Biology of Humans
11. Blood
In the previous several chapters we discussed the nervous and endocrine systems, which control and coordinate body activities. In this chapter, we begin considering systems that maintain the human body. Here, we begin with blood—a critical life-sustaining fluid. We also encounter many of the reasons that blood is so vital as we consider its functions and composition. In addition, we discuss blood type and why it is so important. Finally, we consider how blood forms clots to prevent blood loss from a wound.
Functions of Blood
Blood is sometimes referred to as the river of life. The comparison is apt because, like many a river, blood serves as a transportation system. It carries vital materials to all the cells of the body and carries away the wastes that cells produce. But blood does more than passively move its cargo. Its white blood cells help protect us against disease-causing organisms, and its clotting mechanisms help protect us from excessive blood loss when a vessel is damaged. In addition, buffers in the blood help regulate the acid-base balance of body fluids. Blood also helps regulate body temperature by absorbing heat produced in metabolically active regions and distributing it to cooler regions and to the skin, where the heat is dissipated. So the diverse functions of blood can be grouped into three categories: transportation, protection, and regulation.
· Blood donations save lives.
Composition of Blood
Blood is thicker than water. The reason for the difference is that blood contains cells suspended in its watery fluid. In fact, a single drop of blood contains more than 250 million blood cells. You may recall from Chapter 4 that blood is classified as a connective tissue because it contains cellular elements suspended in a matrix. The liquid matrix is called plasma, and the cellular elements are collectively called the formed elements (Figure 11.1).
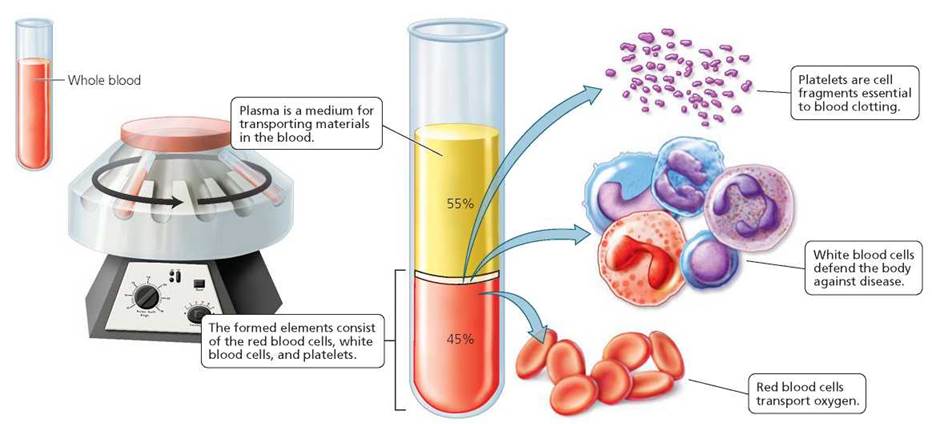
FIGURE 11.1. Blood can be separated into its major components when it is placed in a test tube with a substance that prevents coagulation and then spun in a centrifuge. Whole blood consists of a straw-colored liquid, called plasma, in which cellular elements, called formed elements, are suspended. After separation, the uppermost layer in the test tube consists of plasma. The formed elements are found in two layers below the plasma. Just below the plasma is a thin layer consisting of platelets and white blood cells (leukocytes). The red blood cells (erythrocytes) are packed at the bottom of the test tube.
Plasma
Plasma is a straw-colored liquid that makes up about 55% of blood. Plasma serves as the medium in which materials are transported by the blood. Almost every substance that is transported by the blood is dissolved in the plasma. These include nutrients (such as simple sugars, amino acids, lipids, and vitamins), ions (such as sodium, potassium, and chloride), dissolved gases (including carbon dioxide, nitrogen, and a small amount of oxygen), and every hormone. In addition to transporting materials to the cells, the plasma carries away cellular wastes. For example, urea from protein breakdown and uric acid from nucleic acid breakdown are carried to the kidneys, where they can be removed from the body. Blood also transports carbon dioxide from the cells where it is produced to the lungs for release. Indeed, if wastes were not removed the cells would die.
Despite the amount and variety of substances transported by the blood, most of the dissolved substances (solutes) in the blood are plasma proteins, which make up 7% to 8% of plasma. Plasma proteins help balance water flow between the blood and the cells. You may recall from Chapter 3 that water moves by osmosis across biological membranes from an area of lesser solute concentration to an area of greater solute concentration. Without the plasma proteins, water would be drawn out of the blood by the proteins in cells. As a result, fluid would accumulate in the tissues, causing swelling.
Most of the 50 or so types of plasma proteins fall into one of three general categories: albumins, globulins, and clotting proteins. The albumins make up more than half of the plasma proteins. They are most important in the blood's water-balancing ability. The globulins have a variety of functions. Some globulins transport lipids, including fats and some cholesterol, as well as fat-soluble vitamins. Other globulins are antibodies, which provide protection against many diseases. An example of the third category of plasma protein— the clotting proteins—is fibrinogen, whose role we discuss later in the chapter.
Formed Elements
Among the substances transported by the plasma are the formed elements—platelets, white blood cells, and red blood cells. These cell fragments and cells perform some of the key functions of the blood. Descriptions and functions of the formed elements of the blood are summarized in Table 11.1.
TABLE 11.1. The Formed Elements of Blood
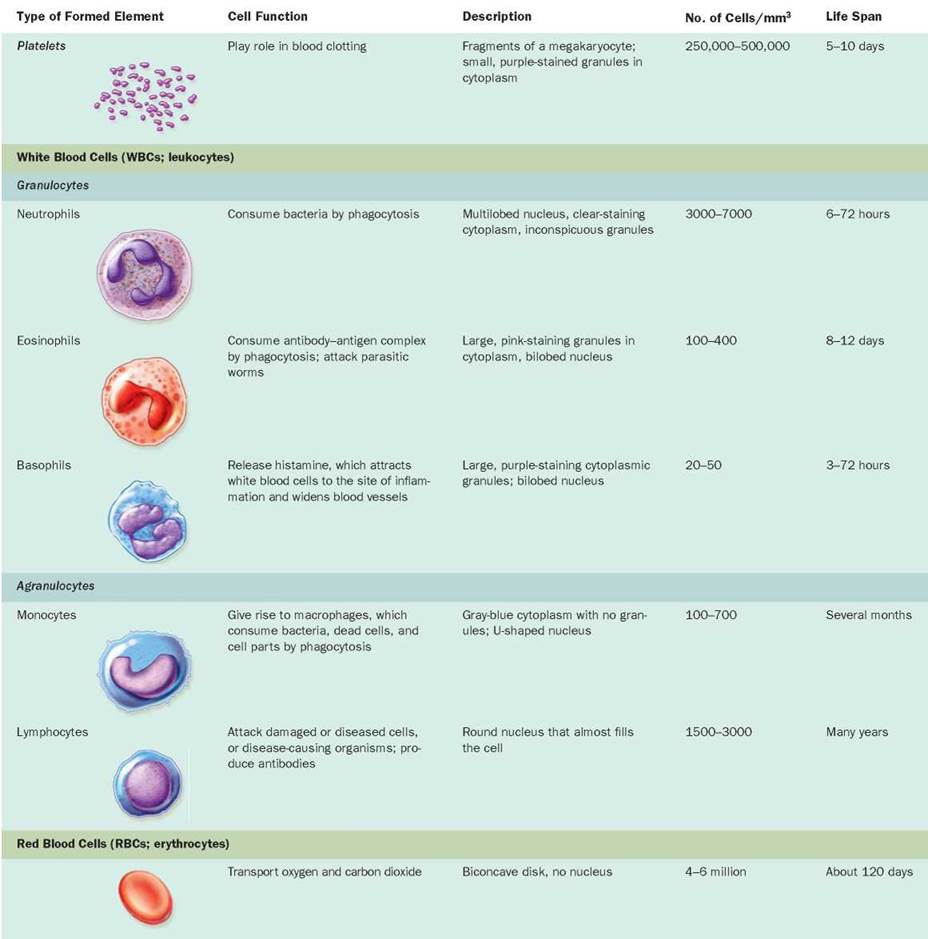
Red bone marrow, a porous connective tissue that fills the cavities within many bones, is the birthplace and nursery for the formed elements. Its spongelike framework supports fat cells, but it also supports the undifferentiated cells called blood stem cells that divide and give rise to all the formed elements (Figure 11.2).
