MCAT Biology and Biochemistry: New for MCAT 2015 (2014)
Chapter 9. The Nervous and Endocrine Systems
The nervous and endocrine systems are presented in the same chapter since their functions are related: They both provide communication, integrating and coordinating the activities of the tissues and organs of the body. The means of communication by the two systems are quite different (although complementary) in many ways. The nervous system communicates through electrochemical signals (action potentials), while the endocrine system uses chemical messengers carried in the blood (hormones). The nervous system in general regulates rapid responses such as those of skeletal muscle or smooth muscle, while the endocrine system takes longer to have an effect and regulates longer-term responses such as metabolism and homeostasis. The two systems are interconnected, with two of the primary endocrine glands—the pituitary and the adrenals—regulated by the nervous system, and with the endocrine system feeding back to modulate the nervous system.
9.1 NEURONAL STRUCTURE AND FUNCTION
Neurons are specialized cells that transmit and process information from one part of the body to another. This information takes the form of electrochemical impulses known as action potentials. The action potential is a localized area of depolarization of the plasma membrane that travels in a wave-like manner along an axon. When an action potential reaches the end of an axon at a synapse, the signal is transformed into a chemical signal with the release of neurotransmitter into the synaptic cleft, a process called synaptic transmission (Section 9.2). The information of many synapses feeding into a neuron is integrated to determine whether that neuron will in turn fire an action potential. In this way the action of many individual neurons is integrated to work together in the nervous system as a whole.
Structure of the Neuron
The basic functional and structural unit of the nervous system is the neuron (Figure 1). The structure of these cells is highly specialized to transmit and process action potentials, the electrochemical signals of the nervous system (Figure 3). Neurons have a central cell body, the soma, which contains the nucleus and is where most of the biosynthetic activity of the cell takes place. Slender projections, termed axons and dendrites, extend from the cell body. Neurons have only one axon (as long as a meter in some cases), but most possess many dendrites. Neurons with one dendrite are termed bipolar; those with many dendrites are multipolar. Neurons generally carry action potentials in one direction, with dendrites receiving signals and axons carrying action potentials away from the cell body. Axons can branch multiple times and terminate in synaptic knobs that form connections with target cells. When action potentials travel down an axon and reach the synaptic knob, chemical messengers are released and travel across a very small gap called the synaptic cleft to the target cell. The nature of the action potential and the transmission of signals across the synaptic cleft are key aspects of nervous system function. [In Figure 1, in what direction does an action potential travel in the axon shown?1 What’s the difference between a neuron and a nerve?2]
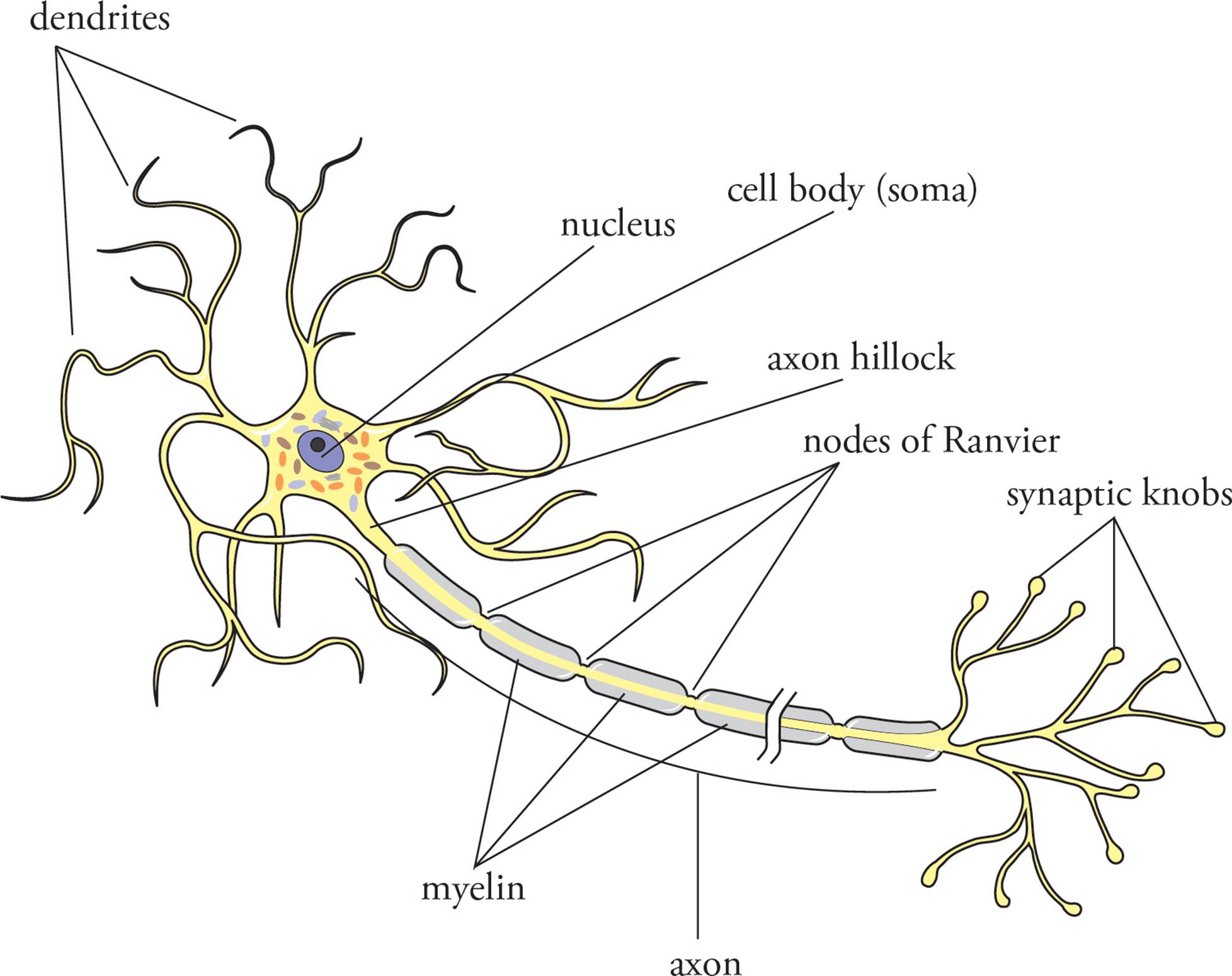
Figure 1 A Multipolar Neuron
• A motor protein called kinesin is one of several different proteins that drive movement of vesicles and organelles along microtubules in axons. Kinesin specifically drives anterograde movement (movement from the soma toward the axon terminus). If a kinesin inhibitor is added to neurons in culture, what is the likely result?3
A) Spontaneous action potentials
B) Cell division
C) Accumulation of material in the synaptic knob
D) Atrophy of axons
The Action Potential
The Resting Membrane Potential
The resting membrane potential is an electric potential across the plasma membrane of approximately –70 millivolts (mV), with the interior of the cell negatively charged with respect to the exterior of the cell.
Two primary membrane proteins are required to establish the resting membrane potential: the Na+/K+ ATPase and the potassium leak channels. The Na+/K+ ATPase pumps three sodium ions out of the cell and two potassium ions into the cell with the hydrolysis of one ATP molecule. [What form of transport is carried out by the Na+/K+ ATPase?4] The result is a sodium gradient with high sodium outside of the cell and a potassium gradient with high potassium inside the cell. Leak channels are channels that are open all the time, and that simply allow ions to “leak” across the membrane according to their gradient. Potassium leak channels allow potassium, but no other ions, to flow down their gradient out of the cell. The combined loss of many positive ions through Na+/K+ ATPases and the potassium leak channels leaves the interior of the cell with a net negative charge, approximately 70 mV more negative than the exterior of the cell; this difference is the resting membrane potential. Note that there are very few sodium leak channels in the membrane (the ratio of K+ leak channels to Na+ leak channels is about 100:1), so the cell membrane is virtually impermeable to sodium.
• Are neurons the only cells with a resting membrane potential?5
• If the potassium leak channels are blocked, what will happen to the membrane potential?6
• What would happen to the membrane potential if sodium ions were allowed to flow down their concentration gradient?7
The resting membrane potential establishes a negative charge along the interior of axons (along with the rest of the neuronal interior). Thus, the cells can be described as polarized; negative on the inside and positive on the outside. An action potential is a disturbance in this membrane potential, a wave of depolarization of the plasma membrane that travels along an axon. Depolarization is a change in the membrane potential from the resting membrane potential of approximately –70 mV to a less negative, or even positive, potential. After depolarization, repolarization returns the membrane potential to normal. The change in membrane potential during passage of an action potential is caused by movement of ions into and out of the neuron through ion channels. The action potential is therefore not strictly an electrical impulse, like electrons moving in a copper telephone wire, but an electrochemicalimpulse.
Depolarization
Key proteins in the propagation of action potentials are the voltage-gated sodium channels located in the plasma membrane of the axon. In response to a change in the membrane potential, these ion channels open to allow sodium ions to flow down their gradient into the cell and depolarize that section of membrane. [What is the effect of opening the voltage-gated sodium channels on the membrane potential?8] These channels are opened by depolarization of the membrane from the resting potential of –70 mV to a threshold potential of approximately –50 mV. Once this threshold is reached, the channels are opened fully, but below the threshold they are closed and do not allow the passage of any ions through the channel. When the channels open, sodium flows into the cell, down its concentration gradient, depolarizing that section of the membrane to about +35 mV before inactivating. Some of the sodium ions flow down the interior of the axon, slightly depolarizing the neighboring section of membrane. When the depolarization in the next section of membrane reaches threshold, those voltage-gated sodium channels open as well, passing the depolarization down the axon (Figure 2). [If an action potential starts at one end of an axon, can it run out of energy and not reach the other end?9]

Figure 2 The Action Potential is a Wave of Membrane Depolarization
• Which one of the following can cause the interior of the neuron to have a momentary positive charge?10
A) Opening of potassium leak channels
B) Activity of the Na+/K+ ATPase
C) Opening of voltage-gated sodium channels
D) Opening of voltage-gated potassium channels
• Given the above description, which of the following best describes the response of voltage-gated sodium channels to a membrane depolarization from –70 mV to –60 mV?11
A) All of the channels open fully.
B) 50% of the channels open fully.
C) All of the channels open 50%.
D) None of the channels open.
Repolarization
With the opening of voltage-gated sodium channels, sodium flows into the cell and depolarizes the membrane to positive values. As the wave of depolarization passes through a region of membrane, however, the membrane does not remain depolarized (Figure 3).
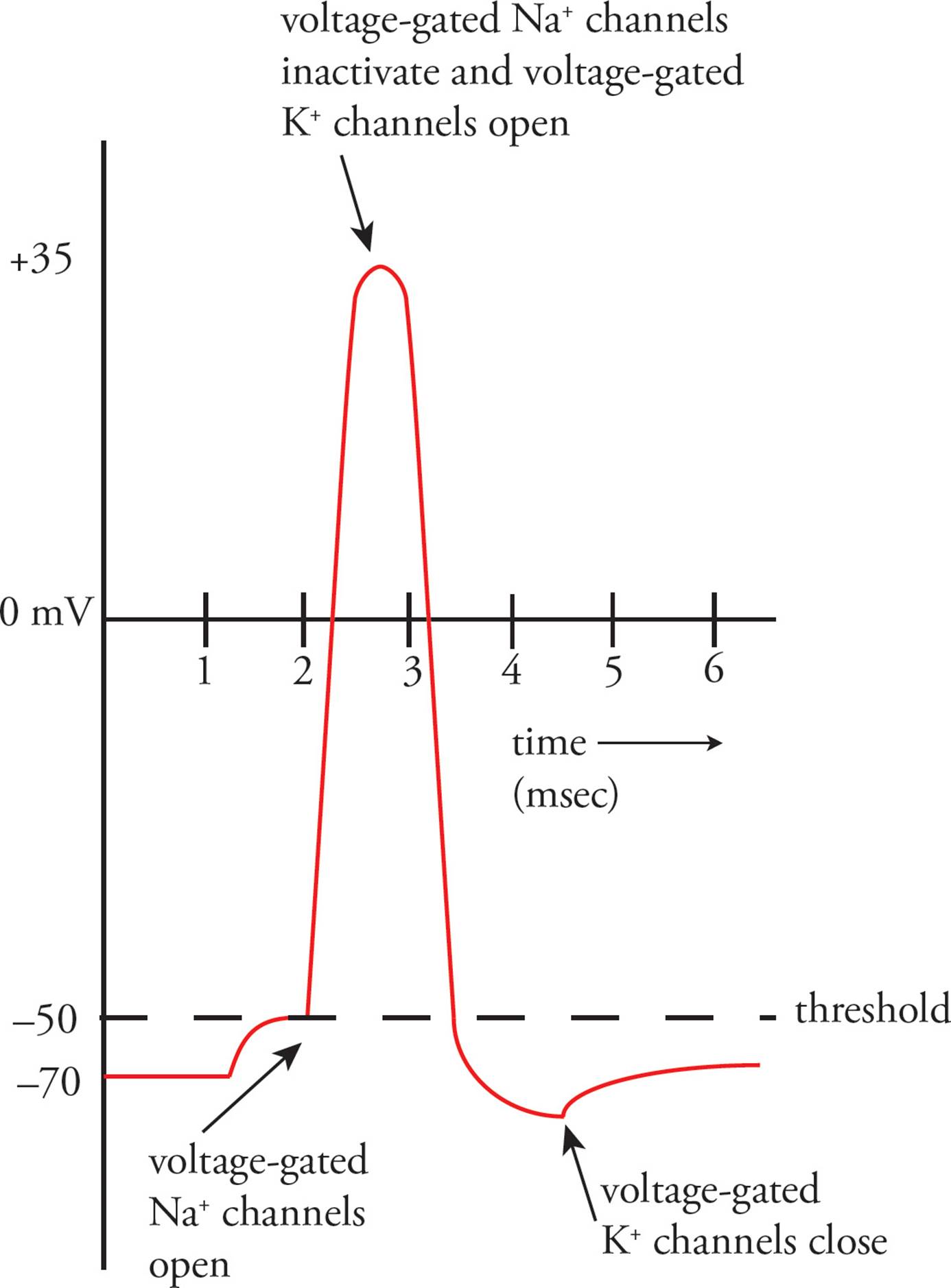
Figure 3 The Action Potential at a Single Location
After depolarization, the membrane is repolarized, re-establishing the original resting membrane potential. A number of factors combine to produce this effect:
1) Voltage-gated sodium channels inactivate very quickly after they open, shutting off the flow of sodium into the cell. The channels remain inactivated until the membrane potential nears resting values again.
2) Voltage-gated potassium channels open more slowly than the voltage-gated sodium channels and stay open longer. Voltage-gated potassium channels open in response to membrane depolarization. As potassium leaves the cell down its concentration gradient, the membrane potential returns to negative values, actually overshooting the resting potential by about 20 mV (to about –90 mV). At this point the voltage-gated potassium channels close.
3) Potassium leak channels and the Na+/K+ ATPase continue to function (as they always do) to bring the membrane back to resting potential. These factors alone would repolarize the membrane potential even without the voltage-gated potassium channels, but it would take a lot longer.
• If a toxin prevents voltage-gated sodium channels from closing, which of the following will occur?12
I. Voltage-gated potassium channels will open but not close.
II. The membrane will not repolarize to the normal resting membrane potential.
III. The Na+/K+ ATPase will be inactivated.
A) I only
B) II only
C) I and II only
D) II and III only
Saltatory Conduction
The axons of many neurons are wrapped in an insulating sheath called myelin (Figure 4). The myelin sheath is not created by the neuron itself, but by cells called Schwann cells13, a type of glial cell, that exist in conjunction with neurons, wrapping layers of specialized membrane around the axons. No ions can enter or exit a neuron where the axonal membrane is covered with myelin. [Would an axon be able to conduct action potentials if its entire length were wrapped in myelin?14] There is no membrane depolarization and no voltage-gated sodium channels in regions of the axonal plasma membrane that are wrapped in myelin. There are periodic gaps in the myelin sheath however, called nodes of Ranvier (Figures 1, 4, and 5). Voltage-gated sodium and potassium channels are concentrated in the nodes of Ranvier in myelinated axons. Rather than impeding action potentials, the myelin sheath dramatically speeds the movement of action potentials by forcing the action potential to jump from node to node. This rapid jumping conduction in myelinated axons is termed saltatory conduction.
