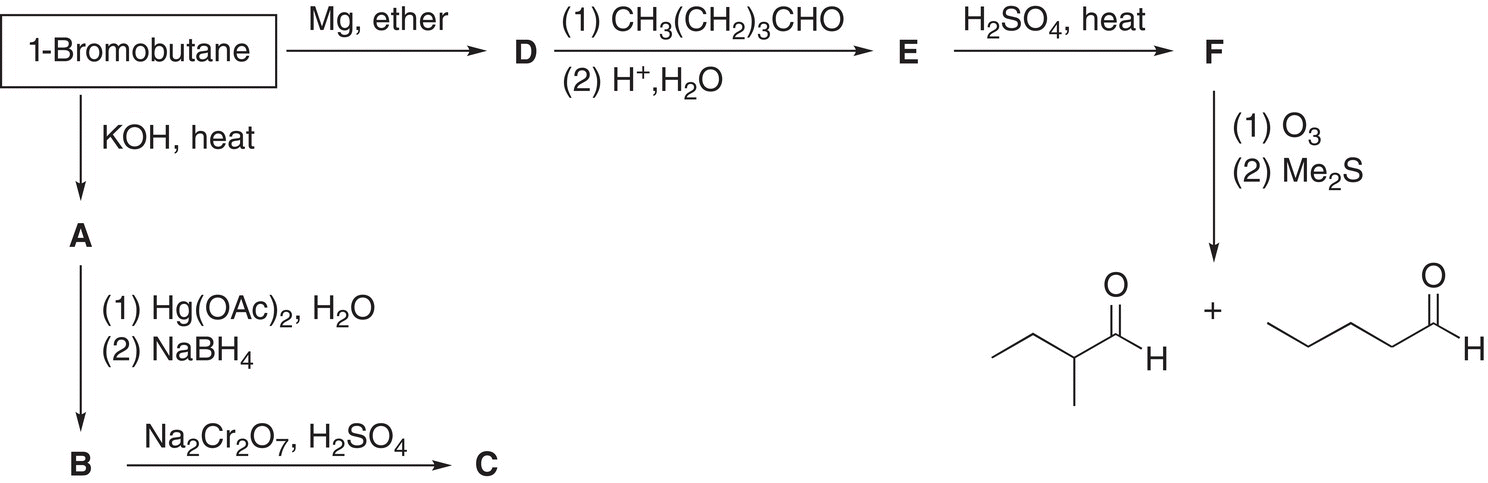Organic Chemistry: Concepts and Applications - Headley Allan D. 2020
Elimination Reactions of Organic Chemistry
End of Chapter Problems
1. 12.10 Give the major organic products of each of the following reactions.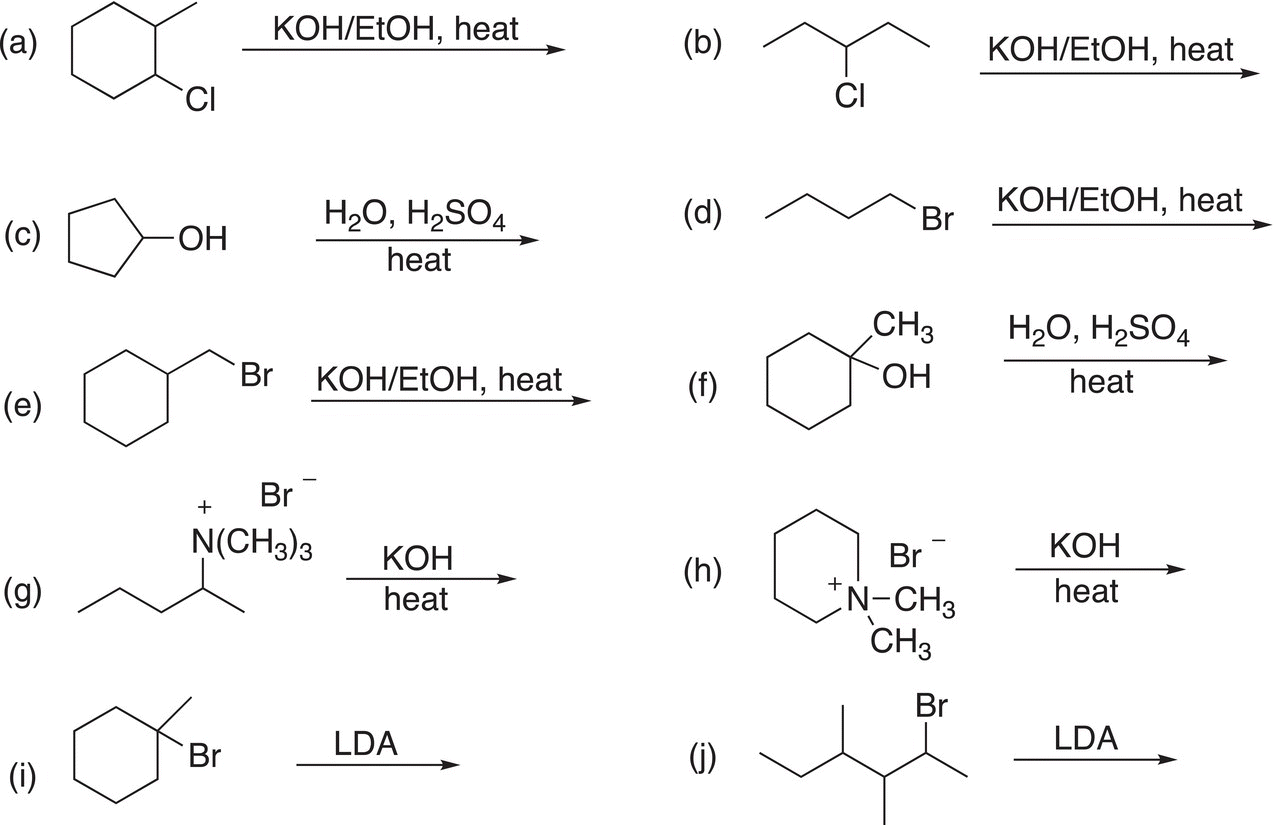
2. 12.11 Give the major organic products for the reaction of 3-methyl-2-pentene with each of the following reagents