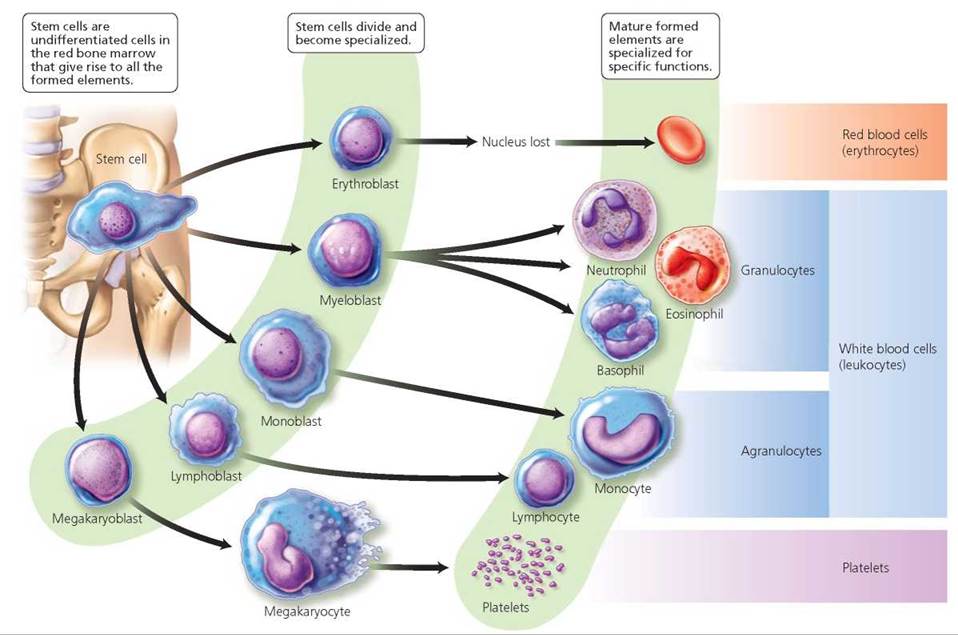
FIGURE 11.2. All formed elements originate In the red bone marrow from an undifferentiated cell called a stem cell. Stem cells divide and differentiate, giving rise to the various types of blood cells.
Platelets
Platelets, sometimes called thrombocytes (thromb, clot; cyte, cell), are essential to blood clotting. They are actually fragments of larger cells called megakaryocytes and are formed in the red bone marrow when megakaryocytes break apart. The fragments are released into the blood at the astounding rate of about 200 billion a day. They then mature during the course of a week, after which they circulate in the blood for about 5 to 10 days. Platelets contain several substances important in stopping the loss of blood through damaged blood vessels. This vital function of platelets is considered later in the chapter.
Stop and think
Thrombocytopenia is a medical term for having relatively few platelets in the blood. Why would this condition cause unusual bruising and bleeding?
White Blood Cells and Defense against Disease
White blood cells (WBCs), or leukocytes (leuk-, white; -cyte, cell), perform certain mundane housekeeping duties—such as removing wastes, toxins, and damaged or abnormal cells—but they also serve as warriors in the body's fight against disease (see Chapters 13). Although leukocytes represent less than 1% of whole blood, we simply could not live without them. We would succumb to the microbes that surround us. Because the number of white blood cells increases when the body responds to microbes, white blood cell counts are often used as an index of infection.
Like the other formed elements, white blood cells are produced in the red bone marrow.1 Unlike platelets and red blood cells, however, white blood cells are nucleated. Moreover, although they circulate in the bloodstream, they are not confined there. By squeezing between neighboring cells that form the walls of blood vessels, white blood cells can leave the circulatory system and move to a site of infection, tissue damage, or inflammation (Figure 11.3). Having slipped out of the capillary into the fluid bathing the cells, white blood cells roam through the tissue spaces. Chemicals released by invading microbes or damaged cells attract the white blood cells and cause them to gather in areas of tissue damage or infection. Certain types of white blood cells may then engulf the "offender" in a process called phagocytosis (phago- to eat; -cyt- cell; see Chapter 3). Our consideration of white blood cells in this chapter is brief; Chapter 13 contains a more detailed discussion of their many tactics for defending our bodies.
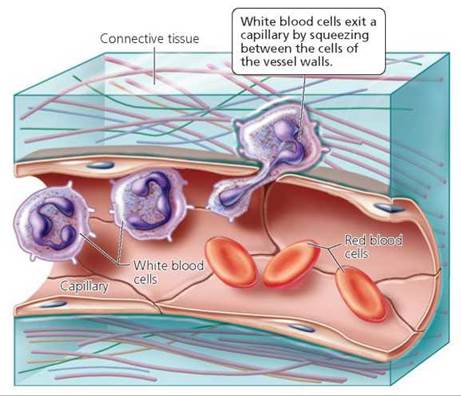
FIGURE 11.3. White blood cells can squeeze between the cells that form the wall of a capillary. They then enter the fluid surrounding body cells and, attracted by chemicals released by microbes or damaged cells, gather at the site of infection or injury.
There are two groups of white blood cells. Granulocytes have granules in their cytoplasm. The granules are actually sacs containing chemicals that are used as weapons to destroy invading pathogens, especially bacteria. Agranulocytes lack cytoplasmic granules or have very small granules. These two groups comprise a total of five types of white blood cells.
Granulocytes All white blood cells are colorless, and thus they are often stained so that they are visible under a microscope. Depending on the color of the granules after they have been stained for microscopic study, the granulocytes are classified as neutrophils, eosinophils, or basophils. Neutrophils have small granules that don't stain with either acidic red stain eosin or with a basic blue stain. The granules in eosinophils pick up a pink color from the stain eosin. Basophils have purple-staining granules (see Table 11.1).
