CHEMISTRY THE CENTRAL SCIENCE
11 LIQUIDS AND INTERMOLECULAR FORCES
EXERCISES
VISUALIZING CONCEPTS
11.1 Does the diagram

best describe a crystalline solid, a liquid, or a gas? Explain. [Section 11.1]
11.2 (a) Which kind of intermolecular attractive force is shown in each case here?
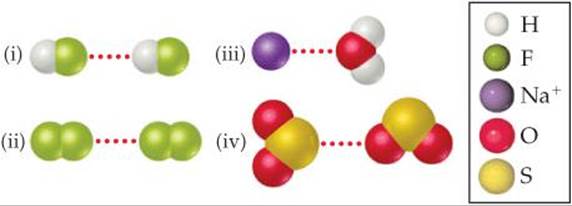
(b) Predict which of the four interactions is the weakest. [Section 11.2]
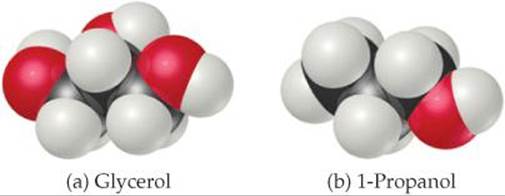
11.3 Do you expect the viscosity of glycerol, C3H5(OH)3, to be larger or smaller than that of 1-propanol, C3H7OH? Explain. [Section 11.3]
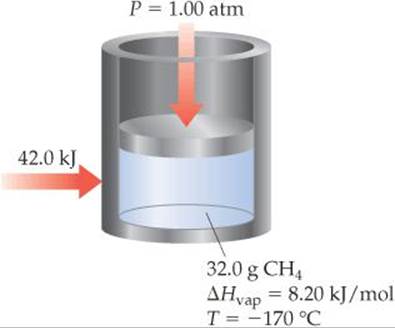
11.4 If 42.0 kJ of heat is added to a 32.0-g sample of liquid methane under 1 atm of pressure at a temperature of –170 °C, what are the final state and temperature of the methane once the system equilibrates? Assume no heat is lost to the surroundings. The normal boiling point of methane is –161.5 °C. The specific heats of liquid and gaseous methane are 3.48 and 2.22 J/g-K, respectively. [Section 11.4]
11.5 Using this graph of CS2 data,
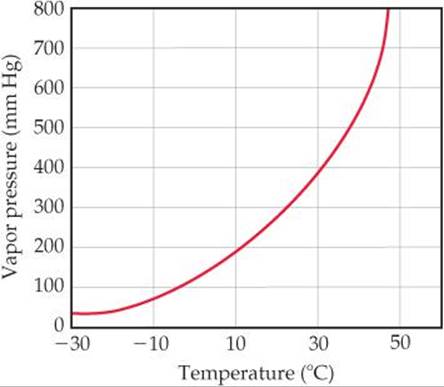
determine (a) the approximate vapor pressure of CS2 at 30 °C, (b) the temperature at which the vapor pressure equals 300 torr, (c) the normal boiling point of CS2. [Section 11.5]
11.6 The molecules
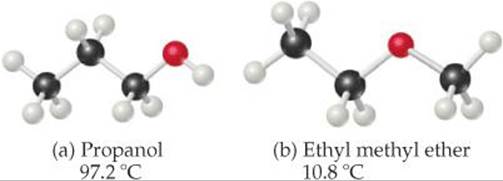
have the same molecular formula (C3H8O) but different normal boiling points, as shown. Rationalize the difference in boiling points. [Sections 11.2 and 11.5]
11.7 The phase diagram of a hypothetical substance is
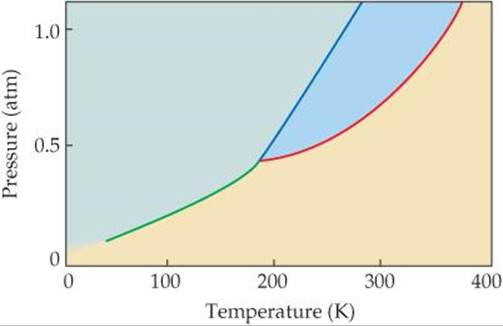
(a) Estimate the normal boiling point and freezing point of the substance.
(b) What is the physical state of the substance under the following conditions: (i) T = 150 K, P = 0.2 atm, (ii) T = 100 K, P = 0.8 atm, (iii) T = 300 K, P = 1.0 atm?