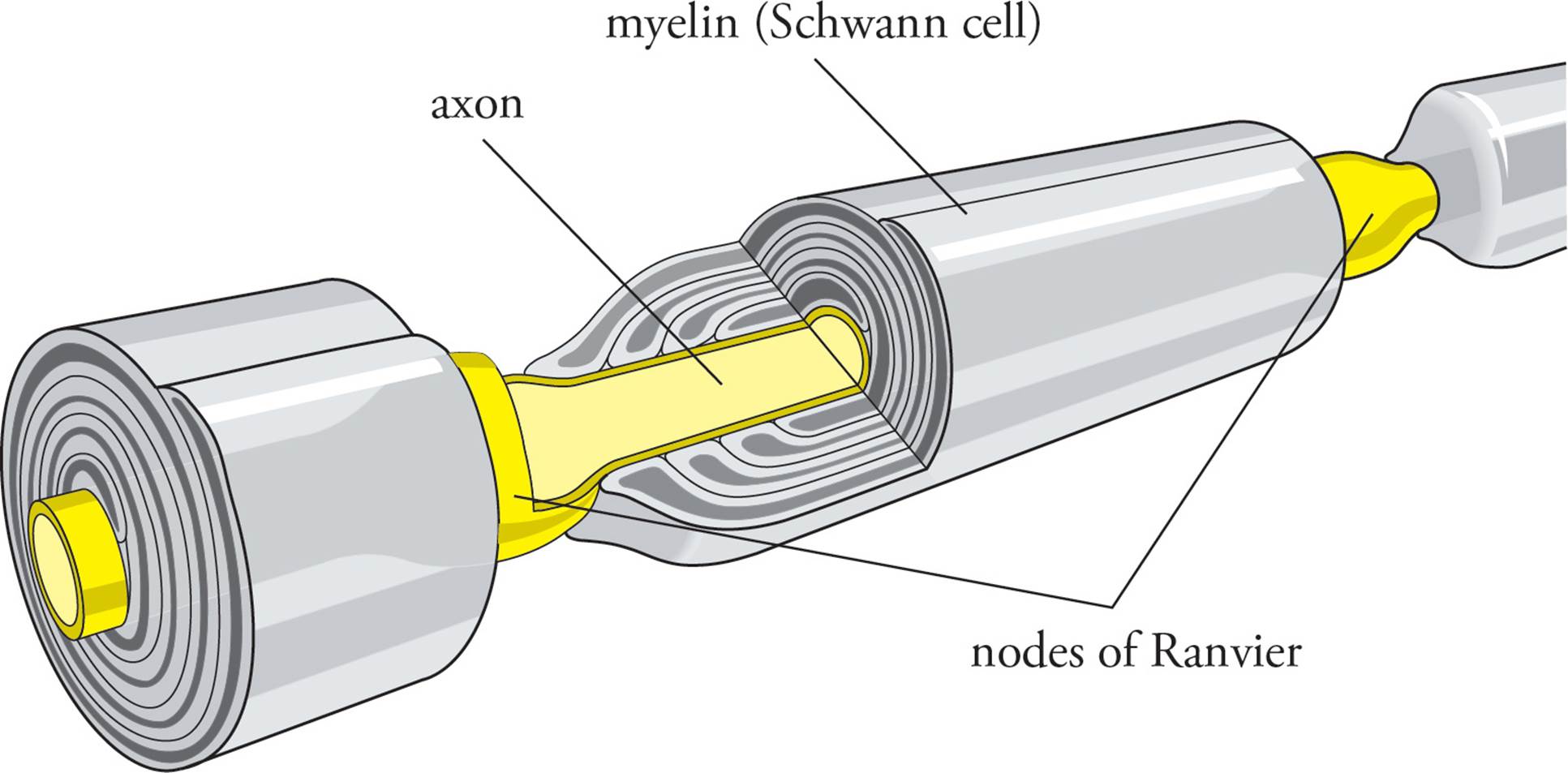
Figure 4 A Schwann Cell Wrapping an Axon with Myelin
• Which one of the following is true concerning myelinated and unmyelinated axons?15
A) The amount of energy consumed by the Na+/K+ ATPase is much less in myelinated axons than in unmyelinated axons.
B) Myelinated axons can conduct many more action potentials per second than can unmyelinated axons.
C) The size of action potential depolarization is much greater in myelinated axons than in unmyelinated axons.
D) Voltage-gated potassium channels do not play a role in repolarization in unmyelinated axons.
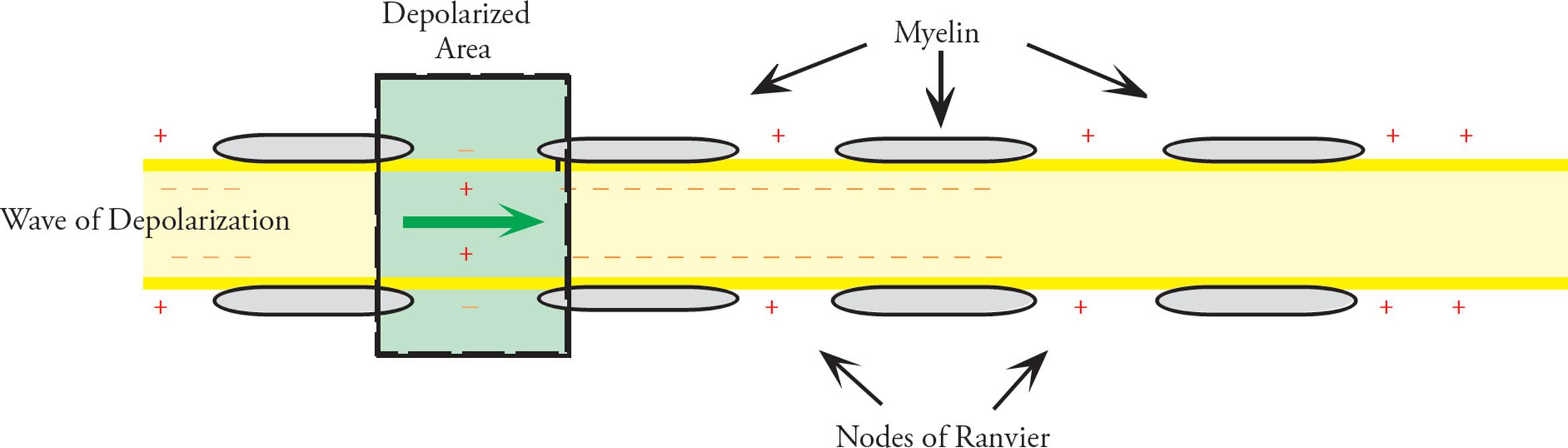
Figure 5 Propagation of the AP in a Myelinated Axon (cross section)
Glial Cells
As mentioned above, the myelin sheath is formed by a type of glial cell called a Schwann cell. However, Schwann cells are not the only type of glial cell. Glial cells are specialized, non-neuronal cells that typically provide structural and metabolic support to neurons (Table 1). Glia maintain a resting membrane potential but do not generate action potentials.
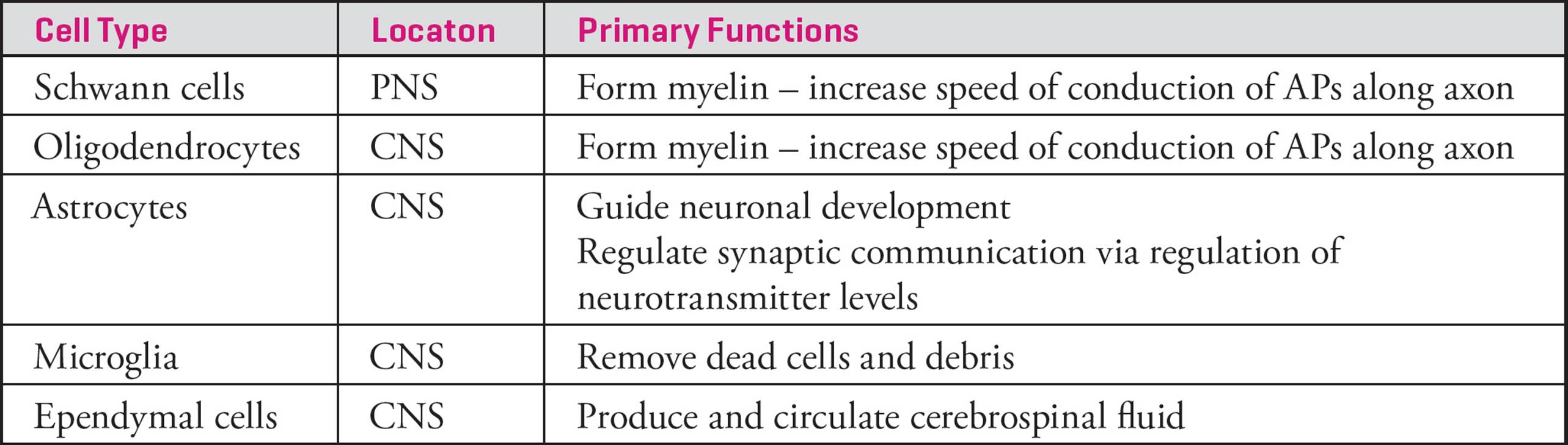
Table 1 Types of Glial Cells and Their Functions
Equilibrium Potentials
During the action potential, the movement of Na+ and K+ ions across the membrane through the voltage-gated channels is passive; driven by gradients. The equilibrium potential is the membrane potential at which this driving force (the gradient) does not exist; in other words, there would be no net movement of ions across the membrane. Note that the equilibrium potential is specific for a particular ion. For example, the Na+ equilibrium potential is positive, approximately +50 mV. Na+ ions are driven inward by their concentration gradient. However, if the interior of the cell is too positive, the positively-charged ions are repelled; in other words, the electrical gradient would drive sodium out. These forces, the chemical gradient driving sodium in and the electrical gradient driving sodium out balance each other at about +50 mV, so this is the equilibrium potential for Na+.
K+, however, has a negative equilibrium potential. K+ ions are driven outward by their concentration gradient. However if the interior of the cell is too negative, the positively-charged ions cannot escape the attraction; the electrical gradient drives potassium in. The chemical gradient driving potassium out and the electrical gradient driving potassium in balance each other at about –90 mV, so this is the equilibrium potential for K+.
The equilibrium potential for any ion is based on the electrochemical gradient for that ion across the membrane, and can be predicted by the Nernst equation:
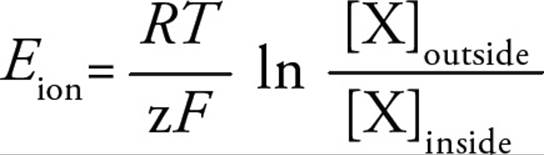
where Eion is the equilibrium potential for the ion, R is the universal gas constant, T is the temperature (in Kelvin), z is the valence of the ion, F is Faraday’s constant, and [X] is the concentration of the ion on each side of the plasma membrane. Note that the relative concentrations of the ion on each side of the membrane create the chemical gradient, while the valence (charge of the ion) helps determine the electrical gradient.
Note that the fact that the resting membrane potential is –70 mV reflects both the differences in the equilibrium potentials for Na+ and K+, and also the relative numbers of leak channels for these two ions. If the cell were completely permeable to K+, the resting potential would be about –90 mV. The fact that the resting potential is very close to the K+ equilibrium potential indicates that there are a large number of K+ leak channels in the membrane; the cell at rest is almost completely permeable to potassium. However, the resting potential is slightly more positive than –90 mV, indicating that there are a few Na+ leak channels allowing Na+ in. Not very many Na+ leak channels, though, or the resting potential would be much more positive; closer to the Na+ equilibrium potential. (This is in fact what we see when the cell does become completely permeable to Na+ at the beginning of the action potential; the membrane potential shoots upward to +35 mV.)
The Refractory Period
Action potentials can pass through a neuron extremely rapidly, thousands each second, but there is an upper limit to how soon a neuron can conduct an action potential after another has passed. The passage of one action potential makes the neuron nonresponsive to membrane depolarization and unable to transmit another action potential, or refractory, for a short period of time. There are two phases of the refractory period, caused by two different factors. During the absolute refractory period, a neuron will not fire another action potential no matter how strong a membrane depolarization is induced. During this time, the voltage-gated sodium channels have been inactivated (not the same as closed) after depolarization. They will not be able to be opened again until the membrane potential reaches the resting potential and the Na+ channels have returned to their “closed” state. During the relative refractory period, a neuron can be induced to transmit an action potential, but the depolarization required is greater than normal because the membrane is hyperpolarized. When repolarization occurs, there is a brief period in which the membrane potential is more negative than the resting potential (Figure 3) caused by voltage-gated potassium channels that have not closed yet. Because it is further from threshold, a greater stimulus is required to open the voltage-gated sodium channels to start an action potential. [If a fruit fly mutant is found that has voltage-gated potassium channels that shut more quickly after repolarization, how would this affect the refractory period in the fly?16]
9.2 SYNAPTIC TRANSMISSION
A synapse is a junction between the axon terminus of a neuron and the dendrites, soma, or axon of a second neuron. It can also be a junction between the axon terminus of a neuron and an organ. There are two types of synapses: electrical and chemical. Electrical synapses occur when the cytoplasms of two cells are joined by gap junctions. If two cells are joined by an electrical synapse, an action potential will spread directly from one cell to the other. Electrical synapses are not common in the nervous system although they are quite important in propagating action potentials in smooth muscle and cardiac muscle. In the nervous system, chemical synapses are found at the ends of axons where they meet their target cell; here, an action potential is converted into a chemical signal. The following steps are involved in the transmission of a signal across a chemical synapse in the nervous system (Figure 6), as well as at the junctions of neurons with other cell types, such as skeletal muscle cells:
1) An action potential reaches the end of an axon, the synaptic knob.
2) Depolarization of the presynaptic membrane opens voltage-gated calcium channels.
3) Calcium influx into the presynaptic cell causes exocytosis of neurotransmitter stored in secretory vesicles.
4) Neurotransmitter molecules diffuse across the narrow synaptic cleft (small space between cells).
5) Neurotransmitter binds to receptor proteins in the postsynaptic membrane. These receptors are ligand-gated ion channels.
6) The opening of these ion channels in the postsynaptic cell alters the membrane polarization.
7) If the membrane depolarization of the postsynaptic cell reaches the threshold of voltage-gated sodium channels, an action potential is initiated.
8) Neurotransmitter in the synaptic cleft is degraded and/or removed to terminate the signal.
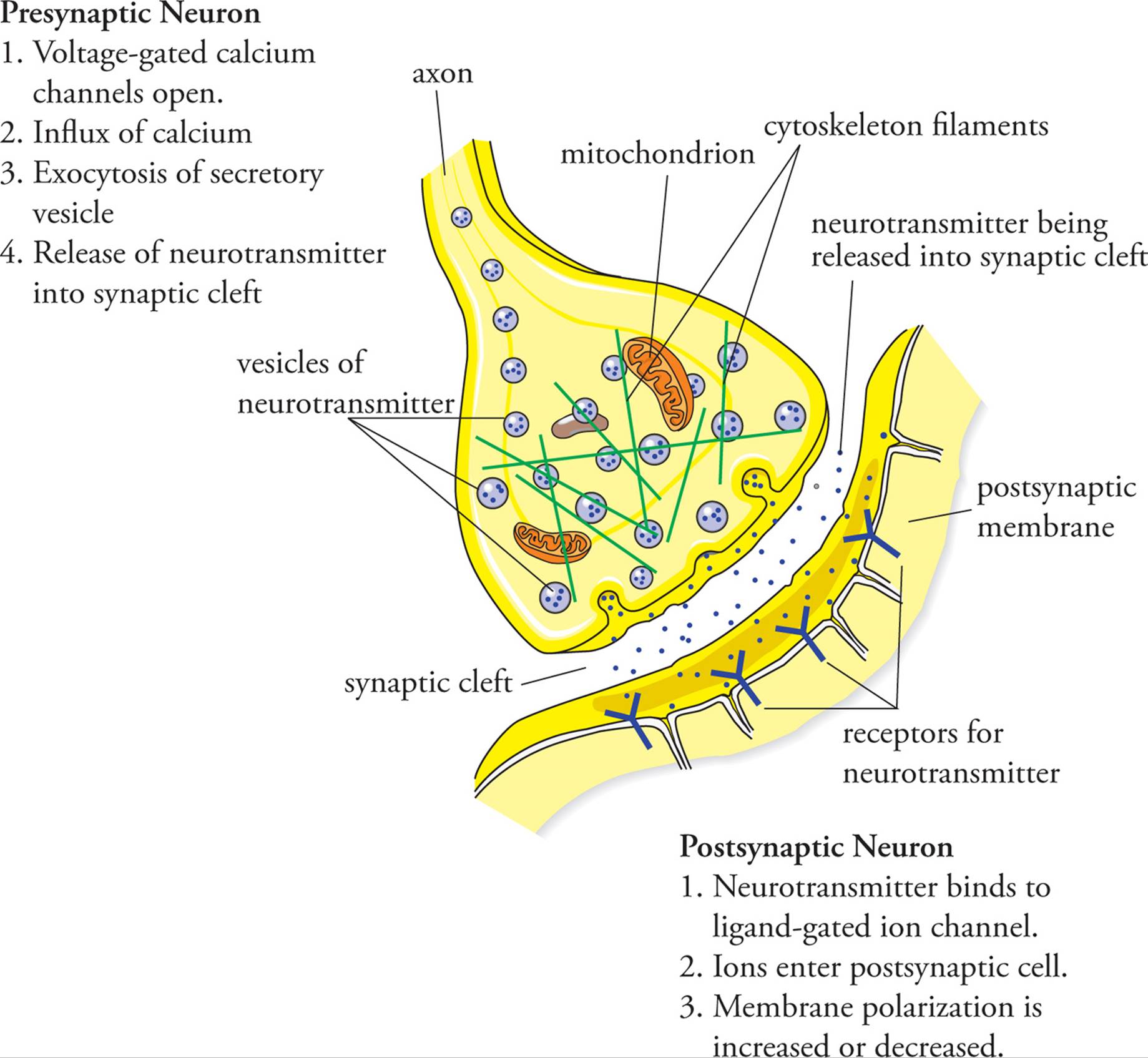
Figure 6 A Typical Synapse
An example of a chemical synapse that is commonly used is the neuromuscular junction between neurons and skeletal muscle. The neurotransmitter that is released at the neuromuscular junction is acetylcholine (ACh). When an action potential reaches such a synapse, acetylcholine is released into the synaptic cleft. The acetylcholine binds to the acetylcholine receptor on the surface of the postsynaptic cell membrane. When acetylcholine binds to its receptor, the receptor opens its associated sodium channel, allowing sodium to flow down a gradient into the cell, depolarizing the postsynaptic cell membrane. Meanwhile, acetylcholine in the synaptic cleft is degraded by the enzyme acetylcholinesterase (AChE).
There are several different neurotransmitters and neurotransmitter receptors. Some of the other neurotransmitters are gamma-aminobutyric acid (GABA), serotonin, dopamine, and norepinephrine. If a neurotransmitter, such as acetylcholine, opens a channel that depolarizes the postsynaptic membrane, the neurotransmitter is termed excitatory. Other neurotransmitters, however, have the opposite effect, making the postsynaptic membrane potential more negative than the resting potential, or hyperpolarized. Neurotransmitters that induce hyperpolarization of the postsynaptic membrane are termed inhibitory. (Note, however, that ultimately it is not the neurotransmitter that determines the effect on the postsynaptic cell, it is the receptor for that neurotransmitter and its associated ion channel. The same neurotransmitter can be excitatory in some cases and inhibitory in others.) Each presynaptic neuron can release only one type of neurotransmitter, although a postsynaptic neuron may respond to many different neurotransmitters.