a) (i) Hg(OAc)2/H2O ii) NaBH4 |
b) (i) BH3 ii) H2O2, OH− |
c) KMnO4, room temp. |
d) Br2/H2O |
e) i) O3 ii) Me2S |
f) KMnO4, heat |
3. 12.12 What is the best reagent(s) to carry out the following transformations?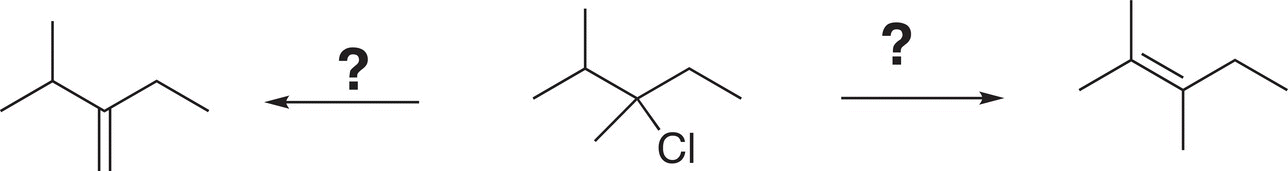
4. 12.13 Using arrow-pushing formulism to indicate electron movement, propose a reasonable mechanism to explain the reaction shown below.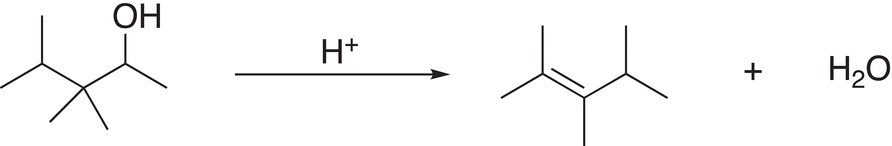
5. 12.14 Using arrow-pushing formulism to indicate electron movement, propose a reasonable mechanism to explain the reaction shown below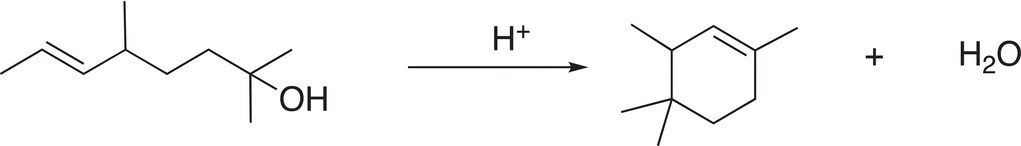
6. 12.15 Using a multistep approach, show how to carry out the following transformations.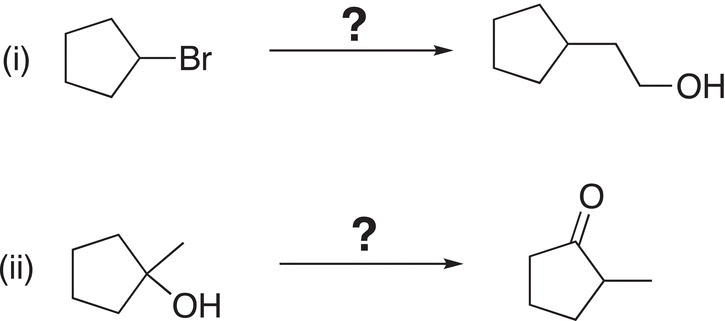
