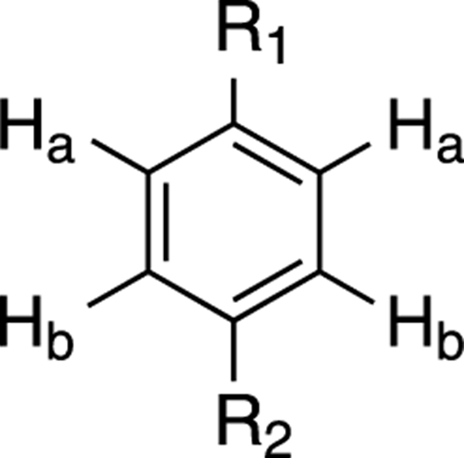
FIGURE 20-6: Seeing symmetry.
 If you can’t see the identical chemical environments from looking at the drawing, you might try building the molecule using a model so that you can turn the model over in your hands and prove to yourself that the two Ha atoms are identical, and the two Hb atoms are identical.
If you can’t see the identical chemical environments from looking at the drawing, you might try building the molecule using a model so that you can turn the model over in your hands and prove to yourself that the two Ha atoms are identical, and the two Hb atoms are identical.
Now that you know that the benzene ring has two substituents that are para to each other, you can write out all the fragments (after double-checking that the number of atoms in our fragments exactly matches the number in the molecular formula). See Figure 20-7.
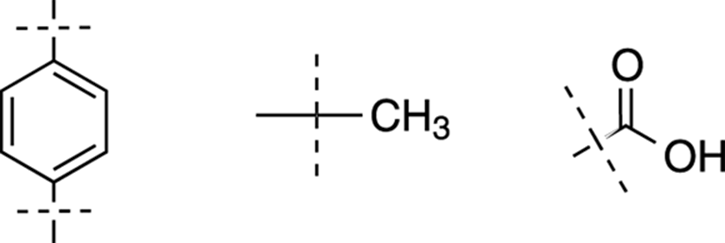
FIGURE 20-7: Structure fragments.
Clue 5: All the king’s horses: Putting the pieces together again
In this case, there’s only one possible way to put the fragments together. The proposed structure is p-toluic acid, shown in Figure 20-8.
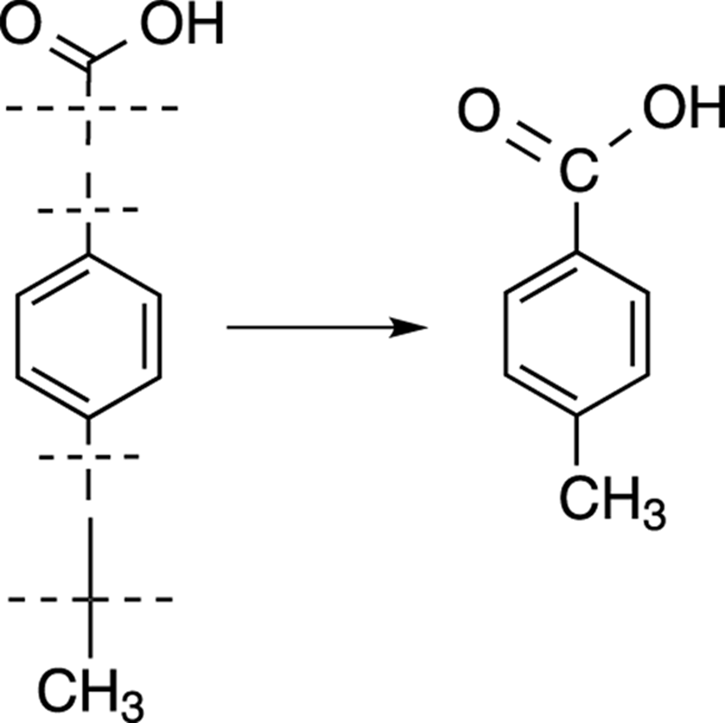
FIGURE 20-8: p-toluic acid.
Clue 6: Confirm the proposed structure
Double-checking to make sure that your proposed structure is the correct one is always a good idea. You check the structure by predicting what the NMR should look like, and then comparing it to the actual spectra to make sure that it matches. So, check this structure (see Figure 20-9):
· The methyl group should correspond to a peak that integrates for 3H and is a singlet. It would be at a slightly higher chemical shift because it’s near an aromatic ring. At δ2.3 ppm, there’s a singlet that integrates for 3H. Check!
· The aromatic ring protons should show two doublets that have a combined integration of 4H between δ6.5 and δ8.5 ppm. Check!
· The carboxylic acid should have a peak at around δ12 ppm that integrates for 1H and is a singlet. Check!