• If a neurotransmitter causes the entry of chloride into the postsynaptic cell, is the neurotransmitter excitatory or inhibitory?17
• If an inhibitor of acetylcholinesterase is added to a neuromuscular junction, then the postsynaptic membrane will:18
A) be depolarized by action potentials more frequently.
B) be depolarized longer with each action potential.
C) be resistant to depolarization.
D) spontaneously depolarize.
• Signals can be sent in only one direction through synapses such as the neuromuscular junction. Which of the following best explains unidirectional signaling at synapses between neurons?19
A) Neurotransmitter is always degraded by the postsynaptic cell.
B) Only the postsynaptic cell has neurotransmitter receptors.
C) Axons can propagate action potentials in only one direction.
D) Only the postsynaptic cell has a resting membrane potential.
Summation
Once an action potential is initiated in a neuron, it will propagate to the end of the axon at a speed and magnitude of depolarization that do not vary from one action potential to another. The action potential is an “all-or-nothing” event. The key regulated step in the nervous system is whether or not a neuron will fire an action potential. Action potentials are initiated when the postsynaptic membrane reaches the threshold depolarization (about –50 mV) required to open voltage-gated sodium channels. The postsynaptic depolarization caused by the release of neurotransmitter by one action potential at one synapse is not generally sufficient to induce this degree of depolarization. A postsynaptic neuron has many different neurons with synapses leading to it, however, and each of these synapses can release neurotransmitter many times per second. The “decision” by a postsynaptic neuron whether to fire an action potential is determined by adding the effect of all of the synapses impinging on a neuron, both excitatory and inhibitory. This addition of stimuli is termed summation.
Excitatory neurotransmitters cause postsynaptic depolarization, or excitatory postsynaptic potentials (EPSPs), while inhibitory neurotransmitters cause inhibitory postsynaptic potentials (IPSPs). One form of summation is temporal summation, in which a presynaptic neuron fires action potentials so rapidly that the EPSPs or IPSPs pile up on top of each other. If they are EPSPs, the additive effect might be enough to reach the threshold depolarization required to start a postsynaptic action potential. If they are IPSPs, the postsynaptic cell will hyperpolarize, moving further and further away from threshold, effectively becoming inhibited. The other form of summation is spatial summation, in which the EPSPs and IPSPs from all of the synapses on the postsynaptic membrane are summed at a given moment in time. If the total of all EPSPs and IPSPs causes the postsynaptic membrane to reach the threshold voltage, an action potential will be fired.
• In which one of the following ways can a presynaptic neuron increase the intensity of signal it transmits?20
A) Increase the size of presynaptic action potentials
B) Increase the frequency of action potentials
C) Change the type of neurotransmitter it releases
D) Change the speed of action potential propagation
9.3 FUNCTIONAL ORGANIZATION OF THE HUMAN NERVOUS SYSTEM
The nervous system must receive information, decide what to do with it, and cause muscles or glands to act upon that decision. Receiving information is the sensory function of the nervous system (carried out by the peripheral nervous system, or PNS), processing the information is the integrative function (carried out by the central nervous system, or CNS), and acting on it is the motor function (also carried out by the PNS).21 Motor neurons carry information from the nervous system toward organs which can act upon that information, known as effectors. [What are the two types of effectors?22] Notice that “motor” neurons do not lead only “to muscle.” Motor neurons, which carry information away from the central nervous system and innervate effectors, are called efferent neurons (remember, efferents go to effectors). Sensory neurons, which carry information toward the central nervous system, are called afferent neurons.
Reflexes
The simplest example of nervous system activity is the reflex. This is a direct motor response to sensory input which occurs without conscious thought. In fact, it usually occurs without any involvement of the brain at all. In the simplest example, a sensory neuron transmits an action potential to a synapse with a motor neuron in the spinal cord, which causes an action to occur. For example, in the muscle stretch reflex, a sensory neuron detects stretching of a muscle (Figure 7). The sensory neuron has a long dendrite and a long axon, which transmits an impulse to a motor neuron cell body in the spinal cord. The motor neuron’s long axon synapses with the muscle that was stretched and causes it to contract. That is why the quadriceps (thigh) muscle contracts when the patellar tendon is stretched by tapping with a reflex hammer. A reflex such as this one, involving only two neurons and one synapse, is known as a monosynaptic reflex arc.
Something else also happens when a physician taps the patellar tendon. Not only does the quadriceps contract, but the hamstring also relaxes. If it did not, the leg would not be able to extend (straighten). The sensory neuron (that detects stretch) synapses with not only a motor neuron for the quadriceps, but also with an inhibitory interneuron. This is a short neuron which forms an inhibitory synapse with a motor neuron innervating the hamstring muscle. When the sensory neuron is stimulated by stretch, it stimulates both the quadriceps motor neuron and the inhibitory interneuron to the hamstring motor neuron. As a result, the quadriceps contracts and the hamstring relaxes. An interneuron is the simplest example of the integrative role of the nervous system. Concurrent relaxation of the hamstring and contraction of the quadriceps is an example of reciprocal inhibition.
• If a reflex occurs without the involvement of the brain, how are we aware of the action?23
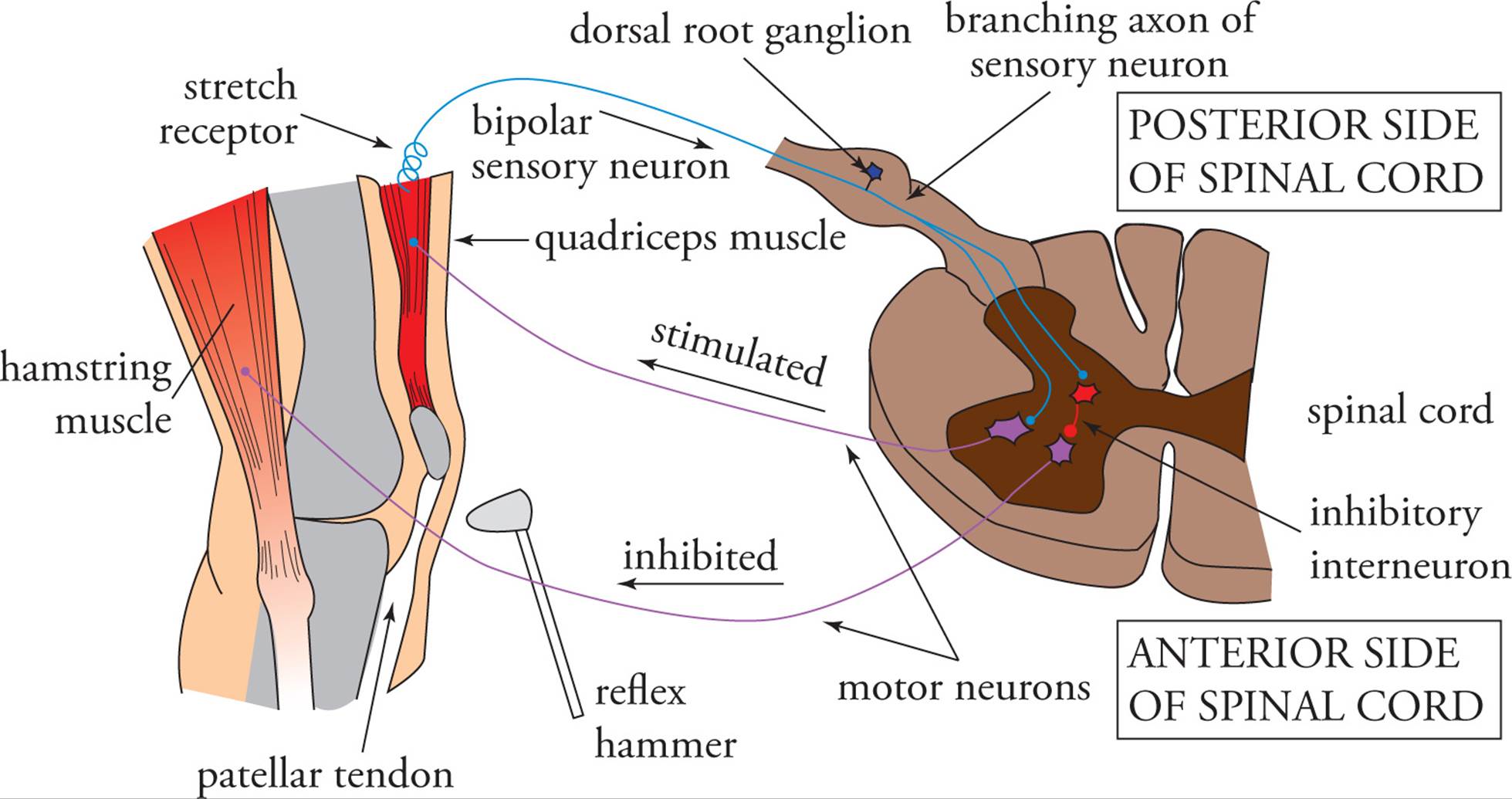
Figure 7 The Muscle Stretch Reflex
Large-Scale Functional Organization
The peripheral nervous system can be subdivided into several functional divisions (Figure 8). The portion of this system concerned with conscious sensation and deliberate, voluntary movement of skeletal muscle is the somaticdivision. The portion concerned with digestion, metabolism, circulation, perspiration, and other involuntary processes is the autonomic division. The somatic and autonomic divisions both include afferent and efferent functions, although the sources of sensory input and the target of efferent nerves are different. The efferent portion of the autonomic division is further split into two subdivisions: sympathetic and parasympathetic. When the sympathetic system is activated, the body is prepared for “fight or flight.” When the parasympathetic system is activated, the body is prepared to “rest and digest.” Table 2 summarizes the main effects of the autonomic system. Notice that many sympathetic effects result from release of epinephrine24 into the bloodstream by the adrenal medulla. The parasympathetic system prepares you to rest and digest food.
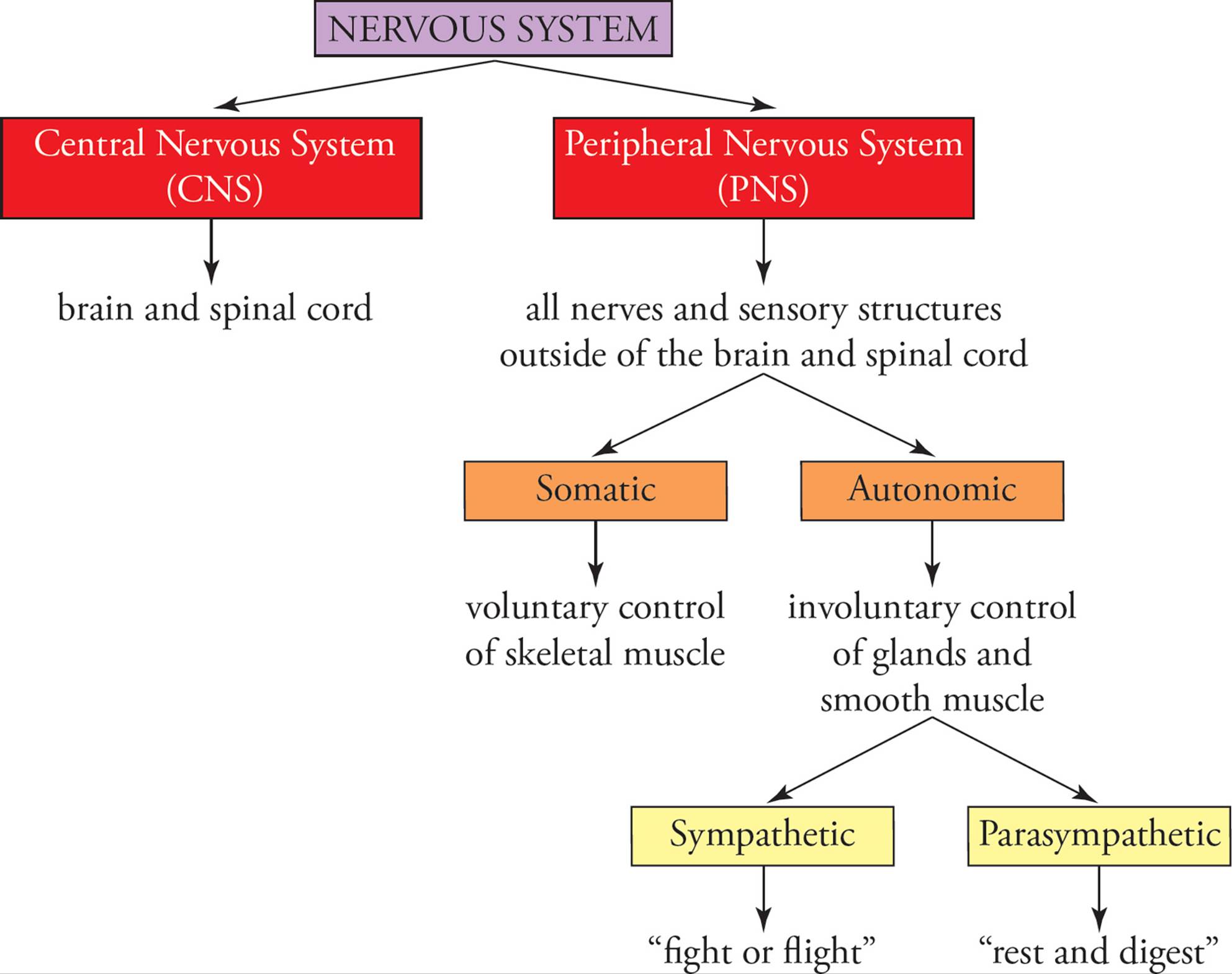
Figure 8 Overall Organization of the Nervous System
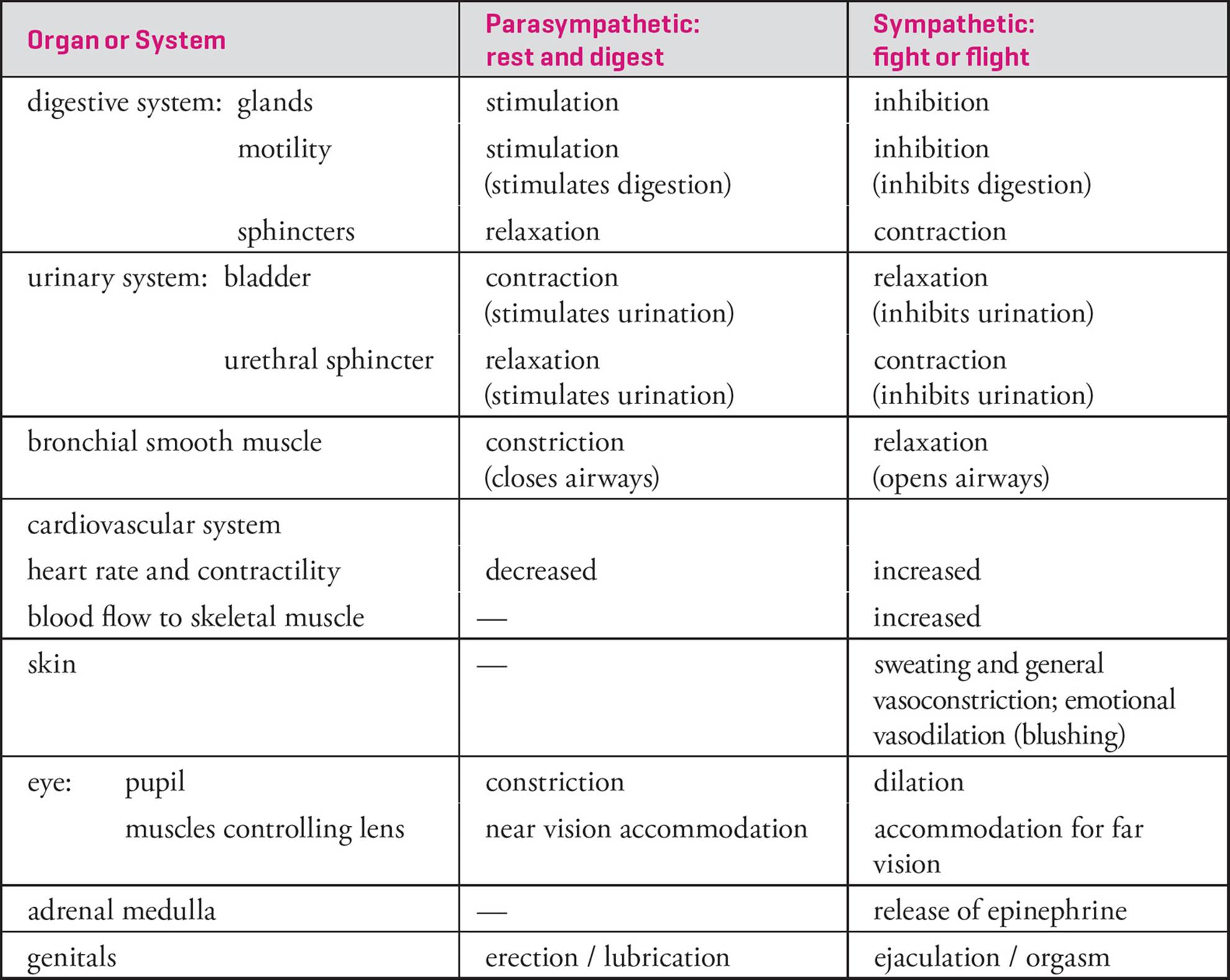
Table 2 Effects of the Autonomic Nervous System
9.4 ANATOMICAL ORGANIZATION OF THE NERVOUS SYSTEM
The main anatomical division of the nervous system is between the central nervous system (CNS) and the peripheral nervous system (PNS). The central nervous system is the brain and spinal cord. The peripheral nervous system includes all other axons, dendrites, and cell bodies. The great majority of neuronal cell bodies are found within the central nervous system. Sometimes they are bunched together to form structures called nuclei. (Don’t confuse this with the nucleic-acid-containing nuclei of cells.) Somas located outside the central nervous system are found in bunches known as ganglia. The anatomy of both the central and the peripheral system will be presented.
CNS Anatomical Organization
The CNS includes the spinal cord and the brain. The brain has three subdivisions: the hindbrain (or the rhombencephalon), the midbrain (or the mesencephalon), and the forebrain (or the prosencephalon). These four regions of the CNS (which will be discussed individually below) perform increasingly complex functions. The entire CNS (brain and spinal cord) floats in cerebrospinal fluid (CSF), a clear liquid that serves various functions such as shock absorption and exchange of nutrients and waste with the CNS.