7. 12.16 Predict the pinacol rearranged product for the following reaction and propose a mechanism to explain your answer.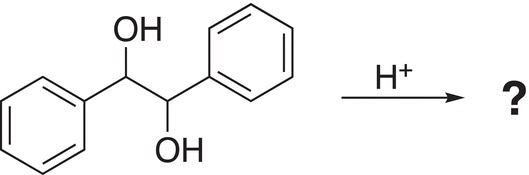
8. 12.17 Briefly explain the outcome of the reactions shown below.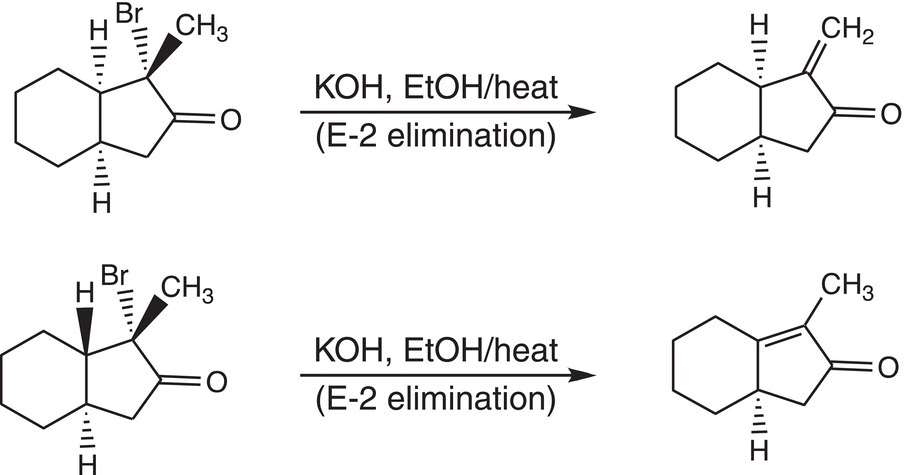
9. 12.18 Complete the following reactions by giving the structures of the major organic product. Indicate stereochemistry where appropriate.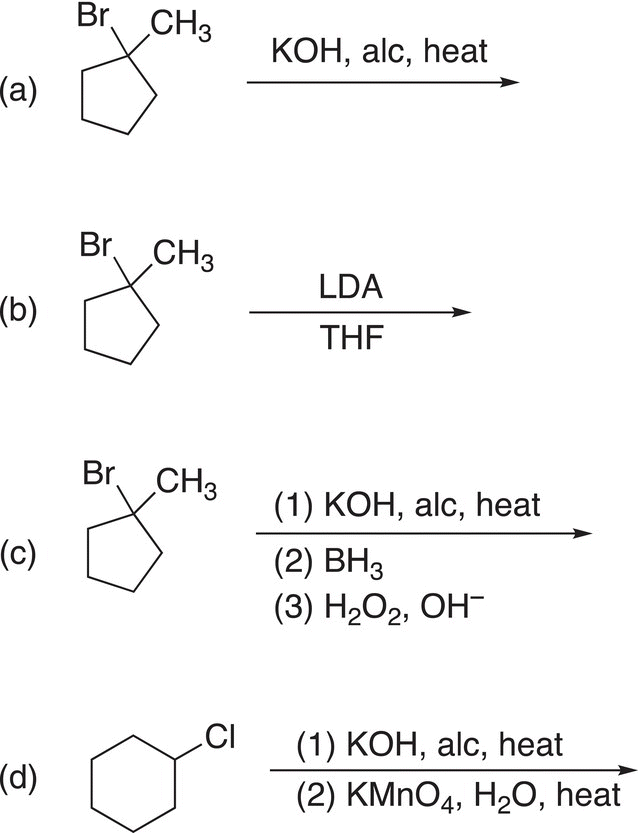
