CHEMISTRY THE CENTRAL SCIENCE
24 THE CHEMISTRY OF LIFE: ORGANIC AND BIOLOGICAL CHEMISTRY

TO COMMUNICATE WITH OTHER members of their species, insects release chemicals called pheromones into their environment.
WHAT'S AHEAD
24.1 GENERAL CHARACTERISTICS OF ORGANIC MOLECULES
We begin with a review of the structures and reactivities of organic compounds.
24.2 INTRODUCTION TO HYDROCARBONS
We consider hydrocarbons, compounds containing only C and H, including the hydrocarbons called alkanes, which contain only single bonds. We also look at isomers, compounds with identical compositions but different molecular structures.
24.3 ALKENES, ALKYNES, AND AROMATIC HYDROCARBONS
We next explore hydrocarbons with one or more ![]() bonds, called alkenes, and those with one or more
bonds, called alkenes, and those with one or more ![]() bonds, called alkynes. Aromatic hydrocarbons have at least one planar ring with delocalized π electrons.
bonds, called alkynes. Aromatic hydrocarbons have at least one planar ring with delocalized π electrons.
24.4 ORGANIC FUNCTIONAL GROUPS
We recognize that a central organizing principle of organic chemistry is the functional group, a group of atoms at which most of the compound's chemical reactions occur.
24.5 CHIRALITY IN ORGANIC CHEMISTRY
We learn that compounds with nonsuperimposable mirror images are chiral and that chirality plays important roles in organic and biological chemistry.
24.6 INTRODUCTION TO BIOCHEMISTRY
We introduce the chemistry of living organisms, known as biochemistry, biological chemistry, or chemical biology. Important classes of compounds that occur in living systems are proteins, carbohydrates, lipids, and nucleic acids.
24.7 PROTEINS
We learn that proteins are polymers of amino acids linked with amide (also called peptide) bonds. Proteins are used for structural support and as molecular transporters and enzymes.
24.8 CARBOHYDRATES
We observe that carbohydrates are sugars and polymers of sugars used primarily as fuel by organisms (glucose) or as structural support in plants (cellulose).
24.9 LIPIDS
We recognize that lipids are a large class of molecules used primarily for energy storage in organisms.
24.10 NUCLEIC ACIDS
We learn that nucleic acids are polymers of nucleotides that contain an organism's genetic information. Deoxyribonucleic acid (DNA) and ribonucleic acid (RNA) are polymers composed of nucleotides.
INSECTS COMMUNICATE BY RELEASING substances called pheromones, which they detect with their antennae. There are sex, alarm, defense, and trail pheromones. For example, isoamyl acetate [3-methylbutyl acetate, (CH3)2CHCH2COOCH3] is an alarm pheromone for bees, attracting other bees and provoking them to sting. Mammals, including humans, may also respond to pheromones, although the identity and function of pheromones in humans are not conclusive. Nevertheless, google “pheromone” and you will find hundreds of sources trying to sell you a pheromone, claiming that it will make you irresistible to the opposite sex.
Although biological systems are almost unimaginably complex, they are nevertheless constructed of molecules of quite modest size, as, for instance, the isoamyl acetate pheromone just described. To understand biology, therefore, we need to understand the chemical behaviors of molecules. This chapter is about the molecules, composed mainly of carbon, hydrogen, oxygen, and nitrogen, that form the basis of organic and biological chemistry.
More than 16 million carbon-containing compounds are known. Chemists make thousands of new compounds every year, about 90% of which contain carbon. The study of compounds whose molecules contain carbon constitutes the branch of chemistry known as organic chemistry. This term arose from the eighteenth-century belief that organic compounds could be formed only by living (that is, organic) systems. This idea was disproved in 1828 by the German chemist Friedrich Wöhler when he synthesized urea (H2NCONH2), an organic substance found in the urine of mammals, by heating ammonium cyanate (NH4OCN), an inorganic (“nonliving”) substance.
The study of the chemistry of living species is called biological chemistry, chemical biology, or biochemistry. In this chapter, we present some of the elementary aspects of both organic chemistry and biochemistry.
24.1 GENERAL CHARACTERISTICS OF ORGANIC MOLECULES
What is it about carbon that leads to the tremendous diversity in its compounds and allows it to play such crucial roles in biology and society? Let's consider some general features of organic molecules and, as we do, review principles we learned in earlier chapters.