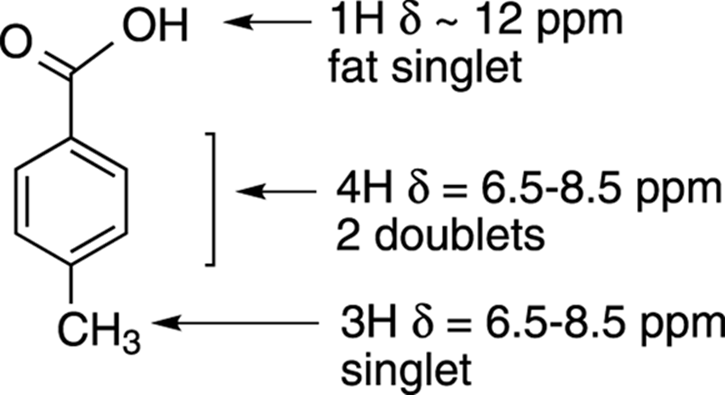
FIGURE 20-9: Checking the proposed structure.
Looks like you can lock this one up!
Example 2: Using the molecular formula, IR, and NMR to deduce the structure of a molecule
Solve the structure of compound C5H10O that has the 1H NMR and IR spectra shown in Figure 20-10.
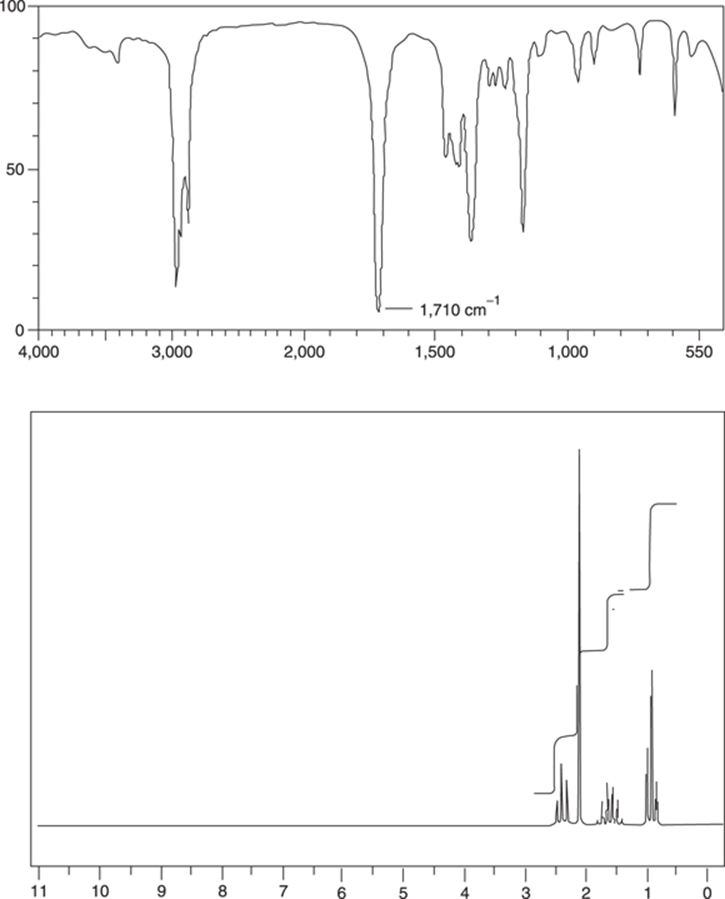
FIGURE 20-10: The IR and NMR spectra for a compound with molecular formula C5H10O.
Clue 1: Determine the degrees of unsaturation
Plugging the molecular formula, C5H10O, into the DOU equation shows that this molecule has one degree of unsaturation. This indicates that the molecule has either one double bond or one ring.
Clue 2: Determine the major functional groups
The IR spectrum shows an intense peak at 1,710 cm–1, which is characteristic of a carbonyl group. (See Chapter 5 for a listing of the functional groups containing a carbonyl group.) The small peaks between 3,400 and 3,700 cm–1 cannot be amine stretches because, according to the molecular formula, the compound doesn’t contain nitrogen, and the peak is not broad and intense enough to be caused by an alcohol (besides, the oxygen is already accounted for with the carbonyl group). This is probably just a carbonyl overtone stretch. No other peaks in the IR are very helpful. (For a list of IR peaks produced by common functional groups, see Chapter 18.) Looking in the 1H NMR for distinct peaks above δ6.5 ppm turns up nothing; the NMR spectrum shows no peaks attributable to aldehyde, aromatic, or acid functional groups. (For a list of chemical shifts in NMR produced by common functional groups, see Chapter 19.)