(c) What is the triple point of the substance? [Section 11.6]
11.8 At three different temperatures, T1, T2, and T3, the molecules in a liquid crystal align in these ways:
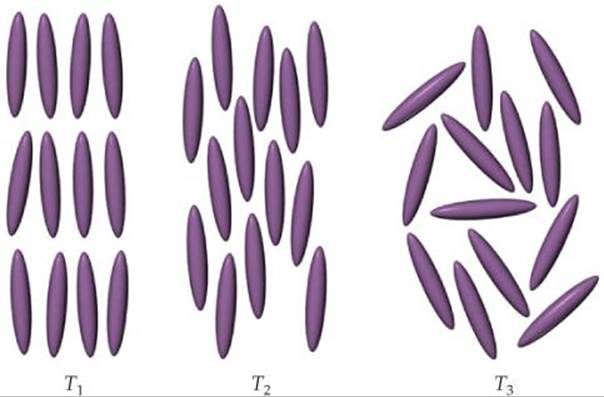
(a) At which temperature or temperatures is the substance in a liquid crystalline state? At those temperatures, which type of liquid crystalline phase is depicted?
(b) Which is the highest of the three temperatures?
MOLECULAR COMPARISONS OF GASES, LIQUIDS, AND SOLIDS (section 11.1)
11.9 List the three states of matter in order of (a) increasing molecular disorder and (b) increasing intermolecular attractions. (c) Which state of matter is most easily compressed?
11.10 (a) How does the average kinetic energy of molecules compare with the average energy of attraction between molecules in solids, liquids, and gases? (b) Why does increasing the temperature cause a solid substance to change in succession from a solid to a liquid to a gas? (c)What happens to a gas if you put it under extremely high pressure?
11.11 Arrange substances CCl4, Si, and Ar in order of increasing boiling point.
11.12 Arrange substances Ga, Ne, and Br2 in order of increasing boiling point.
11.13 At standard temperature and pressure the molar volume of Cl2 and NH3 gases are 22.06 L and 22.40 L, respectively. (a) Given the different molecular weights, dipole moments, and molecular shapes, why are their molar volumes nearly the same? (b) On cooling to 160 K, both substances form crystalline solids. Do you expect the molar volumes to decrease or increase on cooling to 160 K? (c) The densities of crystalline Cl2 and NH3 at 160 K are 2.02 g/cm3 and 0.84 g/cm3, respectively. Calculate their molar volumes. (d) Are the molar volumes in the solid state as similar as they are in the gaseous state? Explain. (e) Would you expect the molar volumes in the liquid state to be closer to those in the solid or gaseous state?
11.14 Benzoic acid, C6H5COOH, melts at 122 °C. The density in the liquid state at 130 °C is 1.08 g/cm3. The density of solid benzoic acid at 15 °C is 1.266 g/cm3. (a) In which of these two states is the average distance between molecules greater? (b) Explain the difference in densities at the two temperatures in terms of the relative kinetic energies of the molecules.
INTERMOLECULAR FORCES (section 11.2)
11.15 Which type of intermolecular attractive force operates between (a) all molecules, (b) polar molecules, (c) the hydrogen atom of a polar bond and a nearby small electronegative atom?
11.16 Based on what you have learned about intermolecular forces, would you say that matter is fundamentally attracted or repulsed by other matter?
11.17 Describe the intermolecular forces that must be overcome to convert these substances from a liquid to a gas: (a) SO2, (b) CH3COOH, (c) H2S.
11.18 Which type of intermolecular force accounts for each of these differences: (a) CH3OH boils at 65 °C; CH3SH boils at 6 °C. (b) Xe is liquid at atmospheric pressure and 120 K, whereas Ar is a gas under the same conditions. (c) Kr, atomic weight 84, boils at 120.9 K, whereas Cl2, molecular weight about 71, boils at 238 K. (d) Acetone boils at 56 °C, whereas 2-methylpropane boils at –12 °C.

11.19 (a) What is meant by the term polarizability? (b) Which of the following atoms would you expect to be most polarizable: N, P, As, Sb? Explain. (c) Put the following molecules in order of increasing polarizability: GeCl4, CH4, SiCl4, SiH4, and GeBr4. (d) Predict the order of boiling points of the substances in part (c).
11.20 True or false:
(a) For molecules with similar molecular weights, the dispersion forces become stronger as the molecules become more polarizable.
(b) For the noble gases the dispersion forces decrease while the boiling points increase as you go down the column in the periodic table.
(c) In terms of the total attractive forces for a given substance dipole–dipole interactions, when present, are always larger than dispersion forces.
(d) All other factors being the same, dispersion forces between linear molecules are greater than dispersion forces between molecules whose shapes are nearly spherical.
11.21 Which member in each pair has the larger dispersion forces: (a) H2O or H2S, (b) CO2 or CO, (c) SiH4 or GeH4?
11.22 Which member in each pair has the stronger intermolecular dispersion forces: (a) Br2 or O2, (b) CH3CH2CH2CH2SH or CH3CH2CH2CH2CH2SH, (c) CH3CH2CH2Cl or (CH3)2CHCl?