• Neutrophils, the most abundant of all white blood cells, are the blood cell soldiers on the front lines. Arriving at the site of infection before the other types of white blood cells, neutrophils immediately begin to engulf microbes by phagocytosis, thus curbing the spread of the infection. After engulfing a dozen or so bacteria, a neutrophil dies. But even in death it helps the body's defense by releasing chemicals that attract more neutrophils to the scene. Dead neutrophils, along with bacteria and cellular debris, make up pus, the yellowish liquid we usually associate with infection.
• Eosinophils contain substances that are important in the body's defense against parasitic worms, such as tapeworms and hookworms. They also lessen the severity of allergies by engulfing antibody-antigen complexes and inactivating inflammatory chemicals.
• Basophils release histamine, a chemical that attracts other white blood cells to the site of infection and causes blood vessels to dilate (widen), thereby increasing blood flow to the affected area. They also play a role in some allergic reactions.
Agranulocytes. The agranulocytes, which lack visible granules in the cytoplasm, are classified as monocytes or lymphocytes.
• Monocytes, the largest of all formed elements, leave the bloodstream and enter various tissues, where they develop into macrophages. Macrophages are phagocytic cells that engulf invading microbes, dead cells, and cellular debris.
• Lymphocytes are classified into two types: B lymphocytes and T lymphocytes. The B lymphocytes give rise to plasma cells, which, in turn, produce antibodies. Antibodies are proteins that recognize specific molecules—called antigens—on the surface of invading microbes or other foreign cells. After recognizing the foreign cell by its antigens, the antibodies help prevent it from harming the body. There are several types of T lymphocytes, specialized white blood cells that play roles in the body's defense mechanisms. We will discuss lymphocytes further in Chapter 13.
Red Blood Cells and Transport of Oxygen
Red blood cells (RBCs), also called erythrocytes (erythro-, red; -cyt-, cell), pick up oxygen in the lungs and ferry it to all the cells of the body. Red blood cells also carry about 23% of the blood's total carbon dioxide, a metabolic waste product. They are by far the most numerous cells in the blood. Indeed, they number 4-6 million/mm3 of blood and constitute approximately 45% of the total blood volume.
Red blood cells and hemoglobin. The shape of red blood cells, as shown in Figure 11.4, is marvelously suited to their function of picking up and transporting oxygen. Red blood cells are quite small, and each is shaped like a biconcave disk—a flattened disk indented on each side. This shape maximizes the surface area of the cell. Because of the greater surface area, oxygen can enter the red blood cell more rapidly than if the disk did not have an indent. A red blood cell is unusually flexible and thus able to squeeze through capillaries (the smallest blood vessels, those in which gas exchange occurs), even those with a diameter much smaller than red blood cells. Each red blood cell is packed with hemoglobin, the oxygen-binding pigment that is responsible for the cells' red color. As a red blood cell matures in the red bone marrow, it loses its nucleus and most organelles. Thus, it is scarcely more than a sac of hemoglobin molecules. Each red blood cell is packed with approximately 280 million molecules of hemoglobin. As can be seen in Figure 11.5, each hemoglobin molecule is made up of four subunits. Each subunit consists of a polypeptide chain and a heme group. The heme group includes an iron ion that actually binds to the oxygen. Therefore, each hemoglobin molecule can carry up to four molecules of oxygen. The compound formed when hemoglobin binds with oxygen is called, logically enough, oxyhemoglobin. Body cells use the oxygen to boost the amount of energy extracted from food molecules in cellular respiration. As oxygen is used, carbon dioxide is produced. Most of the carbon dioxide travels to the lungs dissolved in plasma, but some of it binds to hemoglobin (at a site other than that where the iron atom binds oxygen).
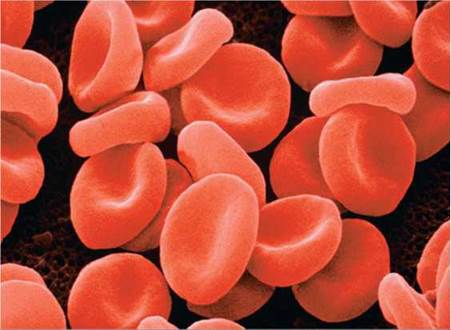
FIGURE 11.4. Red blood cells serve to ferry oxygen from the lungs to the needy tissues. Each red blood cell is packed with the oxygen-binding pigment hemoglobin. A red blood cell is a small disk that is indented on both sides. This design maximizes the surface area for gas exchange.
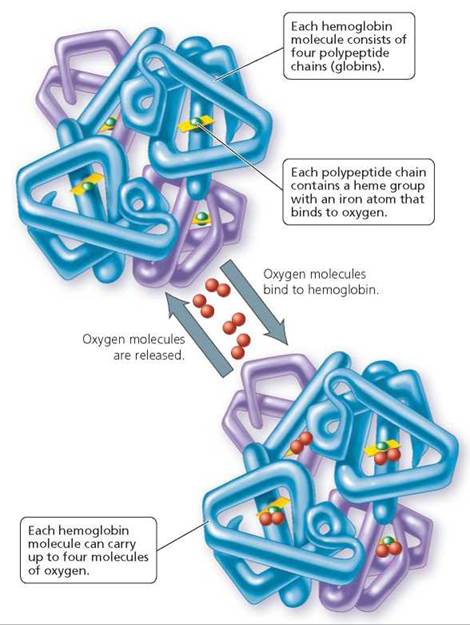
FIGURE 11.5. The structure of hemoglobin, the pigment in red blood cells that transports oxygen from the lungs to the cells
As wonderfully adapted as it is for carrying oxygen, the hemoglobin molecule binds 200 times more readily to carbon monoxide, a product of the incomplete combustion of any carbon-containing fuel. In other words, if concentrations of carbon monoxide and oxygen were identical in inhaled air, for every 1 molecule of hemoglobin that binds to oxygen molecules, 200 molecules of hemoglobin would bind to carbon monoxide molecules. This is the reason carbon monoxide can be deadly. When carbon monoxide binds to the oxygen-binding sites on hemoglobin, it blocks oxygen from binding to it, preventing the blood from carrying life-giving oxygen to the cells. Carbon monoxide is a particularly insidious poison because it is odorless and tasteless. Its primary source is automobile exhaust, but it can also come from indoor sources, including improperly vented heaters and leaky chimneys. Thus, indoor carbon monoxide detectors can save lives.
Life cycle of red blood cells. The creation of a red blood cell, which takes about 6 days, entails many changes in the cell's activities and structure. First, the very immature cell becomes a factory for hemoglobin molecules. And as noted earlier, after the cell is packed with hemoglobin, its nucleus is pushed out. Then a structural metamorphosis occurs, culminating in a mature red blood cell with a typical biconcave shape. Once this change in shape takes place, the cell leaves the bone marrow and enters the bloodstream. Red marrow produces roughly 2 million red blood cells a second, throughout the life of an individual, for a cumulative total of more than half a ton in a lifetime.
A red blood cell lives for only about 120 days. During that time, it travels through approximately 100 km (62 mi) of blood vessels—being bent, bumped, and squeezed along the way. Its life span is probably limited by the lack of a nucleus that would otherwise maintain it and direct needed repairs. Without a nucleus, for instance, protein synthesis needed to replace key enzymes cannot take place, so the cell becomes increasingly rigid and fragile.
