Cracking the SAT Chemistry Subject Test
FORMULA CHEAT SHEET
• density = ![]()
Heat
• q = mcΔT
q = Heat
m = mass
c = specific heat
ΔT = Temperature change
Spontaneity
• ΔG = ΔH – TΔS
ΔG < 0 means that a reaction is spontaneous at this temperature

• ΔT = kmi
Freezing Point Depression
& Boiling Point Depression
i: whole number equaling the number of particles a substance dissolves into
k: solvent constant
m: molality (NOT molarity)
molarity = ![]()
molality = ![]()
mole fraction = ![]()
Ideal Gas Law
• PV = nRT
P = pressure in atm (or mmHg or torr)
V = volume in liters
n = number of moles of gas particles in the container
R = the ideal gas constant
T = temperature in Kelvin
To calculate the ideal gas constant, use the following formula:
R = .08 ![]()
Conversions
• Pressure:
1 ATM = 760 torr = 760 mmHg
• Energy (Heat):
1 Calorie = 1000 calories = 4000 joules
• Temperature:
0°C = 273 Kelvin
25°C = 298 K
Facts to Know
• To find the mass of a mole of a substance, multiply the formula weight by the number of moles.
• 1 mol of gas at STP occupies 22.4 L of volume.
Solubility
• All alkali and ammonium (NH4+) compounds are soluble. The alkali metals are included in group IA (Li+, NA+, K+, Rb+, Cs+).
• All acetates (CH3COO−), chlorates (), nitrates (), and perchlorates () are soluble.
• Pbn+, Hgn+, and Agn+ salts are insoluble, unless they are paired with one of the anions from the previous rule.
Oxidation Rules
1. When oxygen is in a compound, its oxidation state is usually –2 (it has been reduced). One important exception is oxygen in a peroxide such as hydrogen peroxide (H2O2). In a peroxide, oxygen has an oxidation state of –1.
2. When an alkali metal (Li, Na, etc.) is involved in a compound, its oxidation state is always +1 (it's been oxidized).
3. When an alkaline earth metal (Be, Mg, etc.) is involved in a compound, its oxidation state is +2.
4. When a halogen (F, Cl, etc.) is involved in a compound, its oxidation state is often –1. The oxidation state of fluorine in a compound is always –1.
5. When hydrogen is combined with a nonmetal, its oxidation state is +1. When hydrogen is combined with a metal, its oxidation state is –1.
6. In any compound, the sum of all oxidation states is zero.
Constants
• Avogadro's number: 6 × 1023 molecules/mole
Prefixes
Prefixes for Organics
Meth - 1
Eth - 2
Prop - 3
But - 4
Pent - 5
Hex - 6
Hep - 7
Oct - 8
Non - 9
Dec - 10
Prefixes for Scientific Notation
nano n 10-9
micro 10-6
milli m 10-3
centi c 10-2
kilo k 10-3
mega M 106
Electrochemistry
OIL
Oxidation Is Losing Electrons
RIG
Reduction Is Gaining Electrons
AN OX
Oxidation Occurs at Anode
RED CAT
Reduction Occurs at Cathode
• Galvanic Cell: E°cell > 0 means reaction is spontaneous and energy is released (battery).
• Electrolytic Cell: E°cell < 0 means the reaction is NOT spontaneous and requires a voltage source (electro plating).
Equilibrium
• 
Periodic Trends
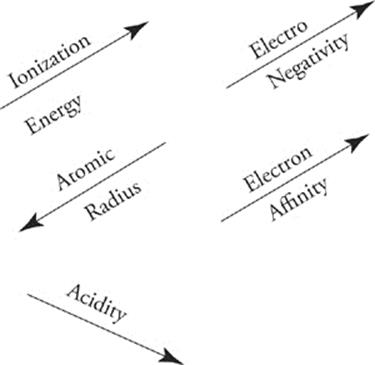
Acid-Base
pH = pKa + log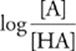
pH = –log10[H+]
pH > 7: Basic
pH = 7: Neutral
pH < 7: Acidic
Strong Acids:
HCI, HI, H2SO4, HBr, HClO4, HNO3
Strong Bases:
All Group I Hydroxides