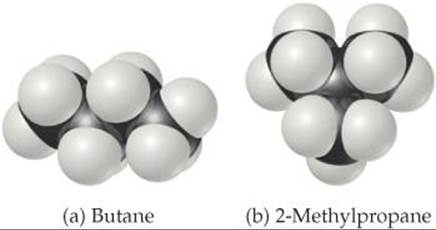
11.23 Butane and 2-methylpropane, whose space-filling models are shown at the top of the next column, are both nonpolar and have the same molecular formula, C4H10, yet butane has the higher boiling point (–0.5 °C compared to –11.7 °C). Explain.
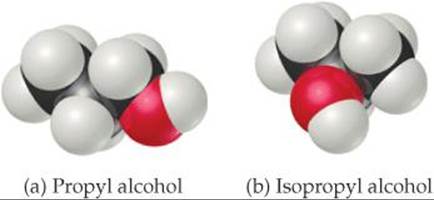
11.24 Propyl alcohol (CH3CH2CH2OH) and isopropyl alcohol [(CH3)2CHOH], whose space-filling models are shown, have boiling points of 97.2 °C and 82.5 °C, respectively. Explain why the boiling point of propyl alcohol is higher, even though both have the molecular formula C3H8O.
11.25 (a) What atoms must a molecule contain to participate in hydrogen bonding with other molecules of the same kind? (b) Which of the following molecules can form hydrogen bonds with other molecules of the same kind: CH3F, CH3NH2, CH3OH, CH3Br?
11.26 Rationalize the difference in boiling points in each pair: (a) HF (20 °C) and HCl (–85 °C), (b) CHCl3 (61 °C) and CHBr3 (150 °C), (c) Br2 (59 °C) and ICl (97 °C).
11.27 Ethylene glycol (HOCH2CH2OH), the major substance in antifreeze, has a normal boiling point of 198 °C. By comparison, ethyl alcohol (CH3CH2OH) boils at 78 °C at atmospheric pressure. Ethylene glycol dimethyl ether (CH3OCH2CH2OCH3) has a normal boiling point of 83 °C, and ethyl methyl ether (CH3CH2OCH3) has a normal boiling point of 11 °C. (a) Explain why replacement of a hydrogen on the oxygen by a CH3 group generally results in a lower boiling point. (b) What are the major factors responsible for the difference in boiling points of the two ethers?
11.28 Identify the type or types of intermolecular forces present in each substance and then select the substance in each pair that has the higher boiling point: (a) propane C3H8 or n-butane C4H10, (b) diethyl ether CH3CH2OCH2CH3 or 1-butanol CH3CH2CH2CH2OH, (c) sulfur dioxide SO2 or sulfur trioxide SO3, (d) phosgene Cl2CO or formaldehyde H2CO.
11.29 Look up and compare the normal boiling points and normal melting points of H2O and H2S. (a) Based on these physical properties, which substance has stronger intermolecular forces? What kind of intermolecular forces exist for each molecule? (b) Predict whether solid H2S is more or less dense than liquid H2S. How does this compare to H2O? Explain. (c) Water has an unusually high specific heat. Is this related to its inter-molecular forces? Explain.
11.30 The following quote about ammonia (NH3) is from a textbook of inorganic chemistry: “It is estimated that 26% of the hydrogen bonding in NH3 breaks down on melting, 7% on warming from the melting to the boiling point, and the final 67% on transfer to the gas phase at the boiling point.” From the standpoint of the kinetic energy of the molecules, explain (a) why there is a decrease of hydrogen-bonding energy on melting and (b) why most of the loss in hydrogen bonding occurs in the transition from the liquid to the vapor state.
11.31 A number of salts containing the tetrahedral polyatomic anion, BF4–, are ionic liquids, whereas salts containing the somewhat larger tetrahedral ion SO42– do not form ionic liquids. Explain this observation.
11.32 The generic structural formula for a 1-alkyl-3-methylimida-zolium cation is
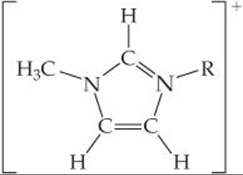
where R is a—CH2(CH2)nCH3 alkyl group. The melting points of the salts that form between the 1-alkyl-3-methylimidazolium cation and the PF6– anion are as follows:
R = CH2CH3 (m.p. = 60 °C),
R = CH2CH2CH3 (m.p. = 40 °C),
R = CH2CH2CH2CH3 (m.p. = 10 °C) and
R = CH2CH2CH2CH2CH2CH3 (m.p. = –61 °C). Why does
the melting point decrease as the length of alkyl group increases?
SELECT PROPERTIES OF LIQUIDS (section 11.3)
11.33 (a) Explain why surface tension and viscosity decrease with increasing temperature. (b) Why do substances with high surface tensions also tend to have high viscosities?
11.34 (a) Distinguish between adhesive forces and cohesive forces. (b) What adhesive and cohesive forces are involved when a paper towel absorbs water? (c) Explain the cause for the U-shaped meniscus formed when water is in a glass tube.