The liver and spleen are the "graveyards" where worn-out red blood cells are removed from circulation. The old, inflexible red blood cells tend to become stuck in the tiny circulatory channels of these organs. Macrophages then engulf and destroy the dying cells. The demolished cells release their hemoglobin, which the liver degrades into its protein (globin) component and heme. The protein is digested to amino acids, which can be used to make other proteins. The iron from the heme is salvaged and sent to the red marrow for recycling.
The remaining part of the heme is degraded to a yellow pigment, called bilirubin, which is excreted by the liver in bile. Bile is released into the small intestine, where it assists in the digestion of fats. It is then carried along the digestive system to the large intestine with undigested food and becomes a component of feces. The color of feces is partly due to bilirubin that has been broken down by intestinal bacteria.
Products formed by the chemical breakdown of heme also create the yellowish tinge in a bruise that is healing. A bruise, or black-and-blue mark, results when tiny blood vessels or capillaries are ruptured and blood leaks into the surrounding tissue. As the tissues use up the oxygen, the blood becomes darker and, viewed through the overlying tissue, looks black or blue. Gradually, the red blood cells degenerate, releasing hemoglobin. The breakdown products of hemoglobin then make the bruise appear yellowish.
Stop and think
Hepatitis is an inflammation of the liver that can be caused by certain viruses or exposure to certain drugs. It impairs the liver's ability to handle bilirubin properly. A symptom of hepatitis is jaundice, a condition in which the skin develops a yellow tone. Why does hepatitis cause jaundice?
A negative feedback mechanism regulates red blood cell production according to the needs of the body, especially the need for oxygen (Figure 11.6). Most of the time, red blood cell production matches red blood cell destruction. However, there are circumstances—blood loss, for instance—that trigger a homeostatic mechanism that speeds up the rate of red blood cell production. This mechanism is initiated by a decrease in the oxygen supply to the body's cells. Certain cells in the kidney sense the reduced oxygen, and they respond by producing the hormone erythropoietin. Erythropoietin then travels to the red marrow, where it steps up both the division rate of stem cells and the maturation rate of immature red blood cells. When maximally stimulated by erythropoietin, the red marrow can increase red blood cell production tenfold—to 20 million cells per second! The resulting increase in red blood cell numbers is soon adequate to meet the oxygen needs of body cells. The increased oxygen-carrying capacity of the blood then inhibits erythropoietin production.
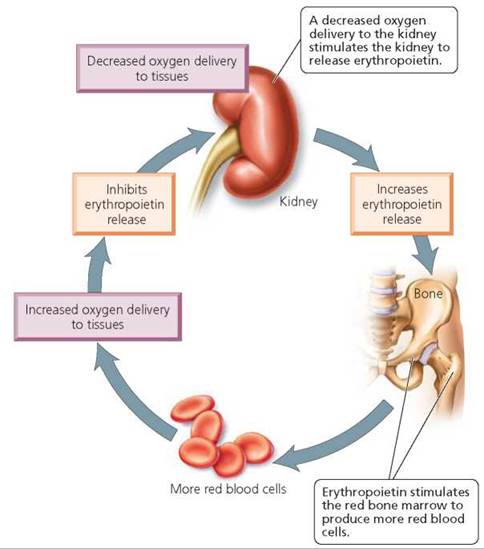
FIGURE 11.6. The production of red blood cells is regulated by a negative feedback relationship between the oxygen-carrying capacity of the blood and the production of erythropoietin.
Blood Cell Disorders
Disorders of red and white blood cells have many different causes. The problems associated with each disorder depend on the type of blood cells affected, because red and white blood cells have different functions.
Disorders of Red Blood Cells
Anemia, a condition in which the blood's ability to carry oxygen is reduced, can result from too little hemoglobin, too few red blood cells, or both. The symptoms of anemia include fatigue, headaches, dizziness, paleness, and breathlessness. In addition, an anemic person's heart often beats faster to compensate for the blood's decreased ability to carry oxygen. The accelerated pumping can cause heart palpitations—the uncomfortable awareness of one's own heartbeat. Although anemia is not usually life threatening, it can affect the quality of life because lack of energy and low levels of productive activity often go hand in hand.
Worldwide, the most common cause of anemia is an insufficiency of iron in the body, which leads to inadequate hemoglobin production. Iron-deficiency anemia can be caused by a diet that contains too little iron, by an inability to absorb iron from the digestive system, or from blood loss—such as might occur because of menstrual flow or peptic ulcers. Treatment for iron- deficiency anemia includes dealing with the cause of the iron depletion, as well as restoring iron levels to normal by eating foods that are rich in iron—such as meat, leafy green vegetables, and fortified cereals—or by taking pills that contain iron.
Blood loss will obviously lower red blood cell counts, but so will any condition that causes the destruction of red blood cells at an amount that exceeds the production of red blood cells. For example, in hemolytic anemias, red blood cells are ruptured because of infections, defects in the membranes of red blood cells, transfusion of mismatched blood, or hemoglobin abnormalities. Sickle-cell anemia is an example of a hemolytic anemia caused by abnormal hemoglobins. This abnormal hemoglobin (hemoglobin S) causes the red blood cells to become deformed to a crescent (or sickle) shape when the blood's oxygen content is low. The misshapen cells are fragile and rupture easily, clogging small blood vessels and promoting clot formation. These events prevent oxygen-laden blood from reaching the tissues and can cause episodes of extreme pain.
Red blood cell numbers also drop when the production of red blood cells is halted or impaired, as occurs in pernicious anemia. The production of red blood cells depends on a supply of vitamin B12. The small intestine absorbs vitamin B12 from the diet with the aid of a chemical called intrinsic factor, which is produced by the stomach lining. People with pernicious anemia do not produce intrinsic factor and are therefore unable to absorb vitamin B12. They are treated with injections of B12. Lead poisoning can also cause anemia. (See the Environmental Issue essay, Lead Poisoning.)
Environmental Issue
Lead Poisoning
What do children in old buildings in America today have in common with the ancient upper-class Romans? Lead poisoning. Today, some children are poisoned by the lead in paint chips that they unintentionally ingest as aging paint peels (or, in very young children, when they chew or bite objects that have been treated with lead paint). Centuries ago, Romans were poisoned by lead in their eating and drinking vessels.
The types of cells that are most sensitive to lead are nerve cells and the bone marrow cells that give rise to red blood cells. The best-known effects of lead on human health are probably those on the nervous system—mental retardation, lowered IQs, reading disabilities, irritability, hyperactivity, and even death. However, the effects of lead on the blood, including anemia and changes in blood enzymes, can also be devastating. Indeed, a blood test is the only way to positively diagnose lead poisoning.
Lead launches a two-pronged attack on the blood's ability to carry oxygen. First, it interferes with the absorption of iron from the digestive system. Second, it inhibits one of the essential enzymes leading to hemoglobin synthesis.
As our example of ancient Romans suggests, lead has been a common pollutant for centuries, encountered in a surprising number of substances. The major causes of lead poisoning in the United States are as follows:
1. House paint. Although lead has not been added to house paint for years, this remains the most prevalent cause of poisoning.