The Structures of Organic Molecules
Because carbon has four valence electrons ([He]2s22p2), it forms four bonds in virtually all its compounds. When all four bonds are single bonds, the electron pairs are disposed in a tetrahedral arrangement. ![]() (Section 9.2) In the hybridization model, the carbon 2s and 2p orbitals are thensp3 hybridized.
(Section 9.2) In the hybridization model, the carbon 2s and 2p orbitals are thensp3 hybridized. ![]() (Section 9.5) When there is one double bond, the arrangement is trigonal planar (sp hybridization). With a triple bond, it is linear (sp hybridization). Examples are shown in
(Section 9.5) When there is one double bond, the arrangement is trigonal planar (sp hybridization). With a triple bond, it is linear (sp hybridization). Examples are shown in ![]() FIGURE 24.1.
FIGURE 24.1.
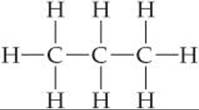
Almost every organic molecule contains C—H bonds. Because the valence shell of H can hold only two electrons, hydrogen forms only one covalent bond. As a result, hydrogen atoms are always located on the surface of organic molecules whereas the C—C bonds form the backbone, or skeleton, of the molecule, as in the propane molecule:
![]() GO FIGURE
GO FIGURE
What is the geometry around the bottom carbon atom in acetonitrile?
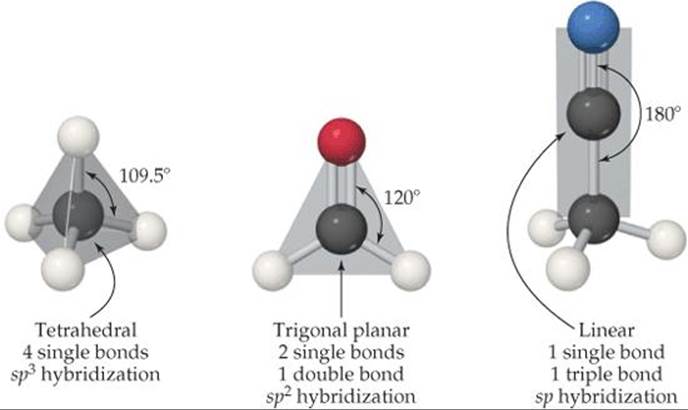
![]() FIGURE 24.1 Carbon geometries. The three common geometries around carbon are tetrahedral as in methane (CH4), trigonal planar as in formaldehyde (CH2O), and linear as in acetonitrile (CH3CN). Notice that in all cases each carbon atom forms four bonds.
FIGURE 24.1 Carbon geometries. The three common geometries around carbon are tetrahedral as in methane (CH4), trigonal planar as in formaldehyde (CH2O), and linear as in acetonitrile (CH3CN). Notice that in all cases each carbon atom forms four bonds.
The Stabilities of Organic Substances
Carbon forms strong bonds with a variety of elements, especially H, O, N, and the halogens. ![]() (Section 8.8) Carbon also has an exceptional ability to bond to itself, forming a variety of molecules made up of chains or rings of carbon atoms. Most reactions with low or moderate activation energy (Section 14.5) begin when a region of high electron density on one molecule encounters a region of low electron density on another molecule. The regions of high electron density may be due to the presence of a multiple bond or to the more electronegative atom in a polar bond. Because of their strength and lack of polarity, both C—C single bonds and C—H bonds are relatively unreactive. To better understand the implications of these facts, consider ethanol:
(Section 8.8) Carbon also has an exceptional ability to bond to itself, forming a variety of molecules made up of chains or rings of carbon atoms. Most reactions with low or moderate activation energy (Section 14.5) begin when a region of high electron density on one molecule encounters a region of low electron density on another molecule. The regions of high electron density may be due to the presence of a multiple bond or to the more electronegative atom in a polar bond. Because of their strength and lack of polarity, both C—C single bonds and C—H bonds are relatively unreactive. To better understand the implications of these facts, consider ethanol:
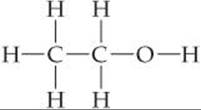
The differences in the electronegativity values of C (2.5) and O (3.5) and of O and H (2.1) indicate that the C—O and O—H bonds are quite polar. Thus, many reactions of ethanol involve these bonds while the hydrocarbon portion of the molecule remains intact. A group of atoms such as the C—O—H group, which determines how an organic molecule reacts (in other words, how the molecule functions), is called a functional group. The functional group is the center of reactivity in an organic molecule.