11.35 Explain the following observations: (a) The surface tension of CHBr3 is greater than that of CHCl3. (b) As temperature increases, oil flows faster through a narrow tube. (c) Raindrops that collect on a waxed automobile hood take on a nearly spherical shape. (d) Oil droplets that collect on a waxed automobile hood take on a flat shape.
11.36 Hydrazine (H2NNH2), hydrogen peroxide (HOOH), and water (H2O) all have exceptionally high surface tensions compared with other substances of comparable molecular weights. (a) Draw the Lewis structures for these three compounds. (b) What structural property do these substances have in common, and how might that account for the high surface tensions?
11.37 The boiling points, surface tensions, and viscosities of water and several alchohols are as follows:
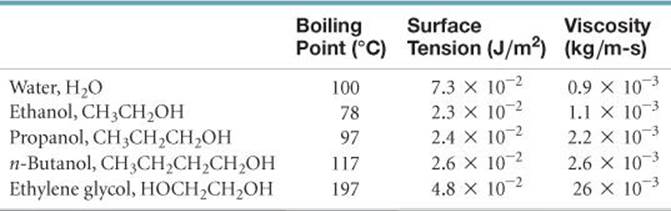
(a) For ethanol, propanol, and n-butanol the boiling points, surface tensions, and viscosities all increase. What is the reason for this increase? (b) How do you explain the fact that propanol and ethylene glycol have similar molecular weights (60 versus 62 amu), yet the viscosity of ethylene glycol is more than 10 times larger than propanol? (c) How do you explain the fact that water has the highest surface tension but the lowest viscosity?
11.38 (a) Would you expect the viscosity of n-pentane, CH3CH2CH2CH2CH3, to be larger or smaller than the viscosity of n-hexane (3.26 × 10–4 kg/m-s from Table 11.4)? (b) If you compared their viscosities at 270 K, would you expect the viscosity of neopentane, (CH3)4C, to be smaller or larger than n-pentane? (See Figure 11.6 to see the shapes of these molecules.)
PHASE CHANGES (section 11.4)
11.39 Name the phase transition in each of the following situations and indicate whether it is exothermic or endothermic: (a) When ice is heated, it turns to water. (b) Wet clothes dry on a warm summer day. (c) Frost appears on a window on a cold winter day. (d) Droplets of water appear on a cold glass of beer.
11.40 Name the phase transition in each of the following situations and indicate whether it is exothermic or endothermic: (a) Bromine vapor turns to bromine liquid as it is cooled. (b) Crystals of iodine disappear from an evaporating dish as they stand in a fume hood. (c) Rubbing alcohol in an open container slowly disappears. (d) Molten lava from a volcano turns into solid rock.
11.41 Explain why any substance's heat of fusion is generally lower than its heat of vaporization.
11.42 Ethyl chloride (C2H5Cl) boils at 12 °C. When liquid C2H5Cl under pressure is sprayed on a room-temperature (25 °C) surface in air, the surface is cooled considerably. (a) What does this observation tell us about the specific heat of C2H5Cl(g) as compared with C2H5Cl(l)? (b)Assume that the heat lost by the surface is gained by ethyl chloride. What enthalpies must you consider if you were to calculate the final temperature of the surface?
11.43 For many years drinking water has been cooled in hot climates by evaporating it from the surfaces of canvas bags or porous clay pots. How many grams of water can be cooled from 35 °C to 20 °C by the evaporation of 60 g of water? (The heat of vaporization of water in this temperature range is 2.4 kJ/g. The specific heat of water is 4.18 J/g-K.)
11.44 Compounds like CCl2F2 are known as chlorofluorocarbons, or CFCs. These compounds were once widely used as refrigerants but are now being replaced by compounds that are believed to be less harmful to the environment. The heat of vaporization of CCl2F2 is 289 J/g. What mass of this substance must evaporate to freeze 200 g of water initially at 15 °C? (The heat of fusion of water is 334 J/g; the specific heat of water is 4.18 J/g-K.)
11.45 Ethanol (C2H5OH) melts at –114 °C and boils at 78 °C. The enthalpy of fusion of ethanol is 5.02 kJ/mol, and its enthalpy of vaporization is 38.56 kJ/mol. The specific heats of solid and liquid ethanol are 0.97 J/g-K and 2.3 J/g-K, respectively. (a) How much heat is required to convert 42.0 g of ethanol at 35 °C to the vapor phase at 78 °C? (b) How much heat is required to convert the same amount of ethanol at –155 °C to the vapor phase at 78 °C?
11.46 The fluorocarbon compound C2Cl3F3 has a normal boiling point of 47.6 °C. The specific heats of C2Cl3F3(l) and C2Cl3F3(g) are 0.91 J/g-K and 0.67 J/g-K, respectively. The heat of vaporization for the compound is 27.49 kJ/mol. Calculate the heat required to convert 35.0 g of C2Cl3F3 from a liquid at 10.00 °C to a gas at 105.00 °C.