2. Drinking water. One out of every five Americans drinks tap water containing excess levels of lead, according to Environmental Protection Agency (EPA) estimates.
3. Air and soil pollution. The primary source of lead in the atmosphere has been leaded gasoline. Leaded gasoline is no longer sold in the United States for use in automobiles, although it is permissible in farm equipment, race cars, and other off-road vehicles.
4. Lead solder on food cans. The lead in the solder used to seal the seams in food and beverage cans can leach into the contents, especially if the contents are acidic foods such as tomatoes or citrus juices. Although the use of lead solder on food cans has been banned in the United States, it is still found on some imported food cans.
5. Lead-containing dishware. China, ceramic, and earthenware dishes may be coated with lead-containing glaze. When the glazes are not properly fired, lead can leach into the food or beverage contained in the dish.
6. Fruits and vegetables grown in lead-contaminated soil. Soil can be contaminated by lead from the atmosphere or by leakage from lead-containing solid wastes, such as lead batteries in automobiles that are dumped into landfills. Important sources of atmospheric lead are the combustion of lead motor fuel, the smelting of ores and other industrial processes, and the incineration of refuse.
Although it is true that blood levels of lead are at an all-time low in the United States, the lead problem has not been eliminated. In 2010 the EPA passed a new Renovation Repair and Painting rule to protect children from lead dust caused by lead paint removal. An important problem now is deciding who should be financially responsible for keeping us safe.

Children often put objects into their mouths. They may unintentionally ingest lead paint chips if the object has been treated with lead.
Questions to Consider
• Who do you think should bear the financial responsibility of removing lead paint from old apartment buildings? The residents? The landlords? The city? The federal government?
• Do you think the residents of buildings with lead paint should be compensated for any lead-related health problems they experience? If so, who do you think should pay—the landlords? The original contractors? The government?
what would you do?
Blood doping is a practice that boosts red blood cell numbers and, therefore, the blood's ability to carry oxygen. In one method, blood is drawn from the body and stored, prompting the body temporarily to boost its production of red blood cells. The stored blood is then returned to the body. Some athletes who compete in aerobic events, such as swimming or cycling, have used blood doping to increase their endurance and speed. Although blood doping is considered unethical and is prohibited in Olympic competition, it is difficult to detect. One sign is an exceptionally high number of red blood cells; another is the presence in the urine of a hormone (erythropoietin) that stimulates red blood cell production—but tests can detect only about 50% of the athletes who practice blood doping. Considering the large percentage of blood-doped athletes who will escape detection even if they are tested, do you think that those who are caught should be punished?
Disorders of White Blood Cells
Infectious mononucleosis ("mono") is a viral disease of the lymphocytes. It is caused by the Epstein-Barr virus, which is common in humans but usually asymptomatic. In mononucleosis, the infection causes an increase in lymphocytes that have an atypical appearance. Because mono often is spread from person to person by oral contact, it is sometimes called the kissing disease; however, it can also be spread by sharing eating utensils or drinking glasses. Mono is most common among teenagers and young adults, particularly those living in dormitories. It often strikes at stressful times, such as during final exams, when immune resistance is low.
The initial symptoms of mono are similar to those of influenza: fever, chills, headache, sore throat, and an overwhelming sense of being ill. Within a few days, the lymph node (commonly called "glands") in the neck, armpits, and groin become painfully swollen. There is no treatment, so mono must simply run its course. The major symptoms generally subside within a few weeks, but fatigue may linger for several months or longer.
Leukemia is a cancer of the white blood cells that causes their uncontrolled multiplication, so that their number increases greatly. These cancerous cells—all descendants of a single abnormal cell—remain immature and are therefore unable to defend the body against infectious organisms. Because they divide more rapidly than do normal cells, the abnormal cells "take over" the bone marrow, preventing the development of all normal blood cells, including red blood cells, white blood cells, and platelets.
Symptoms of leukemia generally result either from the insufficient number of normal blood components or from the invasion of organs by abnormal white blood cells. The increased number of white blood cells crowds out the other formed elements. Insufficient numbers of platelets lead to inadequate clotting, which causes gum bleeding and frequent bruising. Reduced levels of red blood cells lead to anemia, which in turn causes chronic fatigue, breathlessness, and pallor. Because their white blood cells do not function properly, leukemia patients may suffer from repeated respiratory or throat infections, herpes, or skin infections. Also, bone tenderness may be experienced because the immature white blood cells pack the red marrow. Headaches, another symptom of leukemia, may be caused by anemia or by the effects of abnormal white blood cells in the brain.
Treatment of leukemia usually includes radiation therapy and chemotherapy to kill the rapidly dividing cells. In addition, transfusions of red blood cells and platelets may be given to alleviate anemia and prevent excessive bleeding. Today, many patients with acute leukemia are cured with bone marrow transplants. Someone, most often a family member whose tissue type closely matches that of the patient, must be located and agree to serve as a donor. Then the bone marrow in the leukemia patient must be destroyed by irradiation and drugs. Next, the donor's bone marrow is given to the patient intravenously, just like a blood transfusion. The marrow cells find their way to the patient's marrow and begin to grow. Treatment with stem cells from umbilical cord blood seems to be as effective as bone marrow transplantation in helping some patients with leukemia go into remission (the signs and symptoms of leukemia go away). Stem cells from umbilical cord blood are discussed in Chapter 19a, "Special Topic: Stem Cells—A Repair Kit for the Body."
Blood Types
Human blood is classified into different blood types, depending on the presence or absence of certain molecules— mostly proteins—on the surface of a person's red blood cells. Each of your body cells is labeled as "self" (that is, as belonging to your body) by proteins on its surface. If a cell that lacks these self markers enters the body, the body's defense system recognizes that the foreign cell is "nonself" and does not belong. The foreign cell will have different proteins on its surface. To the body's defense system, these proteins are antigens, identifying the cell as foreign and marking it for destruction. As was mentioned earlier, one way the body attacks the foreign cell is by producing proteins called antibodies that specifically target the antigen on the foreign cell's surface. Let's consider the role of antigens and antibodies in blood types and transfusions.
ABO Blood Types
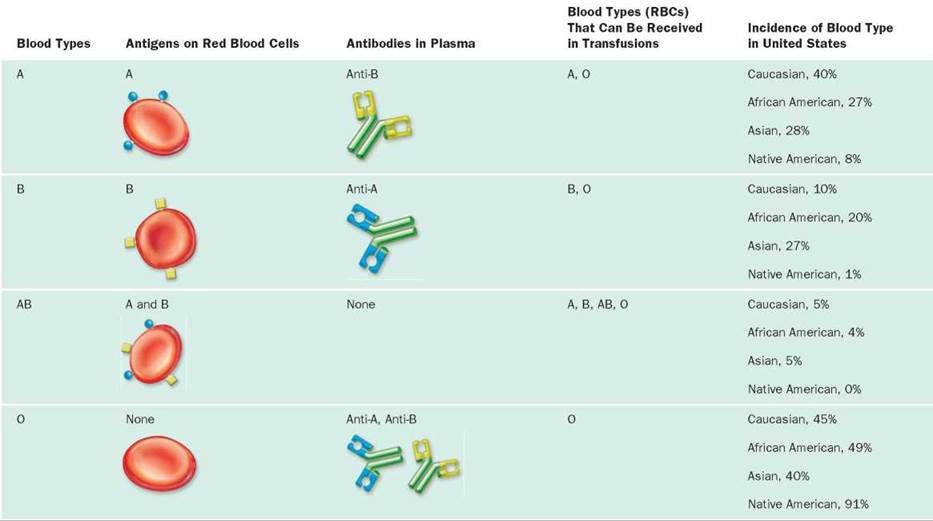
When asked about your blood type, you are probably used to responding by indicating one of the types in the ABO series: A, B, AB, or O. Red blood cells with only the antigen A on their surface are type A. When only the B antigen is on the red blood cell surface, the blood is type B. Blood with both A and B antigens on the red blood cell surface is designated type AB. When neither A nor B antigens are present, the blood is type O.