Clue 3: Determine the peak ratios
The NMR peaks integrate with a relative ratio of 1.0:1.5:1.0:1.5 from left to right (use a ruler if you don’t believe me). Multiplying this entire ratio by two gives a whole number relative ratio of 2:3:2:3. Adding up 2 + 3 + 2 + 3 gives 10, matching the number of hydrogens in the molecular formula. So, you don’t need to multiply the relative ratio by anything to get the actual number of hydrogens. Therefore, the integrations for the three peaks from left to right are 2H, 3H, 2H, and 3H.
Clue 4: Finding the fragments
From Table 20-1, the most likely fragments for 2H, 3H, 2H, and 3H are CH2, CH3, CH2, and CH3, respectively. Adding up all the atoms from these fragments to see if the number of atoms matches the molecular formula shows that one carbon and one oxygen atom are missing. Additionally, from Clue 1 you know that this molecule has one degree of unsaturation; this unsaturation could be accounted for by the double bond in the carbonyl seen in the IR spectrum (refer to Clue 2). Carbonyl groups are found in aldehydes, ketones, esters, and carboxylic acids. Here, however, esters and carboxylic acids can be eliminated because the molecular formula shows only a single oxygen. Because the NMR spectrum doesn’t contain an aldehyde peak (aldehyde protons are seen around δ10 ppm) and the IR spectrum doesn’t show an aldehyde C-H stretch in the at 2,700 cm–1 (see Chapter 15), the carbonyl group is a ketone.
Clue 5: And all the king’s men: Putting the pieces together again
Because the carbonyl is a ketone, the fragments can only be pieced together in two ways, as shown in Figure 20-11. Note that one of these ways gives a symmetric molecule (possible compound A, where the carbonyl group is in the center of the molecule) while the other way gives a molecule that’s not symmetric (possible compound B, where the carbonyl group is on one side of the molecule).
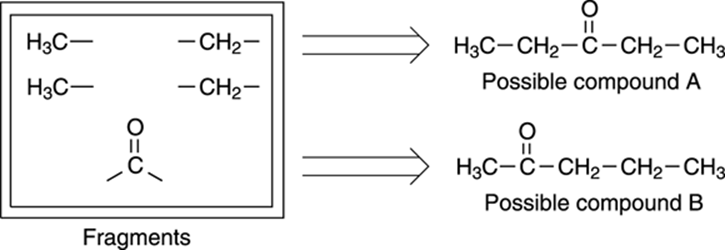
FIGURE 20-11: The two possible structures.
Now that you’ve narrowed down the choices to two, you want to check to see which one is the most consistent with the peak splitting and the chemical shift. Try possible compound A first.
You can easily rule out this compound because it has a center of symmetry — that is, the left-hand side of the molecule is identical to the right-hand side. Therefore, you would expect to see just two peaks in the 1H NMR for this compound rather than the four you see, because both methyl (CH3) groups would be in identical chemical environments, and both methylene (CH2) groups would also be in identical chemical environments.
Clue 6: Confirm the proposed structure
Now check possible compound B (see Figure 20-12).
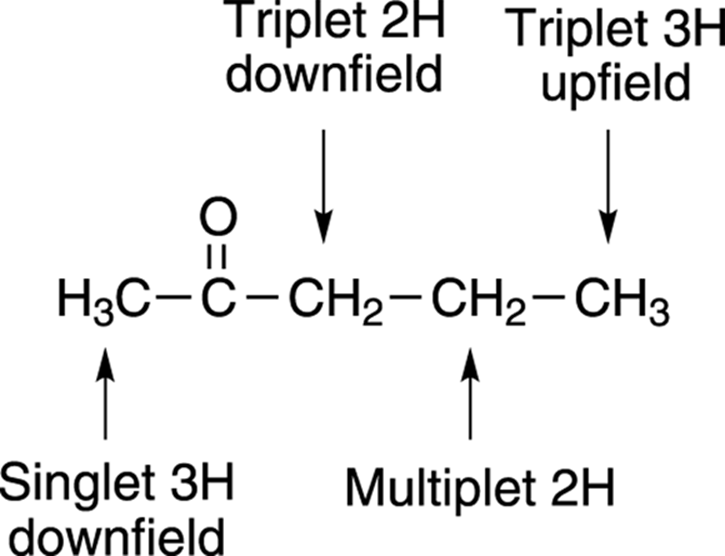
FIGURE 20-12: Confirming the structure.
This compound has no symmetry, so you go carbon atom by carbon atom to check the structure.
· The far-left methyl group has no adjacent hydrogens. Therefore, it would be a singlet and integrate for 3H. Because it’s next to the electron-withdrawing carbonyl group, you would expect it to be shifted downfield (higher ppm). Sure enough, the NMR shows a singlet at δ2.1 ppm that integrates for 3H. Check!
· The methylene (CH2) to the immediate right of the carbonyl group would be expected to be a triplet, because it’s adjacent to two hydrogens. This triplet would integrate for 2H and would be expected to be at a higher chemical shift because it’s next to the electron-withdrawing carbonyl group. Sure enough, at δ2.5 ppm there’s a triplet that integrates for 2H. Check!