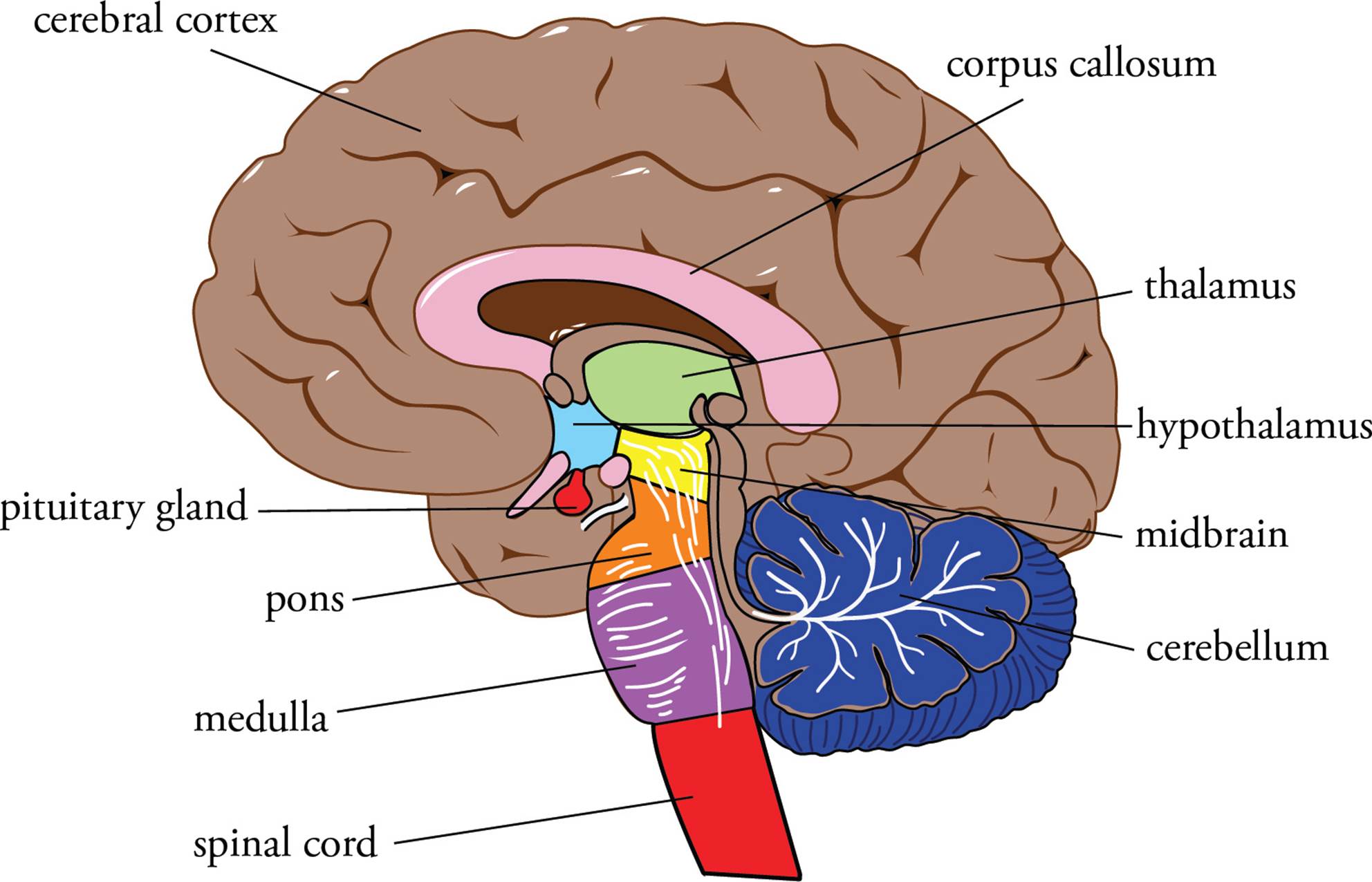
Figure 9 Organization of the CNS (cross-section of the brain)
1) The spinal cord is connected to the brain and is protected by the CSF and the vertebral column. It is a pathway for information to and from the brain. Most sensory data is relayed to the brain for integration, but the spinal cord is also a site for information integration and processing. The spinal cord is responsible for simple spinal reflexes (like the muscle stretch reflex) and is also involved in primitive processes such as walking, urination, and sex organ function.
2) The hindbrain includes the medulla, the pons, and the cerebellum.
• The medulla (or medulla oblongata) is located below the pons and is the area of the brain that connects to the spinal cord. It functions in relaying information between other areas of the brain, and regulates vital autonomic functions such as blood pressure and digestive functions (including vomiting). Also, the respiratory rhythmicity centers are found here.
• The pons is located below the midbrain and above the medulla oblongata. It is the connection point between the brain stem and the cerebellum (see below). The pons controls some autonomic functions and coordinates movement; it plays a role in balance and antigravity posture.
• The cerebellum (or “little brain”) is located behind the pons and below the cerebral hemispheres. It is an integrating center where complex movements are coordinated. An instruction for movement from the forebrain must be sent to the cerebellum, where the billions of decisions necessary for smooth execution of the movement are made. Damage to the cerebellum results in poor hand-eye coordination and balance. Both the cerebellum and the pons receive information from the vestibular apparatus in the inner ear, which monitors acceleration and position relative to gravity.
3) The midbrain is a relay for visual and auditory information and contains much of the reticular activating system (RAS), which is responsible for arousal or wakefulness.
Another term you should be familiar with is brainstem. Together, the medulla, pons and midbrain constitute the brainstem, which contains important processing centers and relays information to or from the cerebellum and cerebrum.
4) The forebrain includes the diencephalon and the telencephalon.
a) The diencephalon includes the thalamus and hypothalamus:
• The thalamus is located near the middle of the brain below the cerebral hemispheres and above the midbrain. It contains relay and processing centers for sensory information.
• The hypothalamus interacts directly with many parts of the brain. It contains centers for controlling emotions and autonomic functions, and has a major role in hormone production and release. It is the primary link between the nervous and the endocrine systems, and by controlling the pituitary gland is the fundamental control center for the endocrine system (discussed later in this chapter).
b) All parts of the CNS up to and including the diencephalon form a single symmetrical stalk, but the telencephalon consists of two separate cerebral hemispheres. Generally speaking, the areas of the left and right hemispheres have the same functions. However, the left hemisphere primarily controls the motor functions of the right side of the body, and the right hemisphere controls those of the left side. Also, in most people, the left side of the brain is said to be dominant. It is generally responsible for speech. The right hemisphere is more concerned with visual-spatial reasoning and music.
• The cerebral hemispheres are connected by a thick bundle of axons called the corpus callosum. A person with a cut corpus callosum has two independent cerebral cortices and to a certain extent two independent minds!25
• The cerebrum is the largest region of the human brain and consists of the large, paired cerebral hemispheres. The hemispheres of the cerebrum consist of the cerebral cortex (an outer layer of gray matter) plus an inner core of white matter connecting the cortex to the diencephalon.26 The gray matter is composed of trillions of somas; the white matter is composed of myelinated axons. (Most axons in the CNS and PNS are myelinated.) The cerebral hemispheres are responsible for conscious thought processes and intellectual functions. They also play a role in processing somatic sensory and motor information. The cerebral cortex is divided into four pairs of lobes, each of which is devoted to specific functions:
i) the frontal lobes initiate all voluntary movement and are involved in complex reasoning skills and problem solving.
ii) the parietal lobes are involved in general sensations (such as touch, temperature, pressure, vibration, etc.) and in gustation (taste).
iii) the temporal lobes process auditory and olfactory sensation and are involved in short-term memory, language comprehension, and emotion.
iv) the occipital lobes process visual sensation.
Figure 10 shows some of the more important cortical areas.
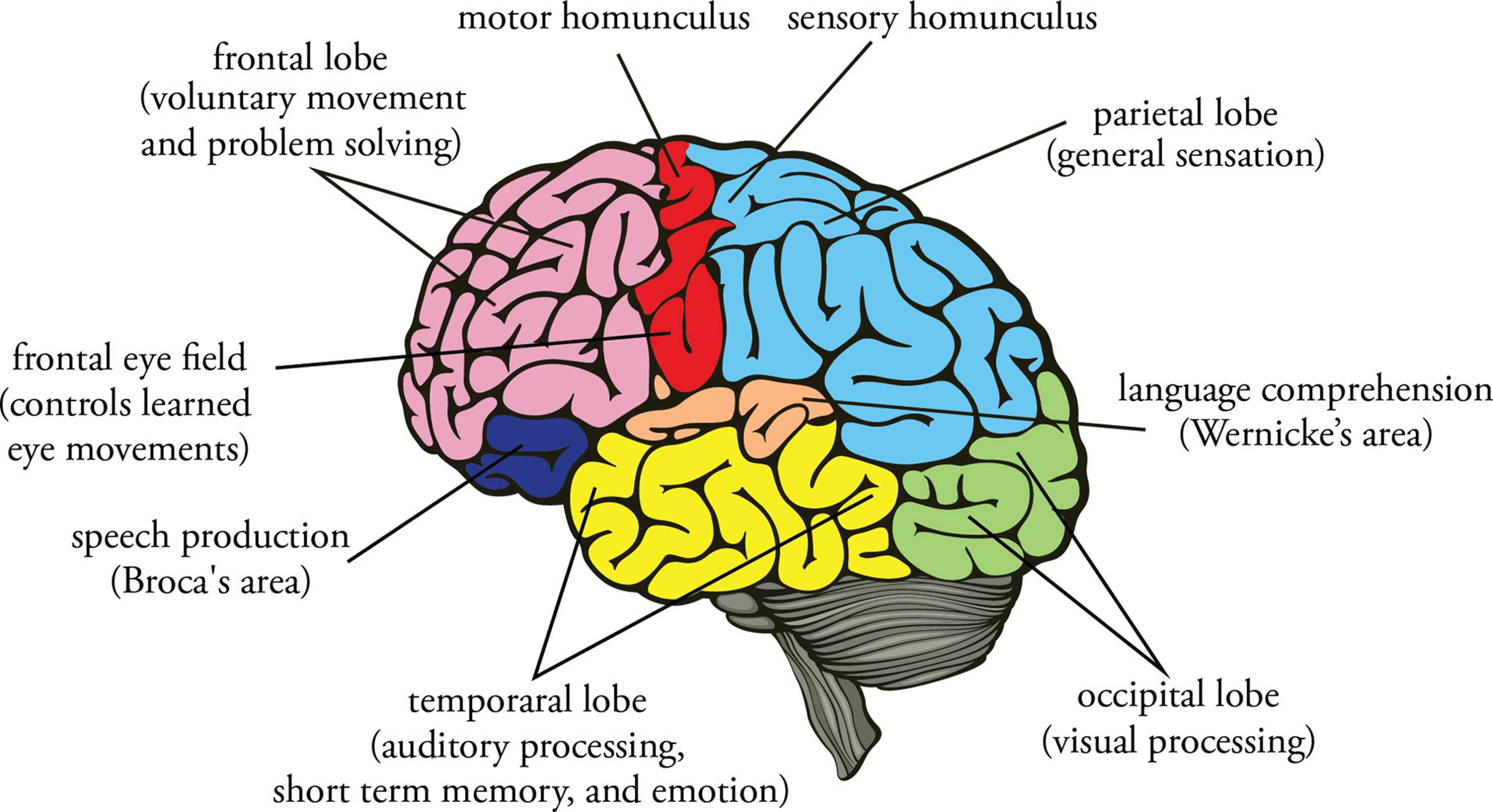
Figure 10 Principal Areas of the Cerebral Cortex
Two last regions of the brain deserve mention:
• The basal nuclei (also called the “cerebral nuclei,” and previously known as the basal ganglia) are composed of gray matter and are located deep within the cerebral hemispheres. They include several functional subdivisions, but broadly function in voluntary motor control and procedural learning related to habits. The basal nuclei and cerebellum work together to process and coordinate movement initiated by the primary motor cortex; the basal nuclei are inhibitory (preventing excess movement), while the cerebellum is excitatory.
• The limbic system is located between the cerebrum and the diencephalon. It includes several substructures (such as the amygdala, the cingulate gyrus, and the hippocampus) and works closely with parts of the cerebrum, diencephalon, and midbrain. The limbic system is important in emotion and memory.
The information above describes the general functions of each region of the brain. Table 3 summarizes the brain functions and provides a little more specific detail for each region.

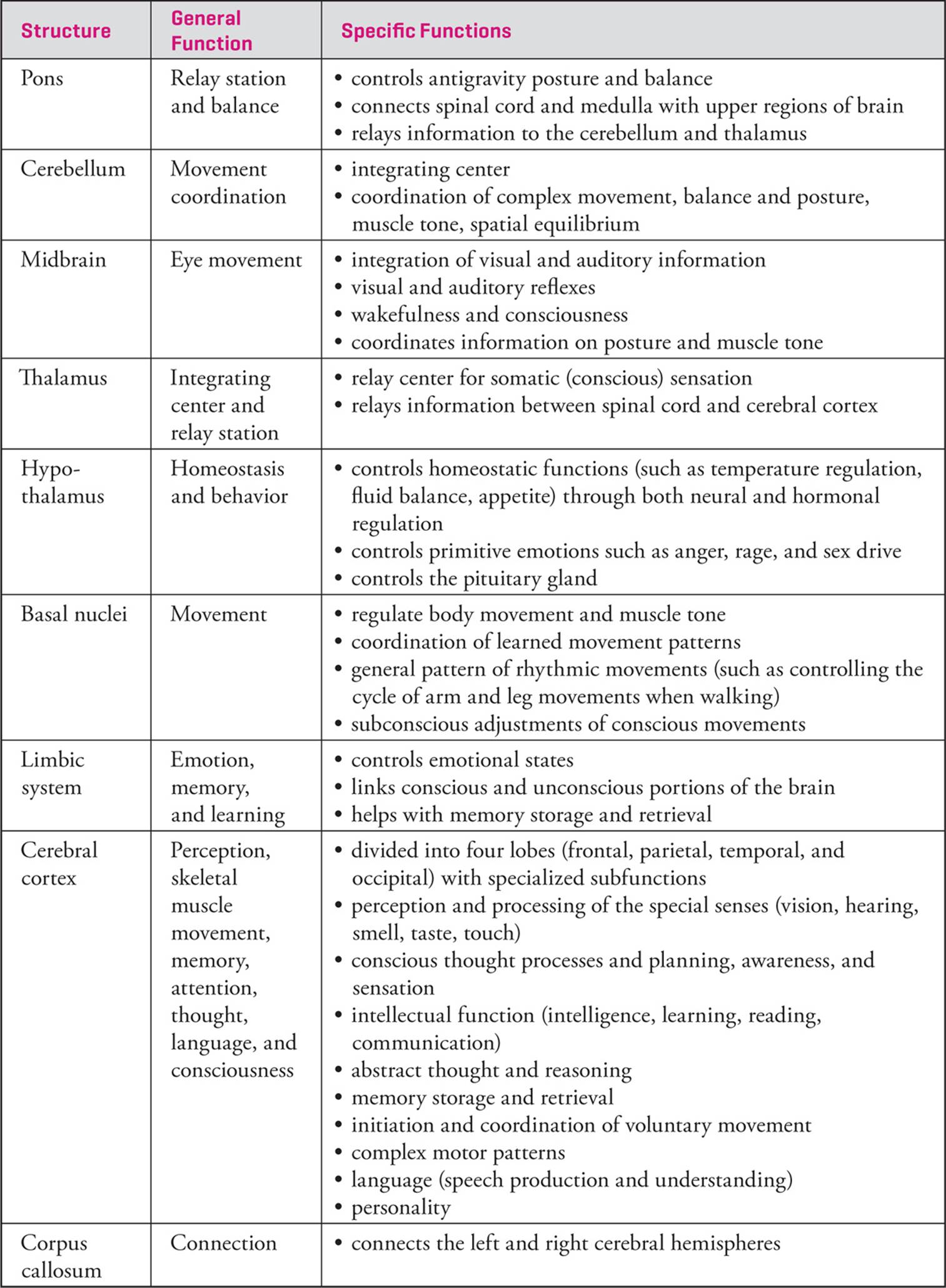
Table 3 Summary of Brain Functions
The motor and sensory regions of the cortex are organized such that a particular small area of cortex controls a particular body part. A larger area is devoted to a body part which requires more motor control or more sensation (Figure 11). For example, more cortex is devoted to the lips than to the entire leg. The body parts represented on the cortex can be sketched. The drawing looks like a distorted person, known as a homunculus (little man).
Figure 11 The Sensory Homunculus
PNS Anatomical Organization
All neurons entering and exiting the CNS are carried by 12 pairs of cranial nerves and 31 pairs of spinal nerves. Cranial nerves convey sensory and motor information to and from the brainstem. Spinal nerves convey sensory and motor information to and from the spinal cord. The different functional divisions of the nervous system have different anatomical organizations (Figure 12).
The vagus nerve is an important example of a cranial nerve, and one that you should be familiar with for the MCAT. The effects of this nerve upon the heart and GI tract are to decrease the heart rate and increase GI activity; as such it is part of the parasympathetic division of the autonomic nervous system. It is a bundle of axons that end in ganglia on the surface of the heart, stomach, and other visceral organs. The many axons constituting the vagus nerve are preganglionic and come from cell bodies located in the CNS. On the surface of the heart and stomach they synapse with postganglionic neurons. The detailed terminology in this paragraph will make more sense to you as you read through the next couple of sections.
Somatic PNS Anatomy
The somatic system has a simple organization:
• All somatic motor neurons innervate skeletal muscle cells, use ACh as their neurotransmitter, and have their cell bodies in the brain stem or the ventral (front) portion of the spinal cord.