![]() GO FIGURE
GO FIGURE
How would replacing OH groups on ascorbic acid with CH3 groups affect the substance's solubility in (a) polar solvents and (b) nonpolar solvents?
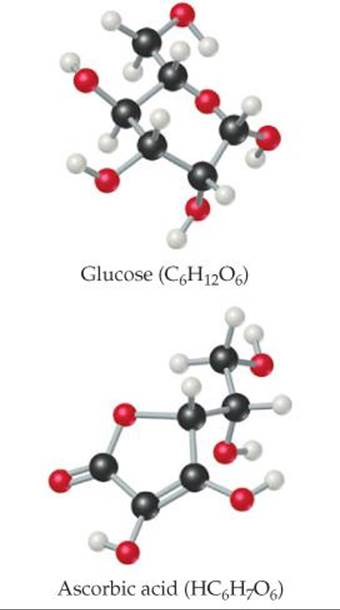
![]() FIGURE 24.2 Organic molecules soluble in polar solvents.
FIGURE 24.2 Organic molecules soluble in polar solvents.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Which bond is most likely to be the location of a chemical reaction: C=N, C—C, or C—H?
Solubility and Acid-Base Properties of Organic Substances
In most organic substances, the most prevalent bonds are carbon–carbon and carbon–hydrogen, which have low polarity. For this reason, the overall polarity of organic molecules is often low, which makes them generally soluble in nonpolar solvents and not very soluble in water. ![]() (Section 13.3) Organic molecules that are soluble in polar solvents are those that have polar groups on the molecule surface, such as glucose and ascorbic acid (
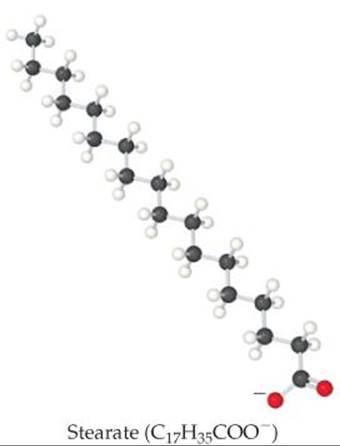
(Section 13.3) Organic molecules that are soluble in polar solvents are those that have polar groups on the molecule surface, such as glucose and ascorbic acid (![]() FIGURE 24.2). Organic molecules that have a long, nonpolar part bonded to a polar, ionic part, such as the stearate ion shown inFigure 24.2, function as surfactants and are used in soaps and detergents.
FIGURE 24.2). Organic molecules that have a long, nonpolar part bonded to a polar, ionic part, such as the stearate ion shown inFigure 24.2, function as surfactants and are used in soaps and detergents. ![]() (Section 13.6) The nonpolar part of the molecule extends into a nonpolar medium such as grease or oil, and the polar part extends into a polar medium such as water.
(Section 13.6) The nonpolar part of the molecule extends into a nonpolar medium such as grease or oil, and the polar part extends into a polar medium such as water.
Many organic substances contain acidic or basic groups. The most important acidic organic substances are the carboxylic acids, which bear the functional group COOH. ![]() (Sections 4.3 and 16.10) The most important basic organic substances are amines, which bear the—NH2,—NHR,—or NR2 groups, where R is an organic group made up of carbon and hydrogen atoms.
(Sections 4.3 and 16.10) The most important basic organic substances are amines, which bear the—NH2,—NHR,—or NR2 groups, where R is an organic group made up of carbon and hydrogen atoms. ![]() (Section 16.7)
(Section 16.7)
As you read this chapter, you will find many concept links (![]() ) to related materials in earlier chapters. We strongly encourage you to follow these links and review the earlier material. Doing so will enhance your understanding and appreciation of organic chemistry and biochemistry.
) to related materials in earlier chapters. We strongly encourage you to follow these links and review the earlier material. Doing so will enhance your understanding and appreciation of organic chemistry and biochemistry.