Normally, a person's plasma contains antibodies against those antigens that are not on his or her own red blood cells. Thus, individuals with type A blood have antibodies against the B antigen (anti-B antibodies2), and those with type B blood have antibodies against A (anti-A antibodies). Because individuals with type AB blood have both antigens on their red blood cells, they have neither antibody. Those with type O blood have neither antigen, so they have both anti-A and anti-B antibodies in their plasma.
In a typical test for blood type, technicians mix a drop of a person's blood with a solution containing anti-A antibodies and mix another drop of the blood with a solution containing anti-B antibodies. If clumping occurs in one of the mixtures, it means the antigen corresponding to the antibody in that mixture is present (Figure 11.7).
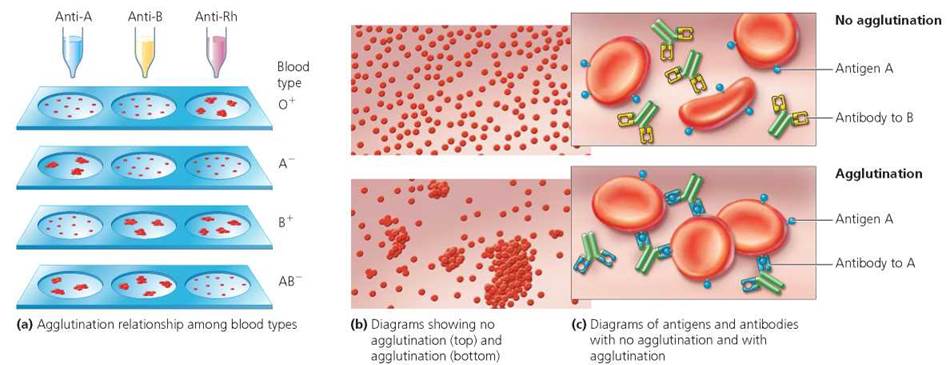
FIGURE 11.7. Blood is typed by mixing it with serum known to contain antibodies specific for a certain antigen. If blood containing that antigen is mixed with the serum, the blood will agglutinate (clump). Thus, one drop of blood is mixed with serum containing anti-A and another drop with serum containing anti-B. A third drop of blood is mixed with serum containing antibodies to Rh. Agglutination in response to an antibody reveals the presence of the antigen.
What blood type will agglutinate to each serum when mixed separately with sera containing anti-A, anti-B, and anti-Rh antibodies?
AB+
Similarly, when a person is given a blood transfusion with donor blood containing foreign antigens, the antibodies in the recipient's blood will cause the donor's cells to clump, or agglutinate. This clumping of the donor's cells is damaging and perhaps even fatal. The clumped cells can get stuck in small blood vessels and block blood flow to body cells. Or they may break open, releasing their cargo of hemoglobin. The hemoglobin then clogs the filtering system in the kidneys, causing death.
It is important to be sure that the blood types of a donor and recipient are compatible, which means that the recipient's blood does not contain antibodies to antigens on the red blood cells of the donor. The plasma of the donor's blood may contain antibodies against antigens on the recipient's red blood cells, but these will be diluted as they enter the recipient's circulation. Therefore, the donor's antibodies are not a major problem. The questions to ask in this case are (1) what, if any, antigens are on the donor's cells and (2) what, if any, antibodies are in the recipient's blood. For example, if a person with blood type A is given a transfusion of blood type B or of type AB, the naturally occurring anti-B antibodies in the recipient's blood will cause the donor's red blood cells to clump because they have the B antigen. The transfusion relationships among blood types in the ABO series are shown in Table 11.2.
TABLE 11.2. Transfusion Relationships among Blood Types
Rh factor
The A and B antigens are not the only important antigens found on the surface of red blood cells. The presence or absence of an Rh factor is also an important component of blood type. The name Rh comes from the beginning of the name of the rhesus monkey, in which an Rh antigen was first discovered. People who have the Rh antigens on their red blood cells are considered Rh-positive (Rh+). When Rh antigens are missing from the red blood cell surface, the individual is considered Rh-negative (Rh-).
An Rh-negative person will not form anti-Rh antibodies unless he or she has been exposed to the Rh antigen. For this reason, an Rh-negative individual should be given only Rh-negative blood in a transfusion. If he or she is mistakenly given Rh-positive blood, it will stimulate the production of anti-Rh antibodies. A transfusion reaction will not occur after the first such transfusion, because it takes time for the body to start making anti-Rh antibodies. After a second transfusion of Rh-positive blood, however, the antibodies in the recipient's plasma will react with the antigens on the red blood cells of the donated blood. This reaction may lead to the death of the patient.
The Rh factor can also have medical importance for pregnancies in which the mother is Rh-negative and the fetus is Rh-positive, a situation that may occur if the father is Rh-positive (see Chapter 20; Figure 11.8). Ordinarily, the maternal and fetal blood supplies do not mix during pregnancy. However, as a result of blood vessel damage, some mixing may occur during a miscarriage or delivery. If the baby's red blood cells, which bear Rh antigens, accidentally pass into the mother's bloodstream, she will produce anti-Rh antibodies. There are usually no ill effects associated with the first introduction of the Rh antigen. However, if antibodies are present in the maternal blood from a previous pregnancy with an Rh-positive child or from a transfusion of Rh-positive blood, the anti-Rh antibodies may pass into the blood of the fetus during a subsequent pregnancy. This transfer can occur because anti-Rh antibodies, unlike red blood cells, can cross the placenta (a structure that forms during pregnancy to allow the exchange of selected substances between the maternal and fetal circulatory systems). These anti-Rh antibodies may destroy the fetus's red blood cells. As a result, the child may be stillborn or very anemic at birth. This condition is called hemolytic disease of the newborn.