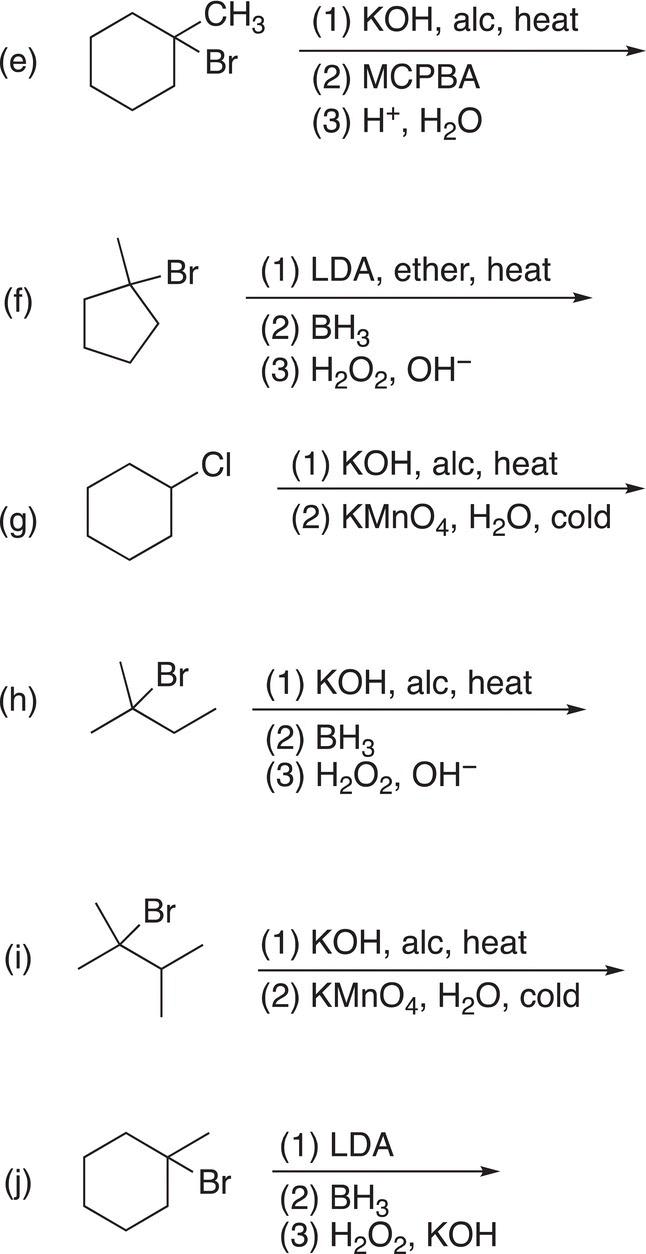
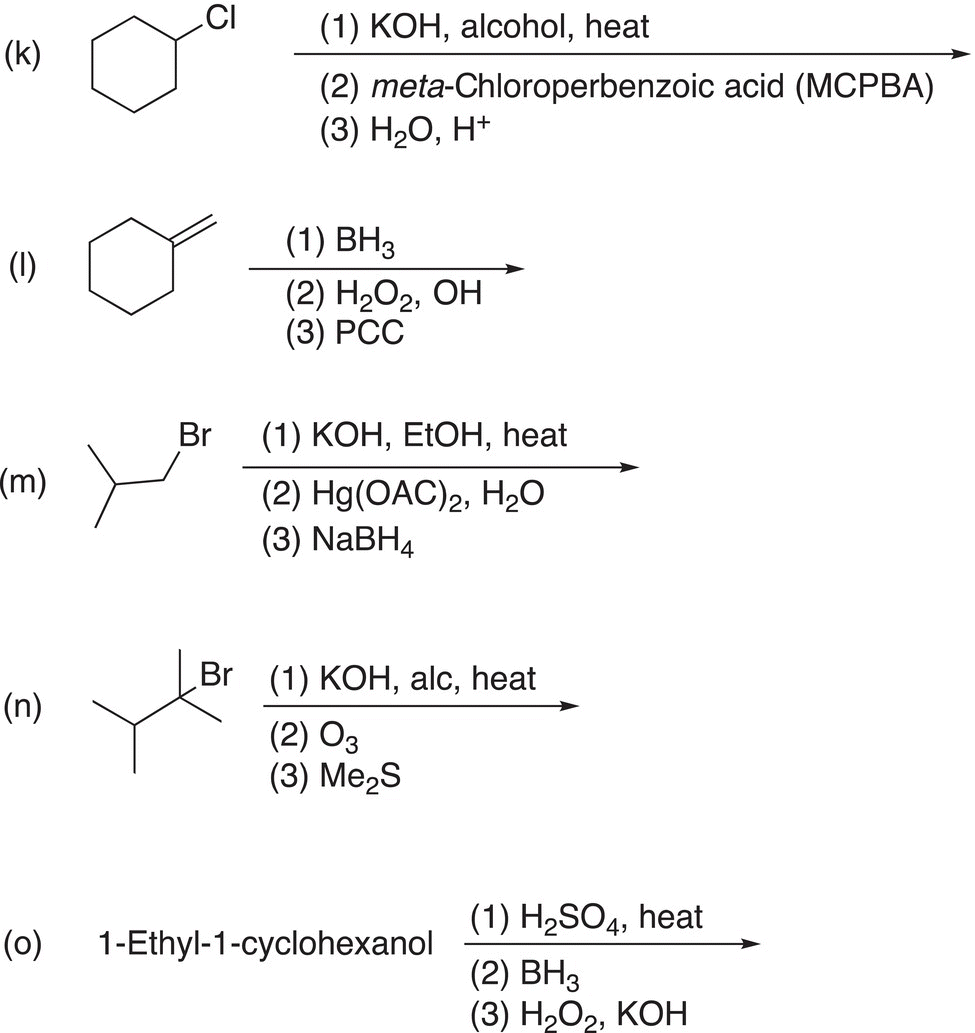
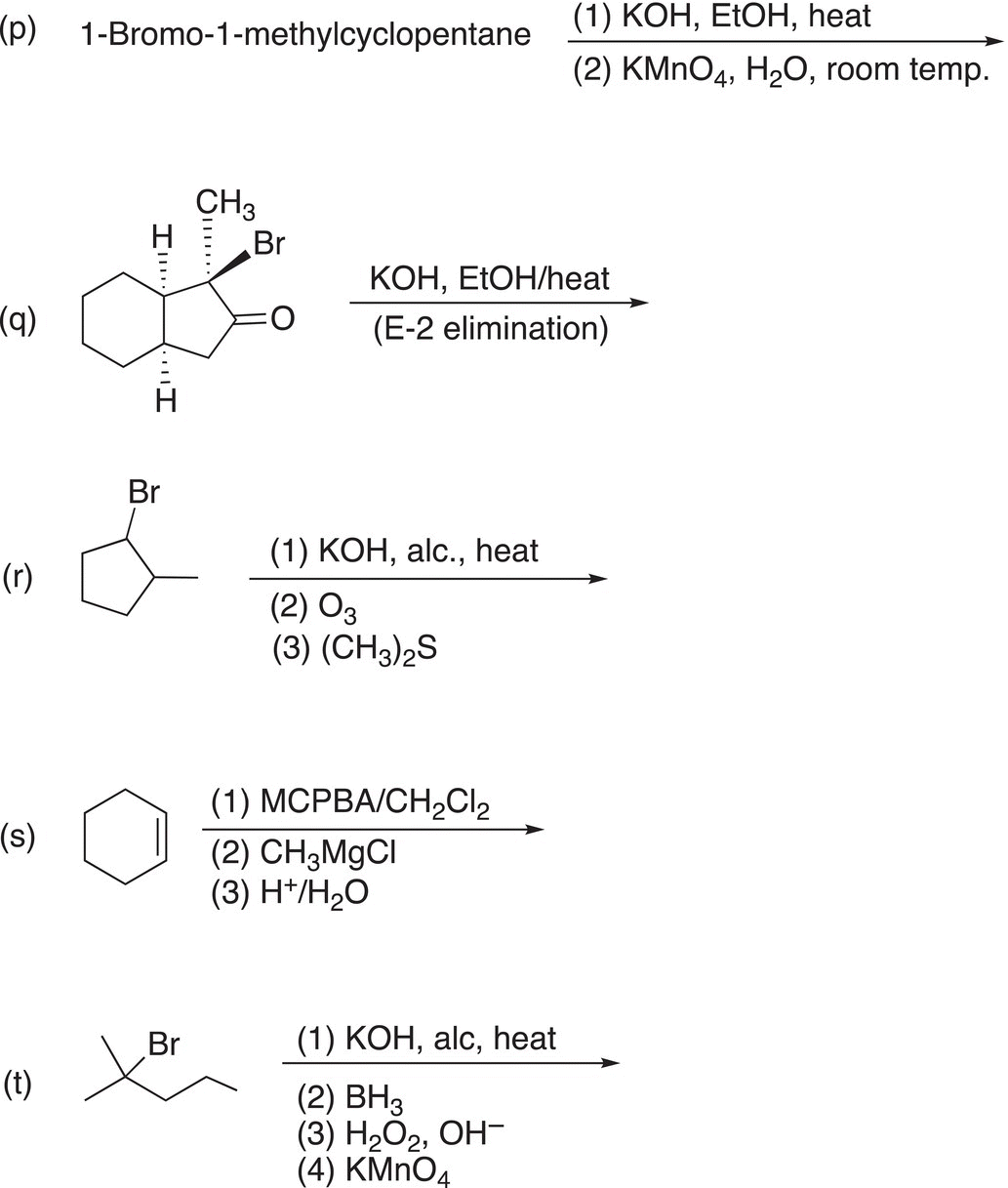
10. 12.19 Treatment of compound A (C7H13Br) with sodium ethoxide (a base) in ethanol at 60 °C converts A into B (C7H12). Ozonolysis of B gives C (shown below) as the only product. Deduce the structures of A and B.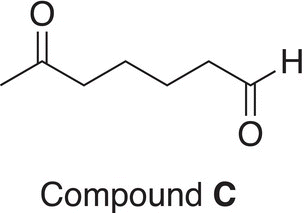