• All somatic sensory neurons have a long dendrite extending from a sensory receptor toward the soma, which is located just outside the CNS in a dorsal root ganglion. The dorsal root ganglion is a bunch of somatic (and autonomic) sensory neuron cell bodies located just dorsal to (to the back of) the spinal cord. There is a pair of dorsal root ganglia for every segment of the spinal cord, and thus the dorsal root ganglia form a chain along the dorsal (back) aspect of the vertebral column. The dorsal root ganglia are protected within the vertebral column but are outside the meninges (protective sheath of the brain and cord) and thus outside the CNS. An axon extends from the somatic sensory neuron’s soma into the spinal cord. In all somatic sensory neurons, the first synapse is in the CNS; depending on the type of sensory information conveyed, the axon either synapses in the cord, or stretches all the way up to the brain stem before its first synapse!
Autonomic PNS Anatomy
Anatomical organization of autonomic efferents is a bit more complex.27 The efferents of the sympathetic and parasympathetic systems consist of two neurons: a preganglionic and a postganglionic neuron. The preganglionic neuron has its cell body in the brainstem or spinal cord. It sends an axon to an autonomic ganglion, located outside the spinal column. In the ganglion, this axon synapses with a postganglionic neuron. The postganglionic neuron sends an axon to an effector (smooth muscle or gland). All autonomic preganglionic neurons release acetylcholine as their neurotransmitter. All parasympathetic postganglionic neurons also release acetylcholine. Nearly all sympathetic postganglionic neurons release norepinephrine (NE, also known as noradrenaline) as their neurotransmitter.
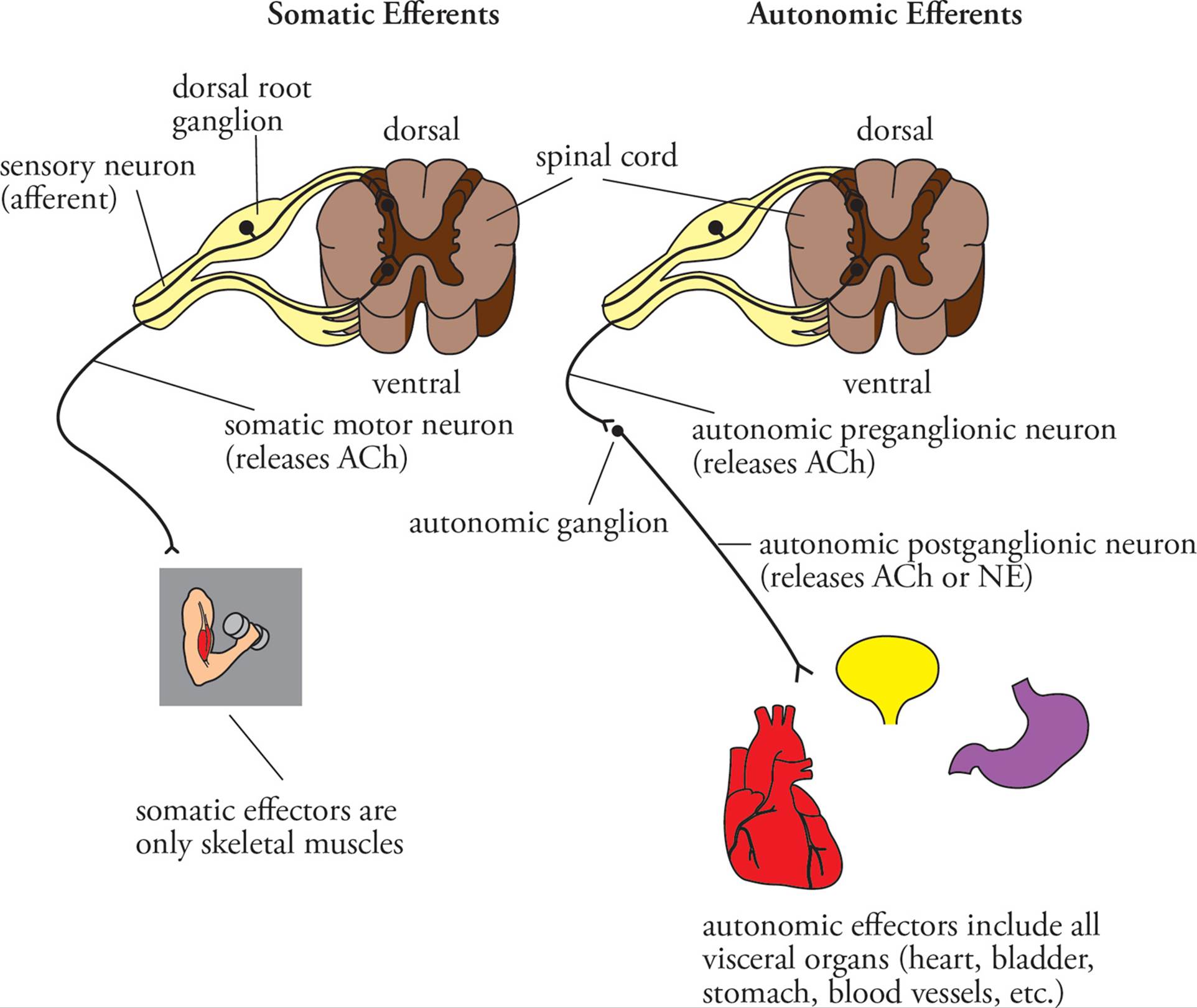
Figure 12 Anatomical Organization of PNS Efferents
All sympathetic preganglionic efferent neurons have their cell bodies in the thoracic (chest) or lumbar (lower back) regions of the spinal cord. Hence the sympathetic system is also referred to as the thoracolumbar system. The parasympathetic system is known as the craniosacral system, because all of its preganglionic neurons have cell bodies in the brainstem (which is in the head or cranium) or in the lowest portion of the spinal cord, the sacral portion. In the sympathetic system, the preganglionic axon is relatively short, and there are only a few ganglia; these sympathetic ganglia are quite large. The sympathetic postganglionic cell sends a long axon to the effector. In contrast, the parasympathetic preganglionic neuron sends a long axon to a small ganglion which is close to the effector. For example, parasympathetic ganglia controlling the intestines are located on the outer wall of the gut. The parasympathetic postganglionic neuron has a very short axon, since the cell body is close to the target.28 These differences are visualized in Figure 13 and summarized in Table 4.
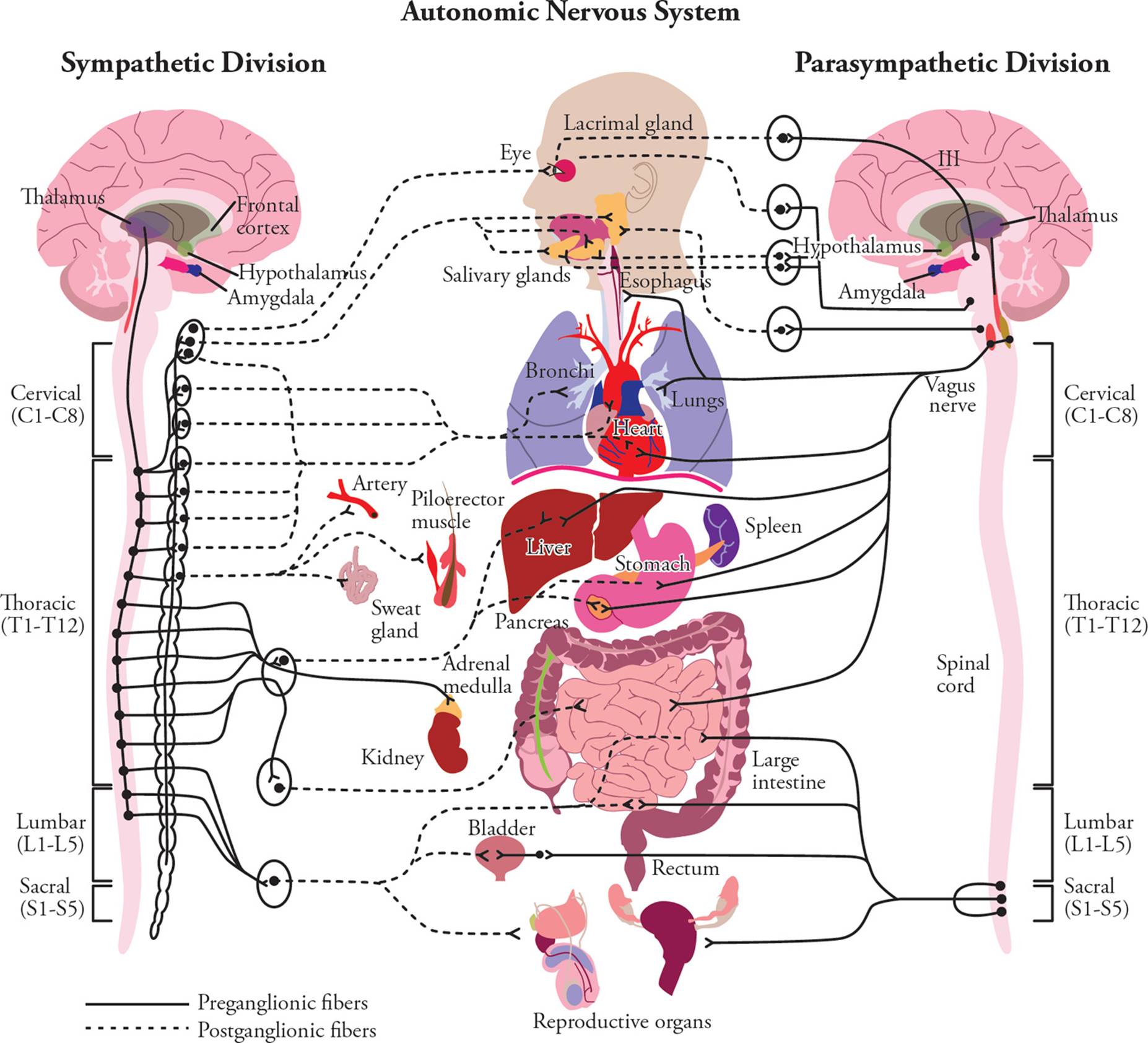
Figure 13 Pre- and Post-Ganglionic Fibers of the Autonomic Nervous System
The autonomic afferent (sensory) neurons are similar to the somatic afferent neurons with one exception: They can synapse in the PNS (at the autonomic ganglia) with autonomic efferent neurons in what is known as a “short reflex.” (Recall that the first synapse of somatic afferent neurons is in the CNS.)
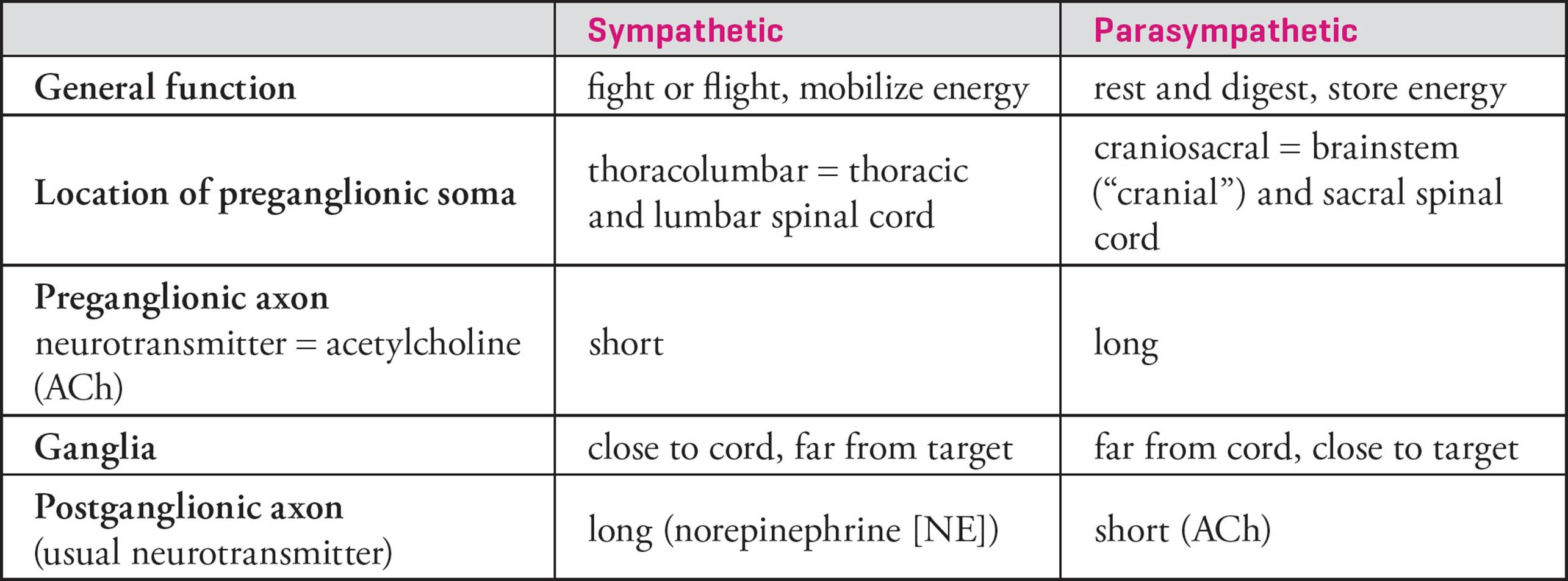
Table 4 Sympathetic vs. Parasympathetic
The Adrenal Medulla
The adrenal gland is named for its location: “Ad-” connotes “above,” and “renal” refers to the kidney. There are two adrenal glands, one above each kidney. The adrenal has an inner portion known as the medulla and an outer portion known as the cortex. The cortex is an important endocrine gland, secreting glucocorticoids (the main one is cortisol), mineralocorticoids (the main one is aldosterone), and some sex hormones.
The adrenal medulla, however, is part of the sympathetic nervous system. It is embryologically derived from sympathetic postganglionic neurons and is directly innervated by sympathetic preganglionic neurons. Upon activation of the sympathetic system, the adrenal gland is stimulated to release epinephrine, also known as adrenaline. Epinephrine is a slightly modified version of norepinephrine, the neurotransmitter released by sympathetic postganglionic neurons. Epinephrine is a hormone because it is released into the bloodstream by a ductless gland. But in many ways it behaves like a neurotransmitter. It elicits its effects very rapidly, and the effects are quite short-lived. Epinephrine release from the adrenal medulla is what causes the sudden flushing and sweating one experiences when severely startled. In general, epinephrine’s effects are those listed in Table 1 for the sympathetic system. Stimulation of the heart is an especially important effect.
9.5 SENSATION AND PERCEPTION
Sensation is the process by which we receive information from the world around us. Sensory receptors detect data, both internally (from within the body) and externally (from the environment), and send this information to the central nervous system for processing. Sensation is the act of receiving information, while perception is the act of organizing, assimilating, and interpreting the sensory input into useful and meaningful information.
Types of Sensory Receptors
Sensory receptors are designed to detect one type of stimulus from either the interior of the body or the external environment. Each sensory receptor receives only one kind of information and transmits that information to sensory neurons, which can in turn convey it to the central nervous system. [How does the brain know the difference between stimulation of visual receptors and olfactory receptors?29] Sensory receptors that detect stimuli from the outside world are exteroceptors and receptors that respond to internal stimuli are interoceptors. A more important distinction between sensory receptors is based on the type of stimulus they detect. The types of sensory receptors are listed below.
1) Mechanoreceptors respond to mechanical disturbances. For example, Pacinian corpuscles are pressure sensors located deep in the skin. The Pacinian corpuscle is shaped like an onion. It is composed of concentric layers of specialized membranes. When the corpuscular membranes are distorted by firm pressure on the skin, the nerve ending becomes depolarized and the signal travels up the dendrite (note that these are graded potential changes—not action potentials). Another important mechanoreceptor is the auditory hair cell. This is a specialized cell found in the cochlea of the inner ear. It detects vibrations caused by sound waves. Vestibular hair cells are located within special organs called semicircular canals, also found in the inner ear. Their role is to detect acceleration and position relative to gravity. An example of an autonomic mechanoreceptor would be a receptor detecting stretch of the intestinal wall.
2) Chemoreceptors respond to particular chemicals. For example, olfactory receptors detect airborne chemicals and allow us to smell things. Taste buds are gustatory receptors. Autonomic chemoreceptors in the walls of the carotid and aortic arteries respond to changes in arterial pH, PCO2, and PO2 levels.
3) Nociceptors are pain receptors.30 They are stimulated by tissue injury. Nociceptors are the simplest type of sensory receptor, generally consisting of a free nerve ending that detects chemical signs of tissue damage. (In that sense the nociceptor is a simple chemoreceptor.) Nociceptors may be somatic or autonomic. Autonomic pain receptors do not provide the conscious mind with clear pain information, but they frequently give a sensation of dull, aching pain. They may also create the illusion of pain on the skin, when their nerves cross paths with somatic afferents from the skin. This phenomenon is known as referred pain.
4) Thermoreceptors are stimulated by changes in temperature. There are autonomic and somatic examples. Peripheral thermoreceptors fall into three categories: cold-sensitive, warm-sensitive, and thermal nociceptors, which detect painfully hot stimuli.
5) Electromagnetic receptors are stimulated by electromagnetic waves. In humans, the only examples are the rod and cone cells of the retina of the eye (also termed photoreceptors). In other animals, electroreceptors and magnetoreceptors are separate. For example, some fish can detect electric fields with electroreceptors, and magnetoreceptors allow animals to sense the Earth’s magnetic field, which can help them navigate during migration.
Encoding of Sensory Stimuli
All sensory receptors need to encode relevant information regarding the nature of the stimulus being detected. There are four properties that need to be communicated to the CNS:
1) Stimulus modality is the type of stimulus. As mentioned above, the CNS determines the stimulus modality based on which type of receptor is firing.
2) Stimulus location is communicated by the receptive field of the sensory receptor sending the signal. Localization of a stimulus can be improved by overlapping receptive fields of neighboring receptors. This works like a Venn diagram, and allows the brain to localize a stimulus activating neighboring receptors to the area in which their receptive fields overlap. Discrimination between two separate stimuli can be improved by lateral inhibition of neighboring receptors.
3) Stimulus intensity is coded by the frequency of action potentials. The dynamic range, or range of intensities that can be detected by sensory receptors, can be expanded by range fractionation—including multiple groups of receptors with limited ranges to detect a wider range overall. One example of this phenomenon is human cone cells responding to different but overlapping ranges of wavelengths to detect the full visual spectrum of light.