11.47 (a) What is the significance of the critical pressure of a substance? (b) What happens to the critical temperature of a series of compounds as the force of attraction between molecules increases? (c) Which of the substances listed in Table 11.5 can be liquefied at the temperature of liquid nitrogen (–196 °C)?
11.48 The critical temperatures (K) and pressures (atm) of a series of halogenated methanes are as follows:
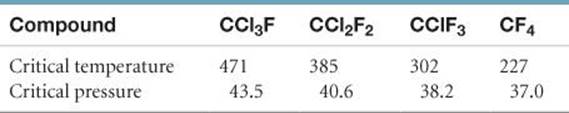
(a) List the intermolecular forces that occur for each compound. (b) Predict the order of increasing intermolecular attraction, from least to most, for this series of compounds. (c) Predict the critical temperature and pressure for CCl4 based on the trends in this table. Look up the experimentally determined critical temperatures and pressures for CCl4, using a source such as the CRC Handbook of Chemistry and Physics, and suggest a reason for any discrepancies.
VAPOR PRESSURE (section 11.5)
11.49 Explain how each of the following affects the vapor pressure of a liquid: (a) volume of the liquid, (b) surface area, (c) inter-molecular attractive forces, (d) temperature, (e) density of the liquid.
11.50 Acetone, H3CCOCH3, has a boiling point of 56 °C. Based on the data given in Figure 11.25, would you expect acetone to have a higher or lower vapor pressure than ethanol at 25 °C?
11.51 (a) Place the following substances in order of increasing volatility: CH4, CBr4, CH2Cl2, CH3Cl, CHBr3, and CH2Br2. Explain. (b) How do the boiling points vary through this series?
11.52 True or false:
(a) CBr4 is more volatile than CCl4.
(b) CBr4 has a higher boiling point than CCl4.
(c) CBr4 has weaker intermolecular forces than CCl4.
(d) CBr4 has a higher vapor pressure at the same temperature than CCl4.
11.53 (a) Two pans of water are on different burners of a stove. One pan of water is boiling vigorously, while the other is boiling gently. What can be said about the temperature of the water in the two pans? (b) A large container of water and a small one are at the same temperature. What can be said about the relative vapor pressures of the water in the two containers?
11.54 Explain the following observations: (a) Water evaporates more quickly on a hot, dry day than on a hot, humid day. (b) It takes longer to cook an egg in boiling water at high altitudes than it does at lower altitudes.
11.55 Using the vapor-pressure curves in Figure 11.25, (a) estimate the boiling point of ethanol at an external pressure of 200 torr; (b) estimate the external pressure at which ethanol will boil at 60 °C; (c) estimate the boiling point of diethyl ether at 400 torr; (d) estimate the external pressure at which diethyl ether will boil at 40 °C.
11.56 Appendix B lists the vapor pressure of water at various external pressures.
(a) Plot the data in Appendix B, vapor pressure (torr) versus temperature (°C). From your plot, estimate the vapor pressure of water at body temperature, 37 °C.
(b) Explain the significance of the data point at 760.0 torr, 100 °C.
(c) A city at an altitude of 5000 ft above sea level has a barometric pressure of 633 torr. To what temperature would you have to heat water to boil it in this city?
(d) A city at an altitude of 500 ft below sea level would have a barometric pressure of 774 torr. To what temperature would you have to heat water to boil it in this city?
(e) For the two cities in parts (c) and (d), compare the average kinetic energies of the water molecules at their boiling points. Are the kinetic energies the same or different? Explain.
PHASE DIAGRAMS (section 11.6)
11.57 (a) What is the significance of the critical point in a phase diagram? (b) Why does the line that separates the gas and liquid phases end at the critical point?
11.58 (a) What is the significance of the triple point in a phase diagram? (b) Could you measure the triple point of water by measuring the temperature in a vessel in which water vapor, liquid water, and ice are in equilibrium under one atmosphere of air? Explain.
11.59 Referring to Figure 11.28, describe all the phase changes that would occur in each of the following cases: (a) Water vapor originally at 0.005 atm and –0.5 °C is slowly compressed at constant temperature until the final pressure is 20 atm. (b) Water originally at 100.0 °C and 0.50 atm is cooled at constant pressure until the temperature is –10 °C.
11.60 Referring to Figure 11.29, describe the phase changes (and the temperatures at which they occur) when CO2 is heated from –80 °C to –20 °C at (a) a constant pressure of 3 atm, (b) a constant pressure of 6 atm.