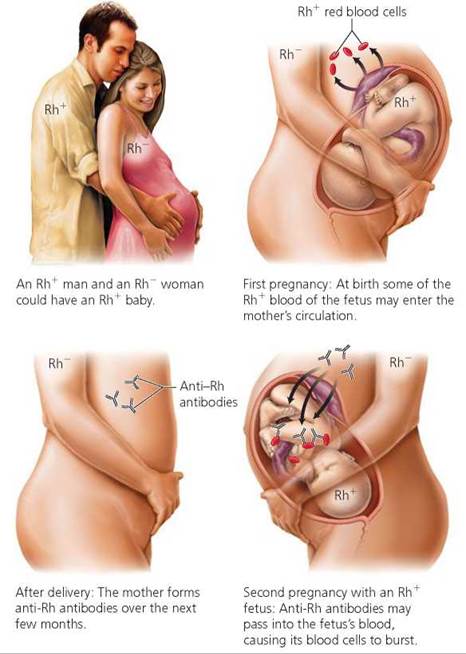
FIGURE 11.8. Rh incompatibility can result when an Rh-negative (Rh-) woman is pregnant with an Rh-positive (Rh+) baby if the woman has been previously exposed to Rh+ blood.
The incidence of hemolytic disease of the newborn has decreased in recent years due to the development of a means of destroying any Rh-positive fetal cells in an Rh-negative mother's blood supply before they can stimulate the mother's cells to produce her own anti-Rh antibodies. The Rh-positive cells are killed by injecting RhoGAM, a serum containing antibodies against the Rh antigens, at about the seventh month of pregnancy and shortly after delivery if the baby is Rh-positive. Rh antigens are thus prevented from being "set" in the memory of the mother's immune system. The injected antibodies disappear after a few months. Therefore, no antibodies linger to affect the fetus in a subsequent pregnancy.
Blood Donation
We have seen that blood is indeed a life-saving fluid, and someone in the United States or Canada needs donated blood every 2 seconds. If you are a healthy person at least 17 years old (16 years in some states), weigh at least 110 lbs, and are willing to spend 10 to 20 minutes, you may be eligible to give the gift of life. When you donate you may feel a slight twinge as the needle is inserted, but the procedure is generally painless. Each donation is a pint of blood. The donated plasma is replaced within hours, and the cells are replaced within weeks. The donated blood can be separated into its components: red blood cells to increase the oxygencarrying capacity of the recipient's blood, platelets to help the recipient's blood clot, and plasma to help the recipient control bleeding.
Blood Clotting
When a blood vessel is cut, several responses are triggered to stop the bleeding. To understand the process of clotting, imagine how you might respond if the garden hose you are using springs a leak. Your initial response might be to squeeze the hose in hopes of stopping the water flow. Likewise, the body's immediate response to blood vessel injury is for the vessel to constrict (squeeze shut).
The next response is to plug the hole (Figure 11.9). Your thumb might do the job on your garden hose; in an injured blood vessel, platelets form a plug that seals the leak. The platelet plug is formed when platelets cling to cables of collagen, a protein fiber on the torn blood vessel surface. When the platelets attach to collagen, they swell, form many cellular extensions, and stick together. Platelets also produce a chemical that attracts other platelets to the wound and makes them stick together even more. Aspirin prevents the formation of this chemical and, therefore, inhibits clot formation. For this reason, a daily dose of aspirin is sometimes prescribed to prevent the formation of blood clots that could block blood vessels nourishing heart tissue and thus cause the death of heart cells (a heart attack). It is also why aspirin can cause excessive bleeding.
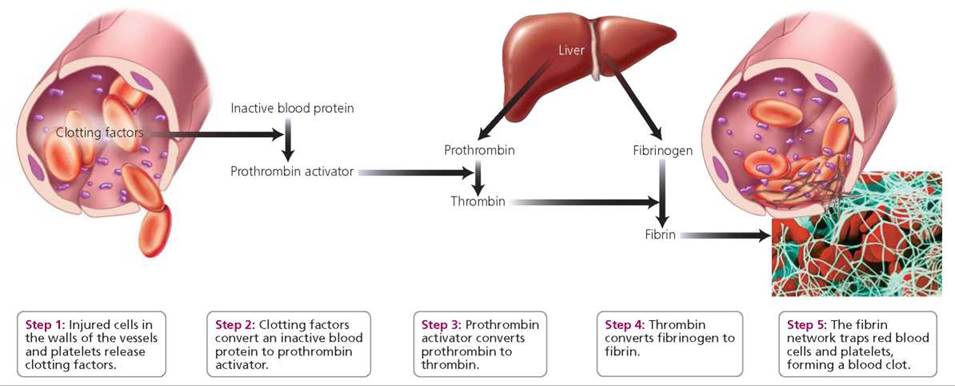
FIGURE 11.9. Selected steps in the blood-clotting process
The next stage in stopping blood loss through a damaged blood vessel is the formation of the clot itself. There are more than 30 steps in the process of clot formation, but here we will describe only the key events. Clot formation begins when clotting factors are released from injured tissue and from platelets. At the site of the wound, the clotting factors convert an inactive blood protein to prothrombin activator,3 which then converts prothrombin—a plasma protein produced by the liver—to an active form, thrombin. Thrombin then causes a remarkable change in another plasma protein produced by the liver, fibrinogen. The altered fibrinogen forms long strands of fibrin, a protein that makes a web that traps blood cells and forms a clot. The clot is a barrier that prevents additional blood loss through the wound in the vessel.
Stop and think
Thromboplastin, a chemical important in initiating clot formation, is released from both damaged tissue and activated platelets. How does this fact explain why a scrape, which causes a great deal of tissue damage, generally stops bleeding more quickly than a clean cut, such as a paper cut or one that might occur with a razor blade?
If even one of the many factors needed for clotting is lacking, the process can be slowed or completely blocked. Vitamin K is needed for the liver to synthesize prothrombin and three other clotting factors. Thus, without vitamin K, clotting does not occur. We have two sources of vitamin K. One is our diet. Vitamin K is found in leafy green vegetables, tomatoes, and vegetable oils. The second source is bacteria living in our intestines and producing vitamin K, some of which we absorb for our own use. Vitamin K is used rapidly by body tissues, so both sources are needed for proper blood clotting. Antibiotic treatment for serious bacterial infections can kill gastrointestinal bacteria and lead to a vitamin K deficiency in as few as 2 days.
Hemophilia is an inherited condition in which the affected person bleeds excessively owing to a fault in one of the genes involved in producing clotting factors. As noted earlier, there are many steps in the clotting process, and so there are many clotting factors. Although a loss of any of the clotting factors could cause hemophilia, the two most common types of hemophilia are caused by insufficient amounts of clotting factors VIII or IX. Because of the way hemophilia is inherited, the condition usually occurs in males (see Chapter 20). Symptoms appear when the affected child first becomes physically active. Crawling, for instance, causes bruises on the elbows and knees, and cuts tend to bleed longer than is normal. Excessive internal bleeding can damage nerves or, when it occurs in joints, permanently cripple the hemophiliac.
Treatment for hemophilia involves restoring the missing clotting factor. Bleeding episodes can be controlled with repeated transfusions of fresh plasma or with injections of concentrated clotting factor. Concentrated clotting factor is made by combining the plasma donations of many people. In fact, each injection requires 2000 to 5000 individual donations. Fortunately, two of the clotting factors (factors VIII and IX) are now being manufactured using recombinant DNA technology.