4) Stimulus duration may or may not be coded explicitly. Tonic receptors fire action potentials as long as the stimulus continues. However, these receptors are subject to adaptation, and the frequency of action potentials decreases as the stimulus continues at the same level (see below). Phasic receptors only fire action potentials when the stimulus begins, and do not explicitly communicate the duration of the stimulus. These receptors are important for communicating changes in stimuli, and essentially adapt immediately if a stimulus continues at the same level.
The ability to adapt to a stimulus is an important property of sensory receptors. This allows the brain to tune out unimportant information from the environment. Adaptation is a decrease in firing frequency when the intensity of a stimulus remains constant. For example, if you walk into a kitchen where someone is baking bread, the bread odor molecules stimulate your olfactory receptors to a great degree and you smell the bread baking. But if you remain in the kitchen for a few minutes, you stop smelling the bread; the continuous input to the olfactory receptors causes them to stop firing even though the odor molecules are still present. This is what allows us to “get used to” certain environments and situations, for example, cold pool water, loud background noise, etc. The receptors don’t stop being able to respond; they can be retriggered if the stimulus intensity increases. For example, if you open up the oven door, you will smell the bread again. Likewise, if you are used to the background noise in a restaurant, but someone drops a plate, you’ll hear it. In other words: the nervous system is programmed to respond to changing stimuli and not so much to constant stimuli, because for the most part, constant stimuli are not a threat whereas changing stimuli might need to be dealt with. (Note that nociceptors do not adapt under any circumstance. We can learn to ignore them, but pain is something that the nervous system wants us to do something about since it is an indication that something is wrong.)
Proprioceptors
This is a broad category including many different types of receptors. Proprioception refers to awareness of self (i.e., awareness of body part position) and is also known as your kinesthetic sense.31 An important example of a proprioceptor is the muscle spindle, a mechanoreceptor. This is a sensory organ specialized to detect muscle stretch. You are already familiar with it because it is the receptor that senses muscle stretch in the deep tendon reflex. Other proprioceptors include Golgi tendon organs, which monitor tension in the tendons, and joint capsule receptors, which detect pressure, tension, and movement in the joints. By monitoring the activity of the musculoskeletal system, the proprioceptive component of the somatic sensory system allows us to know the positions of our body parts. This is most important during activity, when precise feedback is essential for coordinated motion. [What portion of the CNS would you expect to require input from proprioceptors?32]
Gustation and Olfaction
Taste and smell are senses that rely on chemoreceptors in the mouth and nasal passages. Gustation is taste, and olfaction is smell. Much of what is assumed to be taste is actually smell. (Try eating with a bad head cold.) In fact, taste receptors (known as taste buds) can only distinguish five flavors: sweet (glucose), salty (Na+), bitter (basic), sour (acidic), and umami (amino acids and nucleotides). Each taste bud responds most strongly to one of these five stimuli. The taste bud is composed of a bunch of specialized epithelial cells, shaped roughly like an onion. In its center is a taste pore, with taste hairs that detect food chemicals. Information about taste is transmitted by cranial nerves to an area of the brain’s temporal lobe not far from where the brain receives olfactory information.
Olfaction is accomplished by olfactory receptors in the roof of the nasopharynx (nasal cavity). The receptors detect airborne chemicals that dissolve in the mucus covering the nasal membrane. Humans can distinguish thousands of different smells. Olfactory nerves project directly to the olfactory bulbs of the brain. The olfactory bulbs are located in the temporal lobe of the brain near the limbic system, an area important for memory and emotion (which may explain why certain smells can bring back vivid memories and feelings).
Interestingly, the perception of a smell as “good” or “bad” is entirely learned, based on experiences with those smells. There is no smell that is universally noxious to people (though the military has tried to find one in order to develop a “stink” bomb), because different smells can be associated with good or bad experiences based on culture and upbringing.
Pheromones are chemical signals that cause a social response in members of the same species. Though not well understood in humans, pheromones have been studied extensively in insects, particularly those species with complex social structures (such as bees and ants). Pheromones are an important means of communicating information; for example, alarm pheromones will alert the rest of the beehive of danger, food-trail pheromones allow ants to follow a trail to a promising food source, and sex pheromones play an important role in mating between most species. In humans, pheromones are much harder to study.
Hearing and the Vestibular System
Structure of the Ear
The auricle or pinna and the external auditory canal comprise the outer ear. The middle ear is divided from the outer ear by the tympanic membrane or eardrum. The middle ear consists of the ossicles, three small bones called the malleus (hammer), the incus (anvil), and the stapes (stirrup). The stapes attaches to the oval window, a membrane that divides the middle and inner ear. Structures of the inner ear include the cochlea, the semicircular canals, the utricle, and the saccule. The semicircular canals together with the utricle and saccule are important to the sense of balance. The round window is a membrane-covered hole in the cochlea near the oval window. It releases excess pressure. The Eustachian tube (also known as the auditory tube) is a passageway from the back of the throat to the middle ear. It functions to equalize the pressure on both sides of the eardrum and is the cause of the “ear popping” one experiences at high altitudes or underwater.
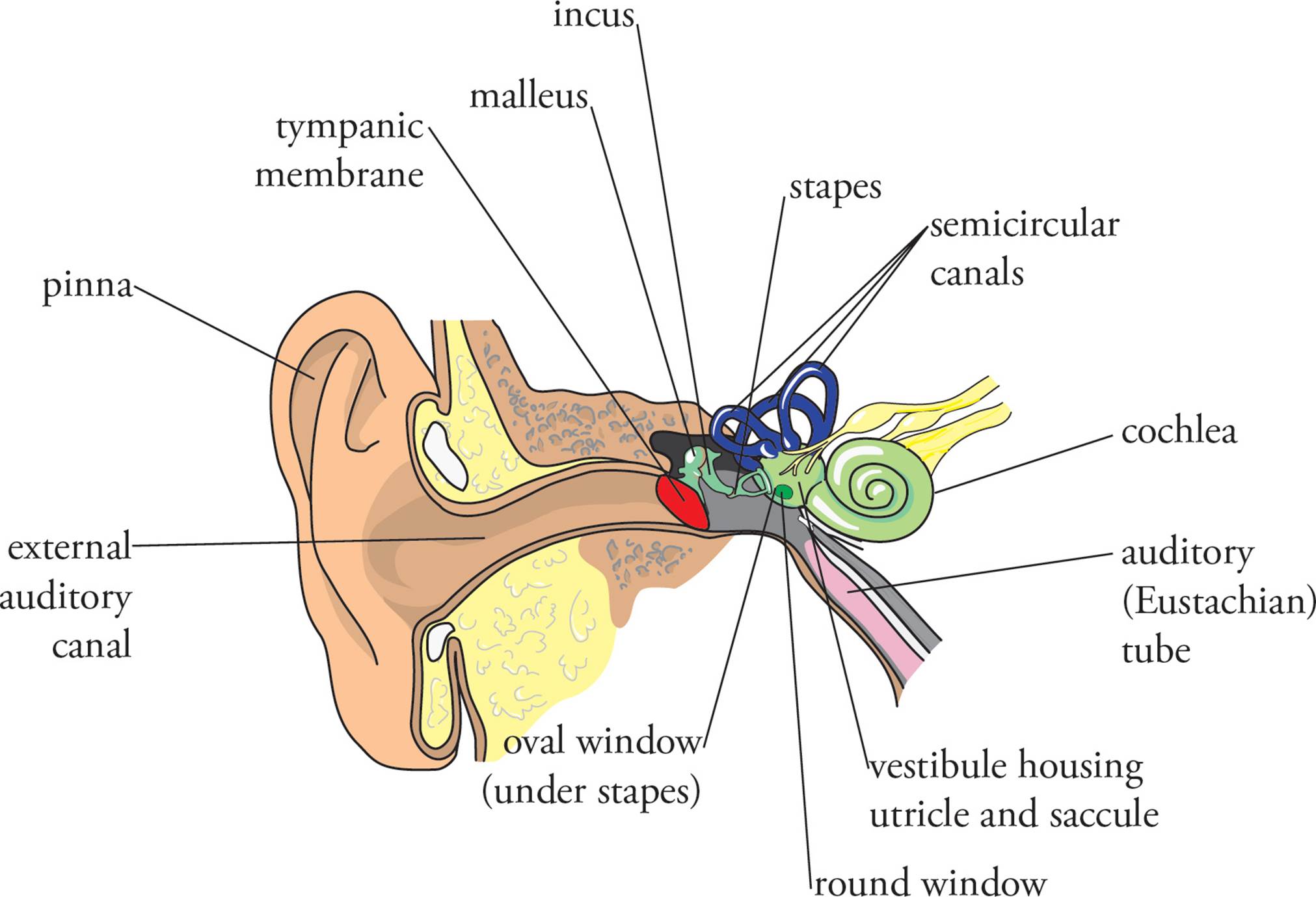
Figure 14 The Ear
Mechanism of Hearing
Sound waves enter the external ear to pass into the auditory canal, causing the eardrum to vibrate. The malleus attached to the eardrum receives the vibrations, which are passed on to the incus and then to the stapes. The bones of the middle ear are arranged in such a way that they amplify sound vibrations passing through the middle ear. The stapes is the innermost of the three middle-ear bones, contacting the oval window. Vibration of the oval window creates pressure waves in the perilymph and endolymph, the fluids in the cochlea. Note that sound vibrations are first conveyed through air, next through bone, and then through liquid before being sensed. The pressure waves in the endolymph cause vibration of the basilar membrane, a thin membrane extending throughout the coiled length of the cochlea. The basilar membrane is covered with the auditory receptor cells known as hair cells. These cells have cilia (hairs) projecting from their apical (top) surfaces (opposite the basilar membrane). The hairs contact the tectorial membrane (tectorial means “roof”), and when the basilar membrane moves, the hairs are dragged across the tectorial membrane and they bend. This displacement opens ion channels in the hair cells, which results in neurotransmitter release. Dendrites from bipolar auditory afferent neurons are stimulated by this neurotransmitter, and thus sound vibrations are converted to nerve impulses. The basilar membrane, hair cells, and tectorial membrane together are known as the organ of Corti. The outer ear and middle ear convey sound waves to the cochlea, and the organ of Corti in the cochlea is the primary site at which auditory stimuli are detected.
Summary: From Sound to Hearing
sound waves → auricle → external auditory canal → tympanic membrane → malleus → incus → stapes → oval window → perilymph → endolymph → basilar membrane → auditory hair cells → tectorial membrane → neurotransmitters stimulate bipolar auditory neurons → brain → perception
Pitch (frequency) of sound is distinguished by which regions of the basilar membrane vibrate, stimulating different auditory neurons. The basilar membrane is thick and sturdy near the oval window and gradually becomes thin and floppy near the apex of the cochlea. Low frequency (long wavelength) sounds stimulate hair cells at the apex of the cochlear duct, farthest away from the oval window, while high-pitched sounds stimulate hair cells at the base of the cochlea, near the oval window. Loudness of sound is distinguished by the amplitude of vibration. Larger vibrations cause more frequent action potentials in auditory neurons.
Locating the source of sound is also an important adaptive function. Having two ears allows for stereophonic (or three-dimensional) hearing. The auditory system can determine the source of a sound based on the difference detected between the two ears. For example, if a horn blasts to your right, your right ear will receive the sound waves slightly sooner and slightly more intensely than your left ear. Sound stimuli are processed in the auditory cortex, located in the temporal lobe of the brain.
In humans, audition is highly adaptive. While we are able to hear a wide range of sounds, those sounds with frequencies within the range corresponding to the human voice are heard best, and we are able to differentiate variations among human voices. For example, when answering the phone, you will recognize your mom’s voice within a fraction of a second.
• If a sensory neuron leading from the ear to the brain fires an action potential more rapidly, how will the brain perceive this change?33
• In some cases of deafness, sound can still be detected by conduction of vibration through the skull to the cochlea. If the auditory nerve is severed, can sound still be detected by conductance through bone?34
• If the bones of the middle ear are unable to move, would this impair the detection of sound by conductance through bone?35
Equilibrium and Balance
The vestibular complex is made up of the three semicircular canals, the utricle, the saccule, and the ampullae. All are essentially tubes filled with endolymph, and like the cochlea, they contain hair cells that detect motion. However, their function is to detect not sound, but rather rotational acceleration of the head. They are innervated by afferent neurons which send balance information to the pons, cerebellum, and other areas. The vestibular complex monitors both static equilibrium and linear acceleration, which contribute to your sense of balance.
Vision: Structure and Function
The eye is the structure designed to detect visual stimuli. The structures of the eye first form an image on the retina, which detects light and converts the stimuli into action potentials to send to the brain. Light enters the eye by passing through the cornea, the clear portion at the front of the eye. Light is bent or refracted as it passes through the cornea (which is highly curved and thus acts as a lens), since the refractive index of the cornea is higher than that of air. The cornea is continuous at its borders with the white of the eye, the sclera. Beneath the sclera is a layer called the choroid. It contains darkly-pigmented cells; this pigmentation absorbs excess light within the eye. Beneath the choroid is the retina, the surface upon which light is focused.
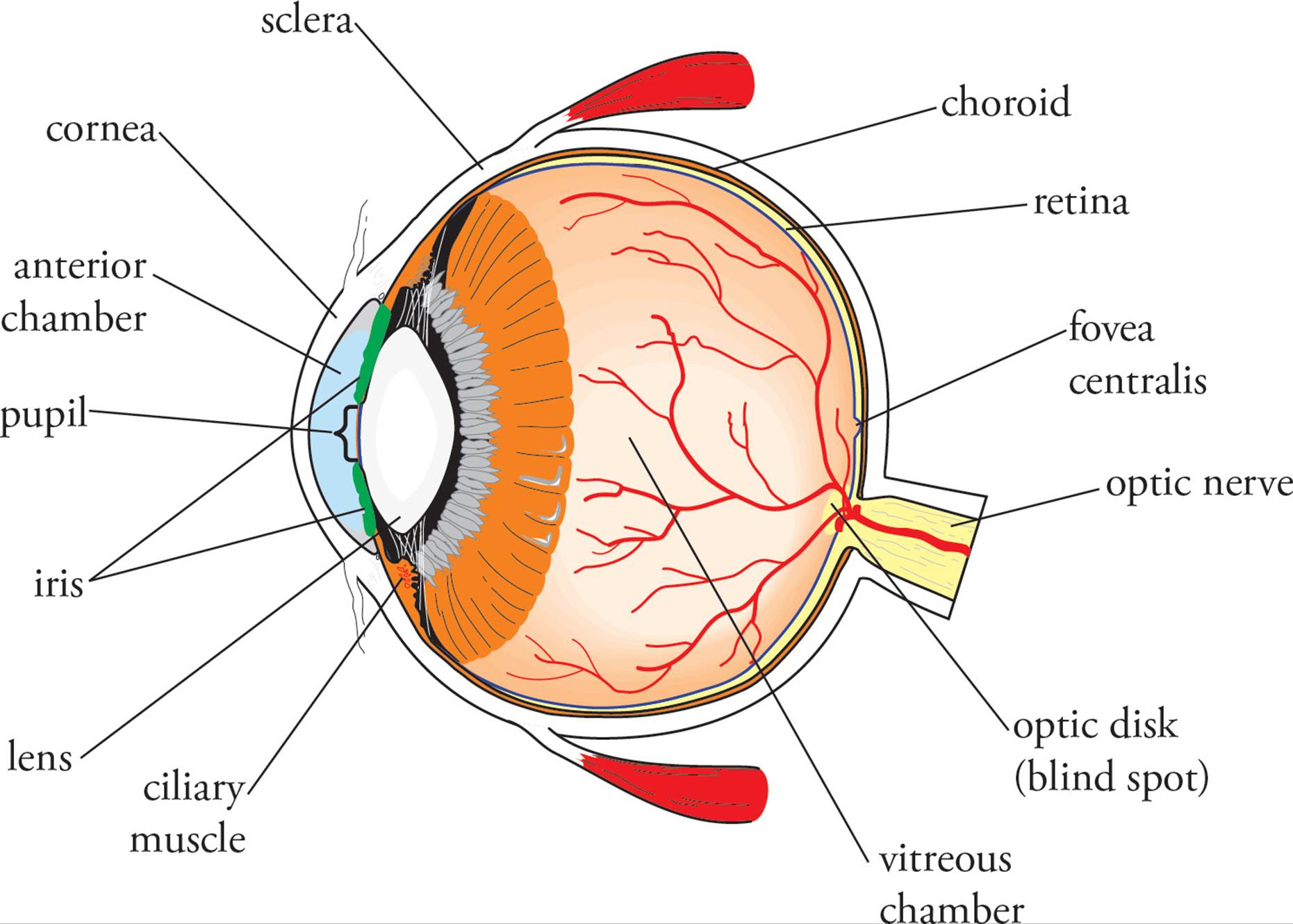
Figure 15 The Eye
Just inside the cornea is the anterior chamber (front chamber), which contains a fluid termed aqueous humor. At the back of the anterior chamber is a membrane called the iris with an opening called the pupil. The iris is the colored part of the eye, and muscles in the iris regulate the diameter of the pupil. Just behind the iris is the posterior chamber, also containing aqueous humor. In the back part of the posterior chamber is the lens. Its role is to fine-tune the angle of incoming light, so that the beams are perfectly focused upon the retina. The curvature of the lens (and thus its refractive power) is varied by the ciliary muscle.
Light passes through the vitreous chamber en route from the lens to the retina. This chamber contains a thick, jelly-like fluid called vitreous humor. The retina is located at the back of the eye. It contains electromagnetic receptor cells (photoreceptors) known as rods and cones which are responsible for detecting light. The rods and cones synapse with nerve cells called bipolar cells. In accordance with the name “bipolar,” these cells have only one axon and one dendrite. The bipolar cells in turn synapse with ganglion cells, whose axons comprise the optic nerve, which travels from each eye toward the occipital lobe of the brain where complex analysis of a visual image occurs. In Figure 16, you may notice that light has to pass through two layers of neurons before it can reach the rods and cones. The neurons are fine enough to not significantly obstruct incoming rays.