11.61 The phase diagram for neon is
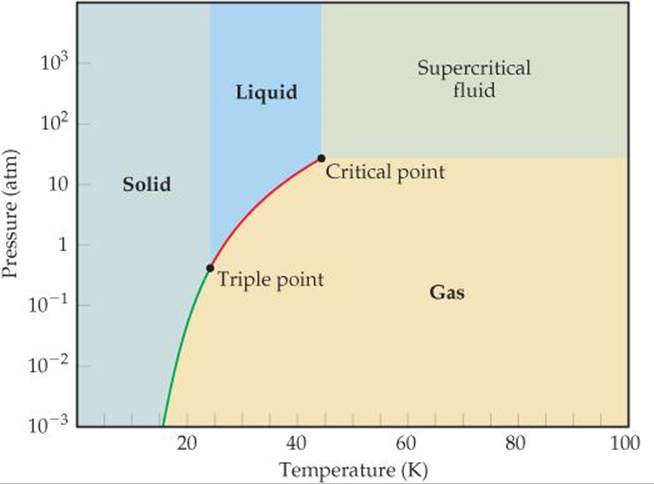
Use the phase diagram to answer the following questions. (a) What is the approximate value of the normal melting point? (b) Over what pressure range will solid neon sublime? (c) At room temperature (T = 25 °C) can neon be liquefied by compressing it?
11.62 Use the phase diagram of neon to answer the following questions. (a) What is the approximate value of the normal boiling point? (b) What can you say about the strength of the intermolecular forces in neon and argon based on the critical points of Ne and Ar? (See Table 11.5.)
11.63 The fact that water on Earth can readily be found in all three states (solid, liquid, and gas) is in part a consequence of the fact that the triple point of water (T = 0.01 °C, P = 0.006 atm) falls within a range of temperatures and pressures found on Earth. Saturn's largest moon Titan has a considerable amount of methane in its atmosphere. The conditions on the surface of Titan are estimated to be P = 1.6 atm and T = –178 °C. As seen from the phase diagram of methane (Figure 11.30), these conditions are not far from the triple point of methane, raising the tantalizing possibility that solid, liquid, and gaseous methane can be found on Titan. (a) What state would you expect to find methane in on the surface of Titan? (b) On moving upward through the atmosphere the pressure will decrease. If we assume that the temperature does not change, what phase change would you expect to see as we move away from the surface?
11.64 At 25 °C gallium is a solid with a density of 5.91 g/cm3. Its melting point, 29.8 °C, is low enough that you can melt it by holding it in your hand. The density of liquid gallium just above the melting point is 6.1 g/cm3. Based on this information, what unusual feature would you expect to find in the phase diagram of gallium?
LIQUID CRYSTALS (section 11.7)
11.65 In terms of the arrangement and freedom of motion of the molecules, how are the nematic liquid crystalline phase and an ordinary liquid phase similar? How are they different?
11.66 What observations made by Reinitzer on cholesteryl benzoate suggested that this substance possesses a liquid crystalline phase?
11.67 The molecules shown in Figure 11.33 possess polar groups (that is, groupings of atoms that give rise to sizable dipole moments within the molecules). How might the presence of polar groups enhance the tendency toward liquid crystal formation?
11.68 One of the more effective liquid crystalline substances employed in LCDs is the molecule
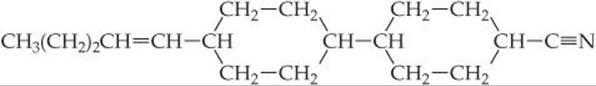
(a) How many double bonds are there in this molecule? (b) Describe the features of the molecule that make it prone to show liquid crystalline behavior.
11.69 For a given substance, the liquid crystalline phase tends to be more viscous than the liquid phase. Why?
11.70 Describe how a cholesteric liquid crystal phase differs from a nematic phase.
11.71 It often happens that a substance possessing a smectic liquid crystalline phase just above the melting point passes into a nematic liquid crystalline phase at a higher temperature. Account for this type of behavior.
11.72 The smectic liquid crystalline phase can be said to be more highly ordered than the nematic phase. In what sense is this true?
ADDITIONAL EXERCISES
11.73 As the intermolecular attractive forces between molecules increase in magnitude, do you expect each of the following to increase or decrease in magnitude? (a) vapor pressure, (b) heat of vaporization, (c) boiling point, (d) freezing point, (e) viscosity, (f) surface tension, (g) critical temperature.
11.74 Suppose you have two colorless molecular liquids, one boiling at –84 °C, the other at 34 °C, and both at atmospheric pressure. Which of the following statements is correct? For each statement that is not correct, modify the statement so that it is correct. (a) The higher-boiling liquid has greater total intermolecular forces than the lower-boiling liquid. (b) The lower-boiling liquid must consist of nonpolar molecules. (c) The lower-boiling liquid has a lower molecular weight than the higher-boiling liquid. (d) The two liquids have identical vapor pressures at their normal boiling points. (e) At –84 °C both liquids have vapor pressures of 760 mm Hg.
11.75 Two isomers of the planar compound 1,2-dichloroethylene are shown here.
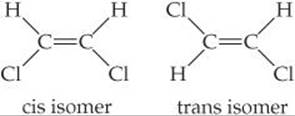
(a) Which of the two isomers will have the stronger dipole–dipole forces? (b) One isomer has a boiling point of 60.3 °C and the other 47.5 °C. Which isomer has which boiling point?