11. 12.20 Give the structures of compounds A through D for the reaction scheme shown below.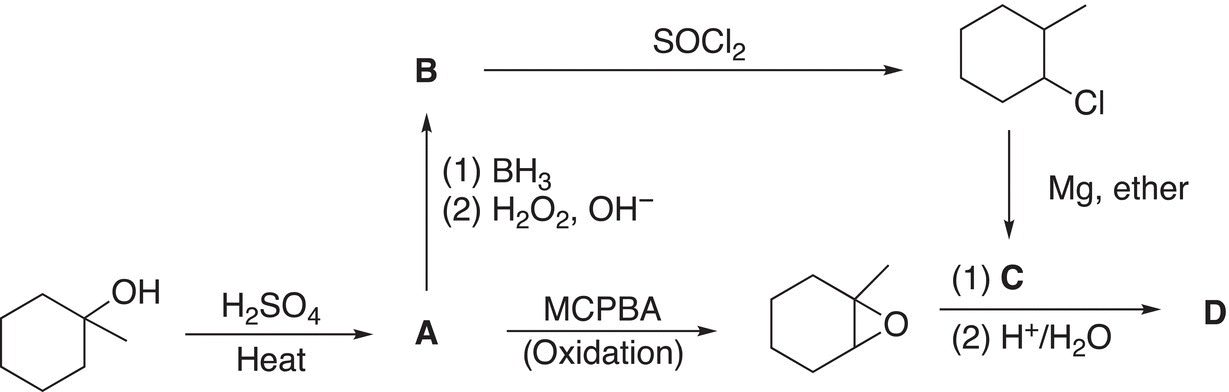
12. 12.21 Give the structural formulas for the lettered compounds of the following sequence of reactions.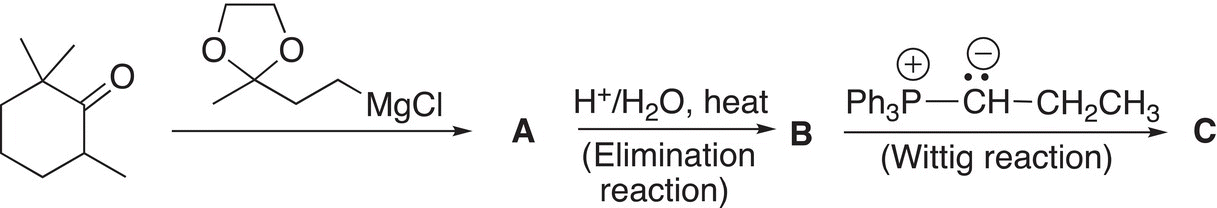
13. 12.22 Give the structures for the lettered compounds in the reaction scheme below.