SAT Physics Subject Test
Chapter 15 Thermal Physics
Chapter 15 Review Questions
See Chapter 17 for solutions.
1. How much heat is required to raise the temperature of a 0.04 kg stainless steel spoon from 20°C to 50°C if the specific heat of stainless steel is 0.50 kJ/kg × °C ?
(A) 200 J
(B) 400 J
(C) 600 J
(D) 800 J
(E) 1,000 J
2. The melting point of copper is 1,080°C and its heat of fusion is 200 kJ/kg. If a copper coin at this temperature is completely melted by the absorption of 2,000 J of heat, what is the mass of the coin?
(A) 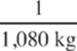
(B) 
(C) 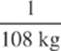
(D) 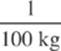
(E) 
3. Water has the specific heat 4.186 kJ/kg·°C, a boiling point of 100°C, and a heat of vaporization of 2,260 kJ/kg. A sealed beaker contains 100 g of water that”s initially at 20°C. If the water absorbs 100 kJ of heat, what will its final temperature be?
(A) 100°C
(B) 119°C
(C) 143°C
(D) 183°C
(E) 239°C
4. On a cold winter day (5°C), the foundation block for a statue is filled with 2.0 m3 of concrete. By how much will the concrete”s volume increase on a very warm summer day (35°C) if its coefficient of volume expansion is 4.0 × 10–5/°C ?
(A) 160 cm3
(B) 1,200 cm3
(C) 1,600 cm3
(D) 2,400 cm3
(E) 3,200 cm3
5. An ideal gas is confined to a container whose volume is fixed. If the container holds n moles of gas, by what factor will the pressure increase if the absolute temperature is increased by a factor of 2 ?
(A) ![]()
(B) 2
(C) 2nR
(D) ![]()
(E) ![]()
6. Two large glass containers of equal volume each hold 1 mole of gas. Container 1 is filled with hydrogen gas (2 g/mol), and Container 2 holds helium (4 g/mol). If the pressure of the gas in Container 1 equals the pressure of the gas in Container 2, which of the following is true?
(A) The temperature of the gas in Container 1 is lower than the temperature of the gas in Container 2.
(B) The temperature of the gas in Container 1 is greater than the temperature of the gas in Container 2.
(C) The value of R for the gas in Container 1 is ![]() the value of R for the gas in Container 2.
the value of R for the gas in Container 2.
(D) The rms speed of the gas molecules in Container 1 is lower than the rms speed of the gas molecules in Container 2.
(E) The rms speed of the gas molecules in Container 1 is greater than the rms speed of the gas molecules in Container 2.
7. Through a series of thermodynamic processes, the internal energy of a sample of confined gas is increased by 560 J. If the net amount of work done on the sample by its surroundings is 320 J, how much heat was transferred between the gas and its environment?
(A) 240 J absorbed
(B) 240 J dissipated
(C) 880 J absorbed
(D) 880 J dissipated
(E) None of the above
8. What”s the total work performed on the gas as it”s transformed from state a to state c, along the path indicated?
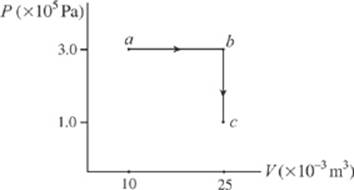
(A) 1,500 J
(B) 3,000 J
(C) 4,500 J
(D) 5,000 J
(E) 9,500 J
9. During each cycle, a heat engine absorbs 400 J of heat from its high-temperature source and discards 300 J of heat into its low-temperature sink. What is the efficiency of this engine?
(A) ![]()
(B) ![]()
(C) ![]()
(D) ![]()
(E) ![]()
10. Of the following, which is the best description of the second law of thermodynamics?
(A) The total energy of the universe is a constant.
(B) The efficiency of a heat engine can never be greater than 50 percent.
(C) The amount of heat required to vaporize a liquid is greater than the amount of heat required to melt a solid of the same substance.
(D) The entropy of the universe is always increasing.
(E) As the altitude increases, the boiling point of water decreases.
Keywords
heat
thermal energy
temperature
degrees Fahrenheit
degrees Celsius
absolute temperature scale
kelvins (K)
triple point of water
solid
liquid
vapor
gas
melts
liquefies
freezes
solidifies
evaporate
condense
sublimate
deposition
specific heat
thermal equilibrium
latent heat of transformation
heat of fusion
heat of vaporization
coefficient of linear expansion
coefficient of volume expansion
pressure
pascal (Pa)
mole
Avogadro”s constant
universal gas constant
ideal gas law
molar mass
thermodynamics
zeroth law of thermodynamics
first law of thermodynamics
state
P–V diagram
entropy
second law of thermodynamics
heat engine
adiabatic
isothermal
efficiency
Carnot cycle
internal energy
Summary
· Objects have energy because of their temperatures, and heat is this energy in transit from one object to another. Temperature is a measure of the concentration of an object”s internal thermal energy.
· When a substance absorbs or gives off heat, either the temperature of the substance can change, or the substance can undergo a phase change, but not both.
· The change of a substance”s temperature depends upon the specific heat of the substance and the amount of the substance present. Use the equation Q = mc ∆T.
· The temperature of a substance remains constant during the substance”s phase transition. Use the formula Q = mL.
· Heat transfer and thermal expansion are related in that when a substance undergoes a temperature change, it changes in size.
· Because the atoms or molecules that make up a gas move freely and rapidly in a chaotic swarm, a confined gas exerts a force in the walls of its container. To find this pressure, use the equation P =![]() .
.
· The ideal gas law, PV = nRT, covers ideal gases, which have the following properties:
1. The volume of the gas molecules is negligible compared to that of the container which holds them.
2. They experience no electrical forces.
3. They undergo elastic collisions.
· The zeroth law of thermodynamics states that when two objects are brought into contact, heat flows from the warmer object to the cooler one.
· The first law of thermodynamics states that energy (in the form of heat) is neither created nor destroyed in any thermodynamic system.
· The second law of thermodynamics states that the total amount of disorder—the total entropy—of a system plus its surroundings will never decrease.
· A heat engine is a device that uses heat to produce useful work. To determine the efficiency of a heat engine, use the formula 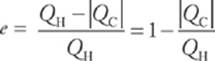 . Unless QC = 0, the engine”s efficiency is always less than 1.
. Unless QC = 0, the engine”s efficiency is always less than 1.
· For a Carnot engine, 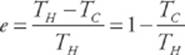
· The change of internal energy is zero for a closed path on a P–V diagram.