11.76 In dichloromethane, CH2Cl2(μ = 1.60 D), the dispersion force contribution to the intermolecular attractive forces is about five times larger than the dipole–dipole contribution. Compared to CH2Cl2, would you expect the relative importance of the dipole–dipole contribution to increase or decrease (a) in dibromomethane (μ = 1.43 D), (b) in difluoromethane (μ = 1.93 D)? Explain.
11.77 When an atom or group of atoms is substituted for an H atom in benzene (C6H6), the boiling point changes. Explain the order of the following boiling points: C6H6 (80 °C), C6H5Cl (132 °C), C6H5Br (156 °C), C6H5OH (182 °C).
11.78 The DNA double helix (Figure 24.30) at the atomic level looks like a twisted ladder, where the “rungs” of the ladder consist of molecules that are hydrogen-bonded together. Sugar and phosphate groups make up the sides of the ladder. Shown are the structures of the adenine-thymine (AT) “base pair” and the guanine-cytosine (GC) base pair:
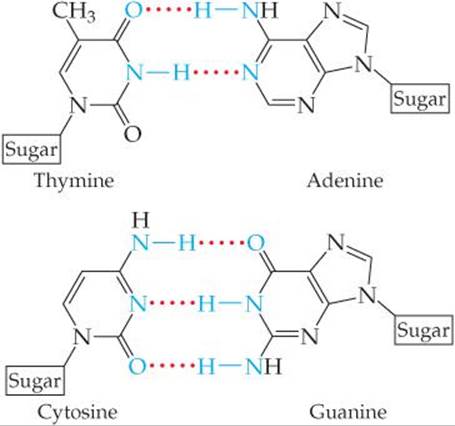
You can see that AT base pairs are held together by two hydrogen bonds, and the GC base pairs are held together by three hydrogen bonds. Which base pair is more stable to heating? Why?
11.79 Ethylene glycol (HOCH2CH2OH) is the major component of antifreeze. It is a slightly viscous liquid, not very volatile at room temperature, with a boiling point of 198 °C. Pentane (C5H12), which has about the same molecular weight, is a nonviscous liquid that is highly volatile at room temperature and whose boiling point is 36.1 °C. Explain the differences in the physical properties of the two substances.
11.80 Use the normal boiling points
propane, C3H8, –42.1 °C
butane, C4H10, –0.5 °C
pentane, C5H12, 36.1 °C
hexane, C6H14, 68.7 °C
heptane, C7H16, 98.4 °C
to estimate the normal boiling point of octane, C8H18. Explain the trend in the boiling points.
11.81 One of the attractive features of ionic liquids is their low vapor pressure, which in turn tends to make them nonflammable. Why do you think ionic liquids have lower vapor pressures than most room-temperature molecular liquids?
11.82 (a) When you exercise vigorously, you sweat. How does this help your body cool? (b) A flask of water is connected to a vacuum pump. A few moments after the pump is turned on, the water begins to boil. After a few minutes, the water begins to freeze. Explain why these processes occur.
[11.83] The following table gives the vapor pressure of hexafluorobenzene (C6F6) as a function of temperature:
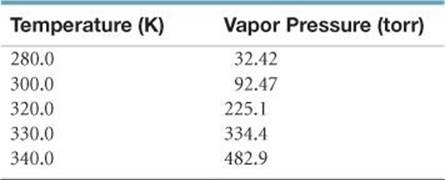
(a) By plotting these data in a suitable fashion, determine whether the Clausius–Clapeyron equation (Equation 11.1) is obeyed. If it is obeyed, use your plot to determine ΔHvap for C6F6. (b) Use these data to determine the boiling point of the compound.
[11.84] Suppose the vapor pressure of a substance is measured at two different temperatures. (a) By using the Clausius–Clapeyron equation (Equation 11.1) derive the following relationship between the vapor pressures, P1 and P2, and the absolute temperatures at which they were measured, T1 and T2:
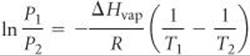
(b) Gasoline is a mixture of hydrocarbons, a major component of which is octane, CH3CH2CH2CH2CH2CH2CH2CH3. Octane has a vapor pressure of 13.95 torr at 25 °C and a vapor pressure of 144.78 torr at 75 °C. Use these data and the equation in part (a) to calculate the heat of vaporization of octane. (c) By using the equation in part (a) and the data given in part (b), calculate the normal boiling point of octane. Compare your answer to the one you obtained from Exercise 11.80. (d) Calculate the vapor pressure of octane at –30 °C.