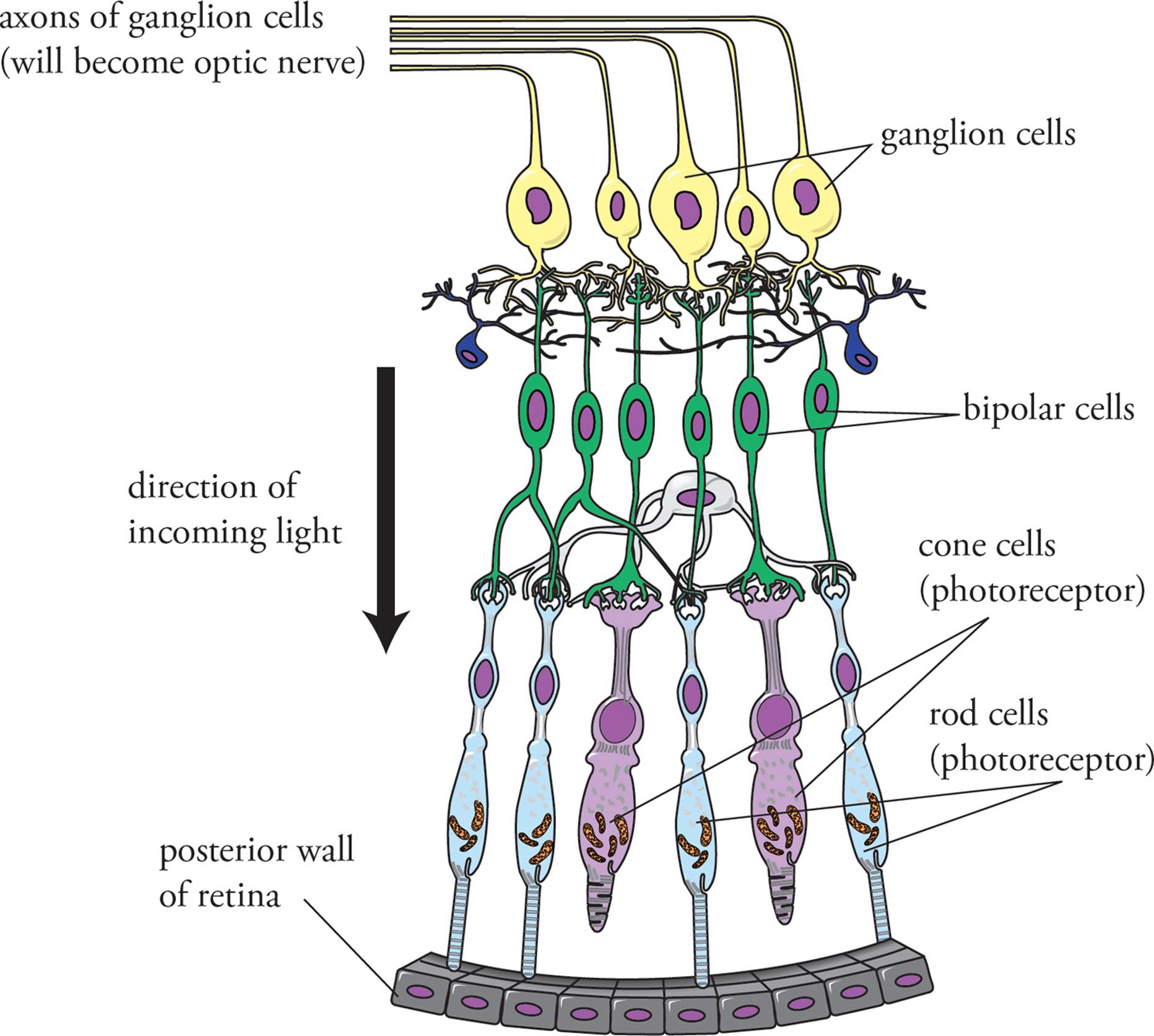
Figure 16 Organization of the Retina
The point on the retina where many axons from ganglion cells converge to form the optic nerve is the optic disk. It is also known as the blind spot (Figure 17) because it contains no photoreceptors. Another special region of the retina is the macula. In the center of the macula is the fovea centralis (focal point), which contains only cones and is responsible for extreme visual acuity. When you stare directly at something, you focus its image on the fovea.

Figure 17 Demonstrating the Blind Spot
The Photoreceptors: Rods and Cones
Rods and cones, named because of their shapes, contain special pigment proteins that change their tertiary structure upon absorbing light. Each protein, called an opsin, is bound to one molecule of retinal, which is derived from vitamin A. In the dark, when the rods and cones are resting, retinal has several trans double bonds and one cis double bond. In this conformation, retinal and its associated opsin keep a sodium channel open. The cell remains depolarized. Upon absorbing a photon of light, retinal is converted to the all-trans form. This triggers a series of reactions that ultimately closes the sodium channel, and the cell hyperpolarizes.
Rods and cones synapse on bipolar cells. Because of their depolarization in the dark, both types of photoreceptors release the neurotransmitter glutamate onto the bipolar cells, inhibiting them from firing. Upon the absorption of a photon of light and subsequent hyperpolarization, the photoreceptor stops releasing glutamate. Because glutamate has an inhibitory effect on the bipolar cells, when glutamate is no longer present, the bipolar cell can depolarize (removal of inhibition causes excitation in this system). This then causes depolarization of the ganglion cells, and an action potential along the axon of the ganglion cell. All of the axons of the ganglion cells together make up the optic nerve to the brain.
Night vision is accomplished by the rods, which are more sensitive to dim light and motion and are more concentrated in the periphery of the retina. Cones require abundant light and are responsible for color vision and high-acuity vision, and hence are more concentrated in the fovea.36 Color vision depends on the presence of three different types of cones. One is specialized to absorb blue light, one absorbs green, and one absorbs red. [What physical difference allows this functional difference?37] The brain perceives hues by integrating the relative input of these three basic stimuli.
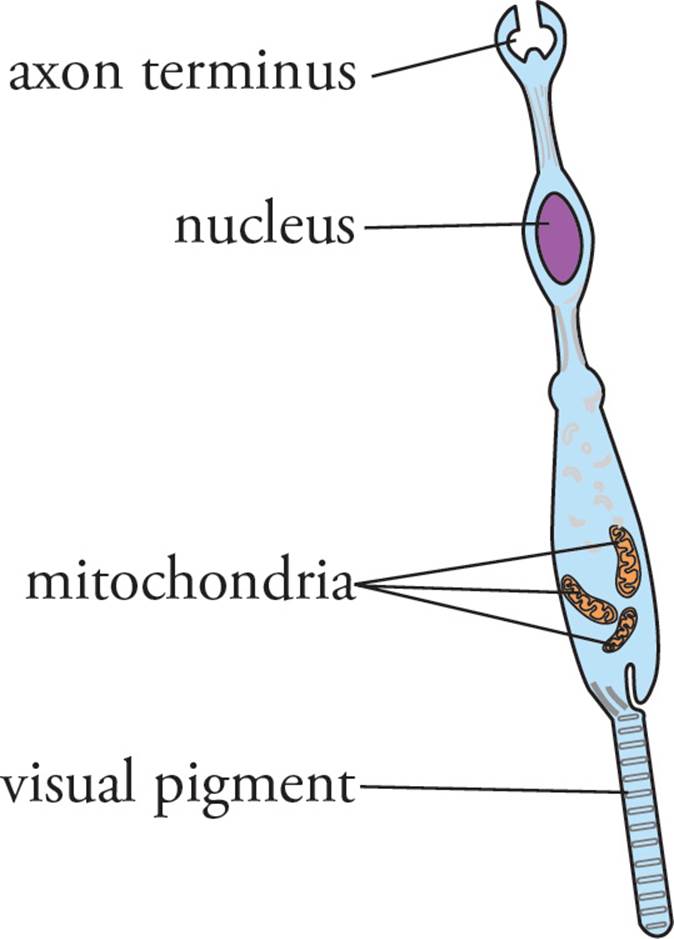
Figure 18 Rod Cell Structure
Defects in Visual Acuity
Normal vision is termed emmetropia. Too much or too little curvature of the cornea or lens results in visual defects. Too much curvature causes light to be bent too much and to be focused in front of the retina. The result is myopia, or nearsightedness. Myopia can be corrected by a concave (diverging) lens, which will cause the light rays to diverge slightly before they reach the cornea. Hyperopia, farsightedness, results from the focusing of light behind the retina. Hyperopia can be corrected by a convex (converging) lens, which causes light rays to converge before reaching the cornea. Presbyopia is an inability to accommodate (focus). It results from loss of flexibility of the lens, which occurs with aging.
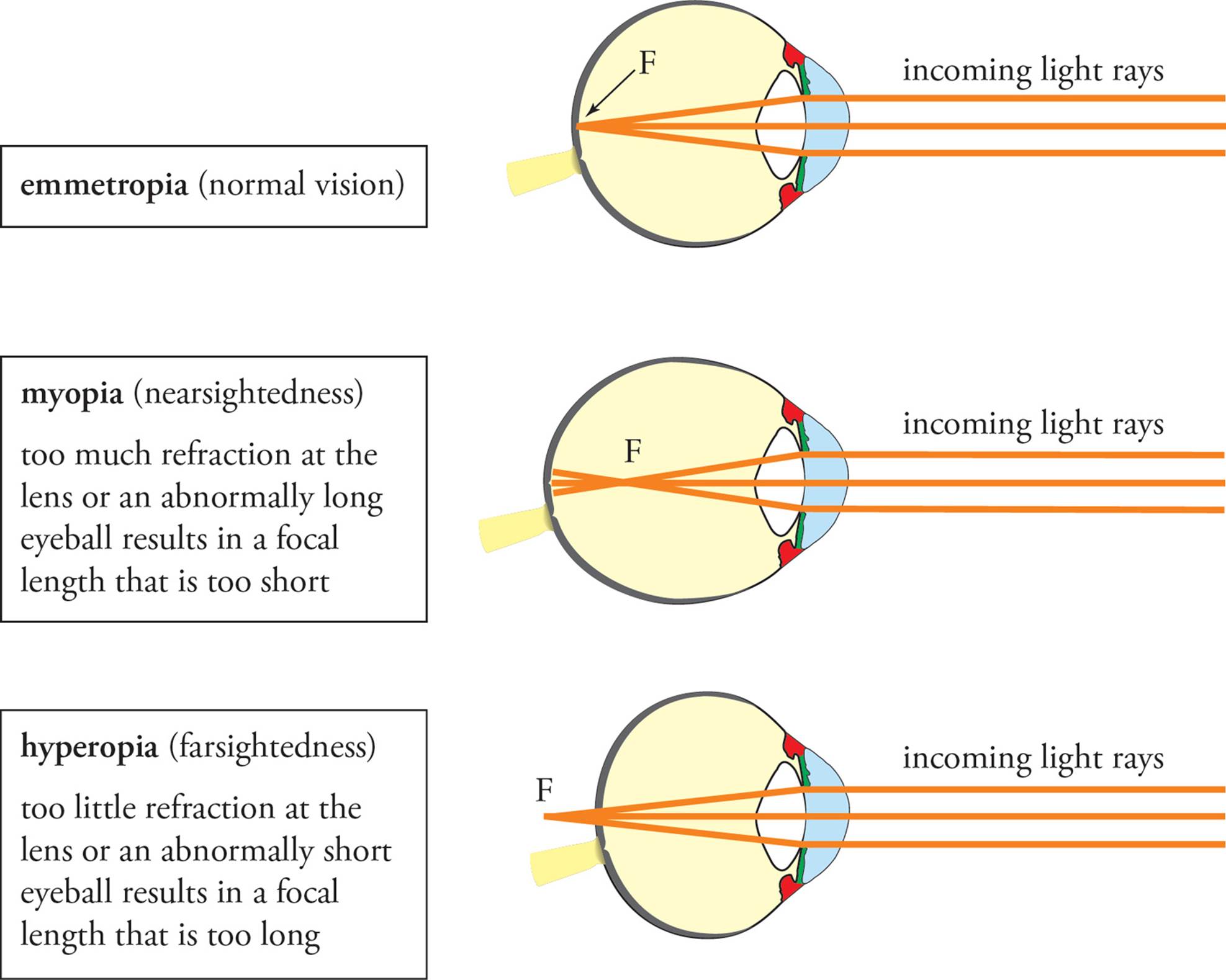
Figure 19 Defects in Visual Acuity (“F” denotes the focal point)
Vision: Information Processing
For humans, vision is the primary sense; even if other information (such as sound or smell) counters visual information, we are more likely to “believe our eyes.” The processing of visual information is extremely complex, and highly reliant on expectations and past experience. Neurons in the visual cortex fire in response to very specific information; feature detecting neurons are specific neurons in the brain that fire in response to particular visual features, such as lines, edges, angles, and movement. This information is then passed along to other neurons that begin to assimilate these distinct features into more complex objects, and so on. Therefore, feature detection theoryexplains why a certain area of the brain is activated when looking at a face, a different area is activated when looking at the letters on this page, etc. In order to process vast amounts of visual information quickly and effectively, our brain employs parallel processing, whereby many aspects of a visual stimulus (such as form, motion, color, and depth) are processed simultaneously instead of in a step-by-step or serial fashion. [Note: parallel processing is also employed for other stimuli as well.] The occipital lobe constructs a holistic image by integrating all of the separate elements of an object, in addition to accessing stored information. For example, the brain is simultaneously processing the individual features of an image, while also accessing stored information, to rapidly come to the conclusion that you are not only viewing a face, but you are specifically viewing your mom’s face. All of this requires a tremendous amount of resources; in fact, the human brain dedicates approximately 30% of the cortex to processing visual information, while only 8% is devoted to processing touch information, and a mere 3% processes auditory information!
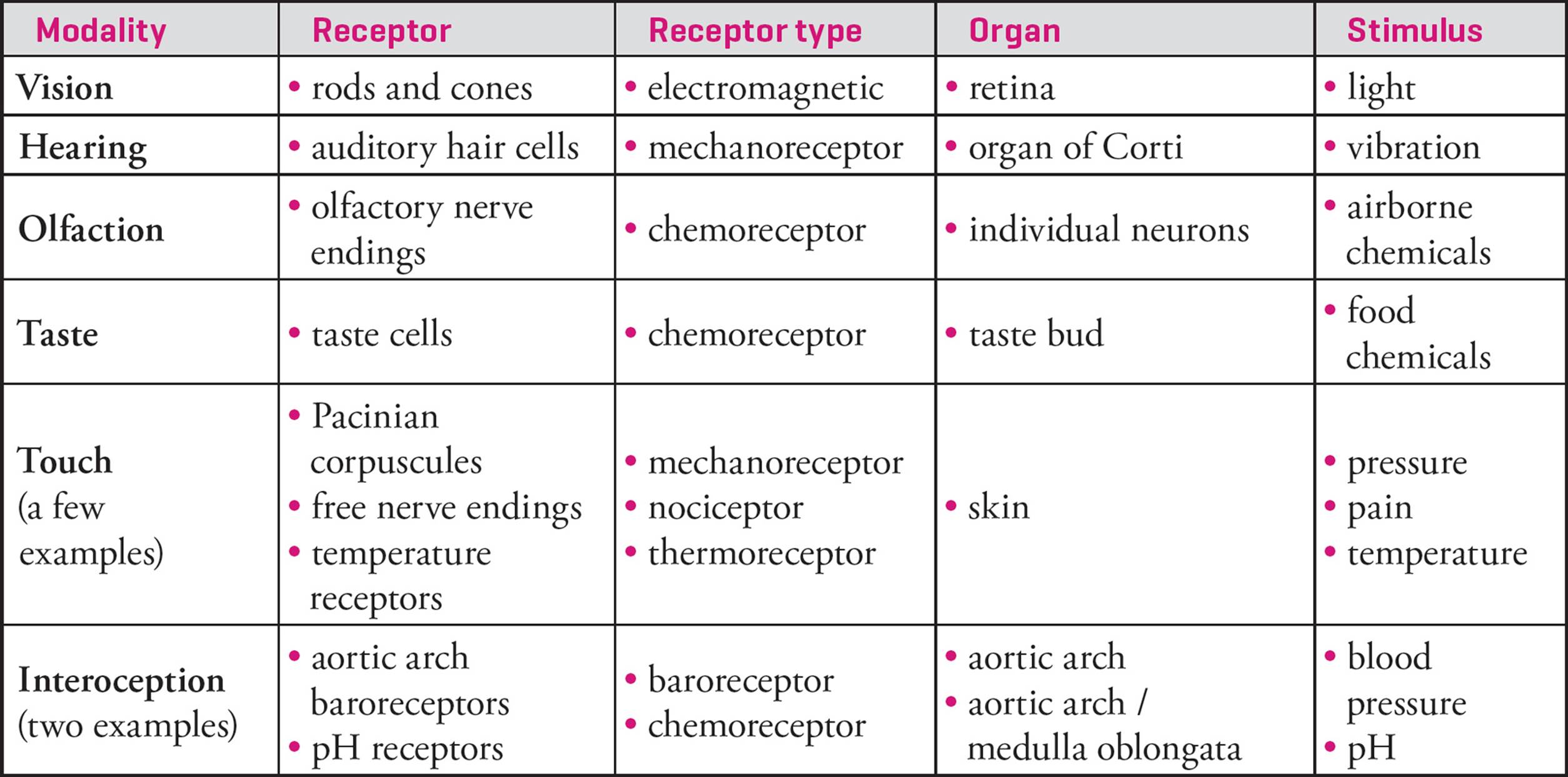
Table 5 Summary of Sensory Modalities
General Sensory Processing
Absolute Thresholds
We are very sensitive to certain types of stimuli. The minimum stimulus intensity required to activate a sensory receptor 50% of the time (and thus detect the sensation) is called the absolute threshold. In other words, for each special sense, the 50% recognition point defines the absolute threshold. (Note that this threshold can vary between individuals and different organisms—the absolute smell threshold for a human and a dog differs greatly.) Absolute thresholds also vary with age. For example, as we age, we gradually lose our ability to detect higher-pitched sounds. [What is the anatomical reason for this?38]
Difference Thresholds
Absolute thresholds are important for detecting the presence or absence of stimuli, but the ability to determine the change or difference in stimuli is also vital. The difference threshold (also called the just noticeable difference, or JND) is the minimum noticeable difference between any two sensory stimuli, 50% of the time. The magnitude of the initial stimulus influences the difference threshold; for example, if you lift a one pound weight and a two pound weight, the difference will be obvious, but if you lift a 100 pound weight and 101 pound weight, you probably won’t be able to tell the difference. Indeed, Weber’s law dictates that two stimuli must differ by a constant proportion in order for their difference to be perceptible. Interestingly, the exact proportion varies by stimulus; but for humans, two objects must differ in weight by 2% [in the weight example above, what is the minimum weight needed to detect a difference between it and the 100 pound weight?39], two lights must differ in intensity by 8%, and two tones must differ in frequency by 0.3%.