· The methylene group farther to the right would be expected to be a multiplet because it’s adjacent to five hydrogens (it would be approximately a sextet, although it could split into as many as 12 peaks; see Chapter 19), and would integrate for 2H. Sure enough, at δ1.5 ppm the NMR spectrum shows a multiplet (that appears to be a sextet) that integrates for 2H. Check!
· The methyl to the far right would be expected to be a triplet because it’s adjacent to two hydrogens and would integrate for 3H. This triplet would be expected to be farther upfield (lower ppm) than the other peaks because it’s the farthest away from the electron-withdrawing carbonyl group. Sure enough, at δ0.9 ppm the NMR spectrum shows a triplet that integrates for 3H. Check!
Everything matches, so you can cuff this one up.
Three Common Mistakes in NMR Problem Solving
In NMR problems, there’s so much information to be considered that making a mistake is easy. But here are three common mistakes that you can try to avoid when solving NMR spectroscopy problems.
Mistake #1: Trying to determine a structure from the chemical shift
Chemical shift regions overlap, so attempting to determine all the fragments of a molecule solely on the basis of chemical shift is almost always a mistake. A few fragments can be determined in some cases — aldehydes, carboxylic acids, and aromatic ring fragments are often given away by the chemical shift — but most fragments cannot. A proton at δ2.5 ppm could be a proton adjacent to a carbonyl group; it could also be a proton on a carbon neighboring a benzene ring or even a proton next to a double bond. It would be impossible to tell the difference among these three possibilities simply by looking at the chemical shift.
Yes, there are chemical shift tables big enough to choke a small beluga whale, but your time will be much better spent understanding the general ideas behind the chemical shift than memorizing all the values for every conceivable proton that you might encounter. Such mindless memorization is a fruitless task anyway. You might know that a proton next to a chlorine will have a chemical shift of δ3 to δ4 ppm, but what about a proton next to two chlorines? Or what about a proton next to two chlorines and a carbonyl group? Too many possibilities exist to simply memorize them all.
A better approach is to learn the ranges of chemical shifts for protons next to the common functional groups (such as those found in Chapter 19; many professors are kind enough to give you a table that they want you to learn), and then understand the idea behind the chemical shift. If you understand that protons next to groups that have a local magnetic field (like double bonds, triple bonds, and aromatic rings) will have a higher chemical shift than isolated protons, and that protons next to electronegative elements will have a higher chemical shift than those that are far from electronegative elements, you’ll be in pretty good shape. If you understand these concepts, you won’t need a table to know that a proton next to two chlorines will have a higher chemical shift than a proton next to one chlorine. This understanding will give you greater flexibility for solving NMR problems.
Mistake #2: Starting with coupling
Unless you really know what you’re doing, trying to solve a structure by looking first at the coupling will result in fistfuls of your pulled-out hair and several empty bottles of aspirin. Unlike integration, coupling doesn’t refer to how many hydrogens (protons) a peak represents; instead, it indicates how many proton neighbors a certain hydrogen has. Because of that fact, determining the pieces of the molecule using the coupling is much more difficult than using the integrations.
Think of this process as analogous to the work of a census taker going house to house gathering information about the number of residents in each house. If you were taking the census, what would make your job easier: having people tell you how many people lived in the house you were visiting, or having people tell you how many people lived next door to the house you were visiting? Doing the census would be easier if the residents told you how many people lived in that particular house, right? To continue with this analogy, the integration tells you how many people (hydrogens) live in the house (the specific carbon); the coupling tells you how many people (hydrogens) live next door.
Another drawback to starting with coupling is that coupling has so many nuances (many of which are beyond the scope of undergraduate-level NMR), and peaks are not always split into the standard-fare singlets, doublets, triplets, and so on. Long-range coupling (which I don’t cover in this book) can complicate things, as can the presence of chiral centers (refer to Chapter 6). Protons that are next to nonequivalent hydrogens can give some pretty complicated splitting patterns, too. Often, you just get a haystack — a big jumble of many peaks clumped together that are hard to resolve. Sometimes the coupling will also be hard to see unless your professor was kind and magnified the peaks for you. Because of all these factors, coupling is most useful for determining the connectivity of the fragments and is less useful in determining the identity of the fragments themselves.
Mistake #3: Confusing integration with coupling
Remember that the integration tells you the relative number of protons a peak represents, but that coupling refers to how many neighbors those protons see. Therefore, the n + 1 rule applies only to coupling, not to integration, and has nothing to do with how many protons a peak represents. A multiplet can integrate for 1H just as a singlet can integrate for 9H. Coupling and integration are independent of one another.