[11.85] The following data present the temperatures at which certain vapor pressures are achieved for dichloromethane (CH2Cl2) and methyl iodide (CH3I):
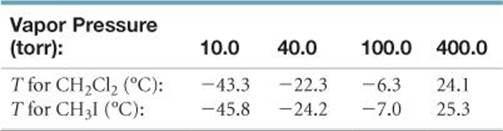
(a) Which of the two substances is expected to have the greater dipole–dipole forces? Which is expected to have the greater dispersion forces? Based on your answers, explain why it is difficult to predict which compound would be more volatile. (b) Which compound would you expect to have the higher boiling point? Check your answer in a reference book such as the CRC Handbook of Chemistry and Physics. (c) The order of volatility of these two substances changes as the temperature is increased. What quantity must be different for the two substances in order for this phenomenon to occur? (d) Substantiate your answer for part (c) by drawing an appropriate graph.
11.86 A watch with a liquid crystal display (LCD) does not function properly when it is exposed to low temperatures during a trip to Antarctica. Explain why the LCD might not function well at low temperature.
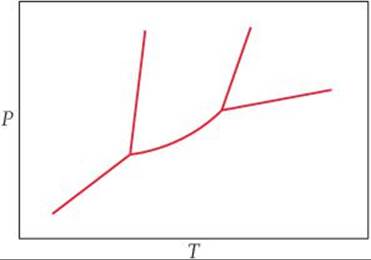
[11.87] A particular liquid crystalline substance has the phase diagram shown in the figure. By analogy with the phase diagram for a non–liquid crystalline substance, identify the phase present in each area.
INTEGRATIVE EXERCISES
11.88 (a) At the molecular level, what factor is responsible for the steady increase in viscosity with increasing molecular weight in the hydrocarbon series shown in Table 11.4? (b) Although the viscosity varies over a factor of more than two in the series from hexane to nonane, the surface tension at 25 °C increases by only about 20% in the same series. How do you account for this? (c) n-Octyl alcohol, CH3(CH2)7OH, has a viscosity of 1.01 × 10–2 kg/m-s, much higher than nonane, which has about the same molecular weight. What accounts for this difference? How does your answer relate to the difference in normal boiling points for these two substances?
11.89 Acetone, (CH3)2CO, is widely used as an industrial solvent. (a) Draw the Lewis structure for the acetone molecule and predict the geometry around each carbon atom. (b) Is the acetone molecule polar or nonpolar? (c) What kinds of intermolecular attractive forces exist between acetone molecules? (d) 1-Propanol, CH3CH2CH2OH, has a molecular weight that is very similar to that of acetone, yet acetone boils at 56.5 °C and 1-propanol boils at 97.2 °C. Explain the difference.
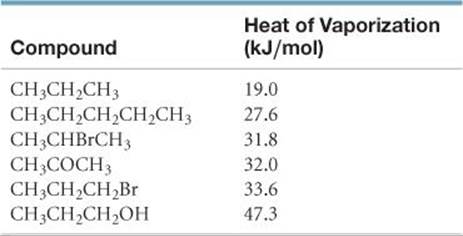
11.90 The table shown here lists the molar heats of vaporization for several organic compounds. Use specific examples from this list to illustrate how the heat of vaporization varies with (a) molar mass, (b) molecular shape, (c) molecular polarity, (d) hydrogen-bonding interactions. Explain these comparisons in terms of the nature of the intermolecular forces at work. (You may find it helpful to draw out the structural formula for each compound.)
11.91 Liquid butane, C4H10, is stored in cylinders to be used as a fuel. The normal boiling point of butane is listed as –0.5 °C. (a) Suppose the tank is standing in the sun and reaches a temperature of 35 °C. Would you expect the pressure in the tank to be greater or less than atmospheric pressure? How does the pressure within the tank depend on how much liquid butane is in it? (b) Suppose the valve to the tank is opened and a few liters of butane are allowed to escape rapidly. What do you expect would happen to the temperature of the remaining liquid butane in the tank? Explain. (c) How much heat must be added to vaporize 250 g of butane if its heat of vaporization is 21.3 kJ/mol? What volume does this much butane occupy at 755 torr and 35 °C?
[11.92] Using information in Appendices B and C, calculate the minimum number of grams of propane, C3H8(g), that must be combusted to provide the energy necessary to convert 5.50 kg of ice at –20 °C to liquid water at 75 °C.
11.93 The vapor pressure of a volatile liquid can be determined by slowly bubbling a known volume of gas through it at a known temperature and pressure. In an experiment, 5.00 L of N2 gas is passed through 7.2146 g of liquid benzene, C6H6, at 26.0 °C. The liquid remaining after the experiment weighs 5.1493 g. Assuming that the gas becomes saturated with benzene vapor and that the total gas volume and temperature remain constant, what is the vapor pressure of the benzene in torr?
11.94 The relative humidity of air equals the ratio of the partial pressure of water in the air to the equilibrium vapor pressure of water at the same temperature times 100%. If the relative humidity of the air is 58% and its temperature is 68 °F, how many molecules of water are present in a room measuring 12 ft × 10 ft × 8 ft?