Signal Detection Theory
Detecting sensory stimuli not only depends on the information itself, but also on our psychological state, including alertness, expectation, motivation, and prior experience. Signal detection theory attempts to predict how and when someone will detect the presence of a given sensory stimulus (the “signal”) amidst all of the other sensory stimuli in the background (considered the “noise”). There are four possible outcomes: a hit (the signal is present and was detected), a miss (the signal was present but not detected), a false alarm (the signal was not present but the person thought it was), and a correct rejection (the signal was not present and the person did not think it was). Signal detection can have important life-or-death consequences—imagine how crucial it is for doctors to be able to detect the signal (perhaps a tumor on a CT scan) from the noise.
Gestalt Psychology
Gestalt is the German word for “whole.” Gestalt psychologists believe that the whole exceeds the sum of its parts; in other words, when humans perceive an object, rather than seeing lines, angles, colors, and shadows, they perceive the whole—a face or a Table or a dog. Beyond merely registering the individual pieces, the human brain perceives the whole. Bottom-up processing begins with the sensory receptors and works up to the complex integration of information occurring in the brain. Top-down processing occurs when the brain applies experience and expectations to interpret sensory information; note that the brain in fact uses a combination of the two: information is received in a bottom-up fashion from sensory receptors while the brain is superimposing assumptions in a top-down manner.
9.6 THE ENDOCRINE SYSTEM
The nervous system and endocrine system represent the two major control systems of the body. The nervous system is fast-acting with relatively short-term effects, whereas the endocrine system takes longer to communicate signals but has generally longer lasting effects. These two control systems are interconnected, as neurons can signal the release of hormones from endocrine glands. [What is one such connection in the sympathetic nervous system?40] A primary connection between the nervous and endocrine systems is the hypothalamic-pituitary axis, which is described in more detail below.
Hormone Types: Transport and Mechanisms of Action
While the nervous system regulates cellular function from instant to instant, the endocrine system regulates physiology (especially metabolism) over a period of hours to days. The nervous system communicates via the extremely rapid action potential. The signal of the endocrine system is the hormone, defined as a molecule which is secreted into the bloodstream by an endocrine gland, and which has its effects upon distant target cells possessing the appropriate receptor. An endocrine gland is a ductless gland whose secretory products are picked up by capillaries supplying blood to the region. (In contrast, exocrine glands secrete their products into the external environment by way of ducts, which empty into the gastrointestinal lumen or the external world.) A hormone receptor is a polypeptide that possesses a ligand-specific binding site. Binding of ligand (hormone) to the site causes the receptor to modify target cell activity. Tissue-specificity of hormone action is determined by whether the cells of a tissue have the appropriate receptor.
Some signaling molecules modify the activity of the cell which secreted them; this is an autocrine activity (auto- means self). For example, a T cell secretes interleukin 2, which binds to receptors on the same T cell to stimulate increased activity.
Hormones can be grouped into one of two classes. Hydrophilic hormones, such as peptides and amino-acid derivatives, must bind to receptors on the cell surface, while hydrophobic hormones, such as the steroid hormones, bind to receptors in the cellular interior.
Peptide Hormones
Peptide hormones are synthesized into the rough ER and modified in the Golgi. Then they are stored in vesicles until needed, when they are released by exocytosis. In the bloodstream they dissolve in the plasma, since they are hydrophilic. Their hydrophilicity also means they cannot cross biological membranes and thus are required to communicate with the interior of the target cell by way of a ___,41 discussed in Chapter 7. To briefly review, the peptide hormone is a first messenger which must bind to a cell-surface receptor. The receptor is a polypeptide with a domain on the inner surface of the plasma membrane that contains the ability to catalytically activate a second messenger. The end result of second messenger activation is that the function of proteins in the cytoplasm is changed. A key feature of second messenger cascades is signal amplification, which allows a few activated receptors to change the activity of many enzymes in the cytoplasm.
Because peptide hormones modify the activity of existing enzymes in the cytoplasm, their effects are exerted rapidly, minutes to hours from the time of secretion. Also, the duration of their effects is brief.
There are two subgroups within the peptide hormone category: polypeptides and amino acid derivatives. An example of a polypeptide hormone is insulin, which has a complex tertiary structure involving disulfide bridges. It is secreted by the β cells of the pancreatic islets of Langerhans in response to elevated blood glucose and binds to a cell-surface receptor with a cytoplasmic domain possessing protein kinase activity. Amino acid derivatives, as their name implies, are derived from single amino acids and contain no peptide bonds. For example, tyrosine is the parent amino acid for the catecholamines (which include epinephrine) and the thyroid hormones. Despite the fact that these two classes are derived from the same precursor molecule, they have different properties. The catecholamines act like peptide hormones, while the thyroid hormones behave more like steroid hormones. Epinephrine is a small cyclic molecule secreted by the adrenal medulla upon activation of the sympathetic nervous system. It binds to cell-surface receptors to trigger a cascade of events that produces the second messenger cyclic adenosine monophosphate (cAMP) and activates protein kinases in the cytoplasm. Thyroid hormones incorporate iodine into their structure. They enter cells, bind to DNA, and activate transcription of genes involved in energy mobilization.
Steroid Hormones
Steroids are hydrophobic molecules synthesized from cholesterol in the smooth endoplasmic reticulum. Due to their hydrophobicity, steroids can freely diffuse through biological membranes. Thus they are not stored but rather diffuse into the bloodstream as soon as they are made. If a steroid hormone is not needed, it will not be made. Steroids’ hydrophobicity also means they cannot be dissolved in the plasma. Instead they journey through the bloodstream stuck to proteins in the plasma, such as albumin. [What holds the steroid bound to a plasma protein?42] The small, hydrophobic steroid hormone exerts its effects upon target cells by diffusing through the plasma membrane to bind with a receptor in the cytoplasm. Once it has bound its ligand, the steroid hormone-receptor complex is transported into the nucleus, where it acts as a sequence-specific regulator of transcription. Because steroid hormones must modify transcription to change the amount and/or type of proteins in the cell, their effects are exerted slowly, over a period of days, and persist for days to weeks.
Steroids regulating sexuality, reproduction, and development are secreted by the testes, ovaries, and placenta. Steroids regulating water balance and other processes are secreted by the adrenal cortex. All other endocrine glands secrete peptide hormones. (Note that although thyroid hormone is derived from an amino acid, its mechanism of action more closely resembles that of the steroid hormones.)
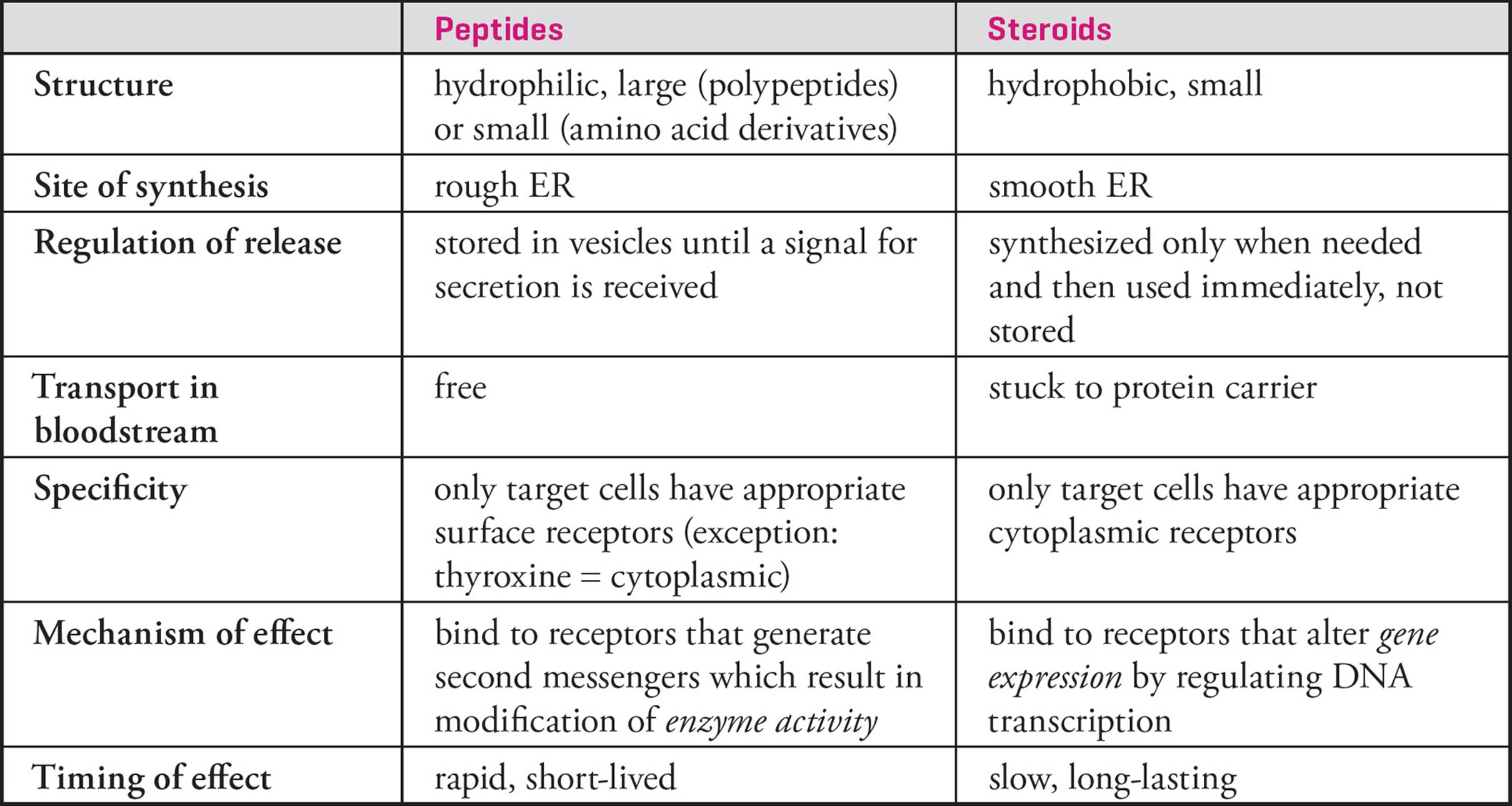
Table 6 Peptide vs. Steroid Hormones
Organization and Regulation of the Human Endocrine System
The endocrine system has many different roles. Hormones are essential for gamete synthesis, ovulation, pregnancy, growth, sexual development, and overall level of metabolic activity. Despite this diversity of function, endocrine activity is harmoniously orchestrated. Maintenance of order in such a complex system might seem impossible to accomplish in a preplanned manner. Regulation of the endocrine system is not preplanned or rigidly structured, but is instead generally automatic. Hormone levels rise and fall as dictated by physiological needs. The endocrine system is ordered yet dynamic. This flexible, automatic orderliness is attribuTable to feedback regulation. The amount of a hormone secreted is controlled not by a preformulated plan but rather by changes in the variable the hormone is responsible for controlling. Continuous circulation of blood exposes target cells to regulatory hormones and also exposes endocrine glands to serum concentrations of physiological variables that they regulate. Thus regulator and that which is regulated are in continuous communication. Concentration of a species X in the aqueous portion of the bloodstream is denoted “serum [X].”
An example of feedback regulation is the interaction between the hormone calcitonin and serum [Ca2+]. The function of calcitonin is to prevent serum [Ca2+] from peaking above normal levels, and the amount of calcitonin secreted is directly proportional to increases in serum [Ca2+] above normal. When serum [Ca2+] becomes elevated, calcitonin is secreted. Then when serum [Ca2+] levels fall, calcitonin secretion stops. The falling serum [Ca2+] level (that which is regulated) feeds back to the cells which secrete calcitonin (regulators). The serum [Ca2+] level is a physiological endpoint which must be maintained at constant levels. This demonstrates the role of the endocrine system in maintaining homeostasis, or physiological consistency.
An advantage of the endocrine system and its feedback regulation is that very complex arrays of variables can be controlled automatically. It’s as if the variables controlled themselves. However, some integration (a central control mechanism) is necessary. Superimposed upon the hormonal regulation of physiological endpoints is another layer of regulation: hormones that regulate hormones. Such meta-regulators are known as tropic hormones.
For example, adrenocorticotropic hormone (ACTH) is secreted by the anterior pituitary. The role of ACTH is to stimulate increased activity of the portion of the adrenal gland called the cortex, which is responsible for secreting cortisol (among other steroid hormones). ACTH is a tropic hormone because it does not directly affect physiological endpoints, but merely regulates another regulator (cortisol). Cortisol regulates physiological endpoints, including cellular responses to stress and serum [glucose]. Feedback regulation applies to tropic hormones as well as to direct regulators of physiological endpoints; the level of ACTH is influenced by the level of cortisol. When cortisol is needed, ACTH is secreted, and when the serum [cortisol] increases sufficiently, ACTH secretion slows.
You may have noticed that in both of our examples the effect of feedback was inhibitory: The result of hormone secretion inhibits further secretion. Inhibitory feedback is called negative feedback or feedback inhibition. Most feedback in the endocrine system (and if you remember, most biochemical feedback) is negative. There are few examples of positive feedback, which we will not discuss here. A key example will be discussed in Chapter 14 with the reproductive systems.
There is yet another layer of control. Many of the functions of the endocrine system depend on instructions from the brain. The portion of the brain which controls much of the endocrine system is the hypothalamus, located at the center of the brain. The hypothalamus controls the endocrine system by releasing tropic hormones that regulate other tropic hormones, called releasing and inhibiting factors or releasing and inhibiting hormones.
For example (Figure 20), the hypothalamus secretes corticotropin releasing hormone (CRH, also known as CRF, where “F” stands for factor). The role of CRH is to cause increased secretion of ACTH. Just as ACTH secretion is regulated by feedback inhibition from cortisol, CRH secretion, too, is inhibited by cortisol. You begin to see that regulatory pathways in the endocrine system can get pretty complex.
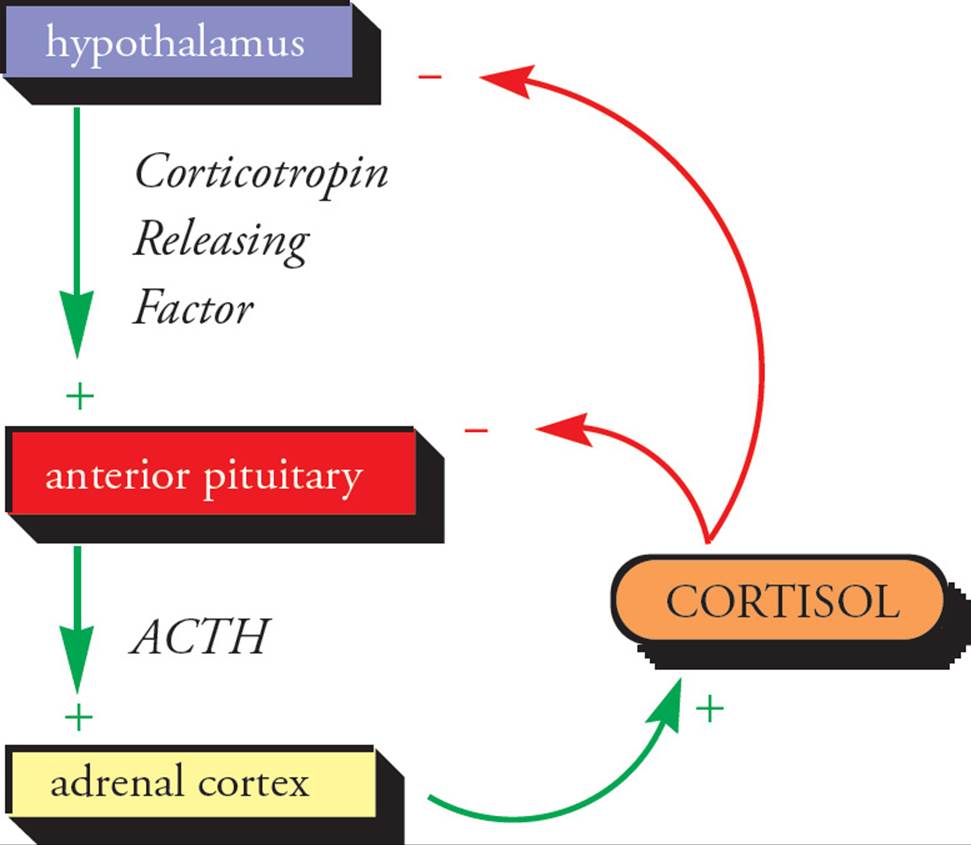
Figure 20 Feedback Regulation of Cortisol Secretion
Understanding that the hypothalamus controls the anterior pituitary and that the anterior pituitary controls most of the endocrine system is important. Damage to the connection between the hypothalamus and the pituitary is fatal, unless daily hormone replacement therapy is given. This endocrine control center is given a special name: hypothalamic-pituitary control axis (Figure 21). The hypothalamus exerts its control of the pituitary by secreting its hormones into the bloodstream, just like any other endocrine gland; what’s unique is that a special miniature circulatory system is provided for efficient transport of hypothalamic releasing and inhibiting factors to the anterior pituitary. This blood supply is known as the hypothalamic-pituitary portal system. You will also hear the term hypothalamic-hypophysial portal system. Hypophysis is another name for the pituitary gland.