The failure of blood to clot can shorten the life of a person with hemophilia. But in people without hemophilia, the formation of unnecessary blood clots can have much more immediate health consequences because clots can disrupt blood flow. A blood clot lodged in an unbroken blood vessel is called a thrombus. A blood clot that drifts through the circulatory system is called an embolus. Emboli can drift through the circulatory system until they become stuck in a narrow vessel. When the tiny vessels that nourish the heart or brain become clogged with a clot, the consequences can be severe—a heart attack or stroke, which can be disabling or even fatal. After a wound has healed, clots are normally dissolved by an enzyme called plasmin, which is formed from an inactive protein, plasminogen. Plasmin dissolves clots by digesting the fibrin strands that form the framework of the clot.
Stop and think
Heparin is a drug that inactivates thrombin. It is sometimes administered to patients for the purpose of inhibiting the clotting response. How would heparin act to achieve these ends?
Looking ahead
In this chapter, we looked at the composition and functions of blood. In the next chapter, we will consider the blood vessels through which blood travels and the heart that pumps the blood.
_____________________________________________________
1 One type, the lymphocytes, also may be produced in the lymph nodes and other lymphoid tissues.
2 It is not certain why these antibodies form without exposure to red blood cells bearing the foreign antigen. It may be that either the bacteria that invade our bodies or the foods we eat contain a small amount of A and B antigens—enough to stimulate antibody production.
3 Prothrombin activator is an enzyme called prothrombinase.
Highlighting the Concepts
Functions of Blood (p. 199)
• Blood transports vital material to cells and carries wastes away from cells. White blood cells defend against disease. Blood clotting prevents excessive blood loss. Blood also helps regulate body temperature.
Composition of Blood (pp. 199-205)
• Blood is a type of connective tissue that contains formed elements in a liquid (plasma) matrix.
• Plasma is the liquid portion of the blood, consisting of water and certain dissolved components, mostly proteins. (The substances transported by the blood are also dissolved in the plasma. These include nutrients, ions, gases, hormones, and waste products.) Plasma proteins aid in the balance of water flow between blood and cells. These proteins are grouped into three categories: albumins, globulins, or clotting proteins (such as fibrinogen).
• Blood stem cells are undifferentiated cells that divide and give rise to all of the formed elements—namely, platelets, white blood cells, and red blood cells.
• Platelets play an important role in blood clotting. They are fragments of a larger cell called a megakaryocyte.
• White blood cells (leukocytes) help the body fight off disease and help remove wastes, toxins, and damaged cells.
• There are five types of leukocytes: neutrophils, eosinophils, basophils, monocytes, and lymphocytes. Neutrophils are the most abundant and immediately phagocytize foreign microbes. Eosinophils help the body defend against parasitic worms and play a role in allergic reactions. Basophils can increase the flow of blood by releasing histamines. Monocytes are the largest leukocytes and, after maturing into macrophages, actively fight chronic infections. Lymphocytes take part in body defense responses.
• Red blood cells (erythrocytes) are flexible cells packed with hemoglobin, an oxygen-binding pigment. Red blood cell production is controlled by erythropoietin, a hormone produced by the kidney in response to low oxygen. Worn and dead red blood cells are removed from circulation and broken down in the liver and spleen.
Blood Cell Disorders (pp. 205-207)
• Anemia is a reduction in the blood's ability to carry oxygen. There are several forms, including iron-deficiency anemia; hemolytic anemias, such as sickle-cell anemia; and pernicious anemia, caused by the stomach's failure to produce intrinsic factor, which is needed to absorb the vitamin necessary for red blood cell production.
• Infectious mononucleosis is a viral disease of lymphocytes. Leukemia is a cancer of the white blood cells in which there are high numbers of white blood cells, but they do not function properly to defend against infectious agents.
Blood Types (pp. 207-209)
• Blood types are determined by the presence of certain proteins (antigens) on the surface of red blood cells: the ABO and Rh groups. The plasma contains antibodies against A and B antigens if the antigens are not present on the red blood cells. Antibodies to Rh antigens are formed only after exposure to Rh-positive blood. If blood containing foreign antigens is introduced during a transfusion, a reaction between the antibodies and antigens can cause red blood cells to clump and clog the recipient's bloodstream, which can lead to death.
• Rh incompatibility becomes important to an Rh-negative woman who has been previously exposed to Rh-positive blood and who is pregnant with an Rh-positive fetus. Antibodies to the Rh antigens can cross the placenta and destroy the red blood cells of the fetus; this condition is called hemolytic disease of the newborn.
Blood Clotting (pp. 209-211)
• The prevention of blood loss involves three mechanisms: blood vessel constriction, platelet plug formation, and clotting. Clotting is initiated when platelets and damaged tissue release clotting factors that lead to the production of a chemical called prothrombin activator, which converts the blood protein prothrombin to thrombin. Thrombin then converts the blood protein fibrinogen to fibrin. Strands of fibrin make a mesh that traps red blood cells and forms the clot. Hemophiliacs are missing an important clotting factor and, therefore, bleed excessively.
Reviewing the Concepts
1. What is plasma? What are its functions? p. 200
2. What are the three categories of plasma proteins? p. 200
3. List the three types of formed elements, and describe the function of each. pp. 200-204
4. Compare the size, structure, and numbers of leukocytes with the size, structure, and numbers of erythrocytes. p. 203
5. White blood cells function in defending the body against foreign invaders. List the five types of white blood cells, and describe the role each plays in body defense. p. 203
6. Describe the characteristics of red blood cells that make them specialized for delivering oxygen to the tissues. pp. 203-204
7. Describe the function of hemoglobin. p. 204
8. Where are red blood cells produced? How is the production of red blood cells controlled? pp. 204-205
9. Describe what happens to worn or damaged red blood cells and their hemoglobin. p. 204
10. Why would pernicious anemia be treated with regular injections of vitamin B12 instead of by dietary supplements of this vitamin? p. 205
11. What determines a person's blood type? pp. 207-208
12. What happens if a person is given a blood transfusion with blood of an incompatible type? p. 207
13. What blood type(s) can a person with type B blood receive? Explain. pp. 207-208
14. What is hemolytic disease of the newborn? What causes it? p. 209
15. After a blood vessel is cut, what mechanisms prevent excessive blood loss? pp. 209-210
16. Describe the steps involved in blood clotting. pp. 209-210
17. What is the difference between the blood clotting that occurs after an injury and the clumping that occurs after a mismatched transfusion? pp. 207, 209-210
18. Type B blood contains which antibodies?
a. A
b. B
c. both A and B
d. neither A nor B
19. An important function of white blood cells is
a. blood clotting.
b. transportation of oxygen.
c. fighting infection.
d. maintaining blood pressure.
20. A condition in which the oxygen-carrying capacity of the blood is low is
a. infection.
b. anemia.
c. leukemia.
d. a kidney problem.
21. The primary function of red blood cells is to _____.
22. _____ is a protein in red blood cells that transports oxygen.
23. The protein that forms a net that traps red blood cells and platelets and forms blood clots is _____.
Applying the Concepts
1. Erin has leukemia, and her white blood cell count is elevated. Why does Erin have an elevated risk of infection?
2. Jose is a runner for his college track team from New York City. The championship meet is in Denver, which is called the Mile-High City because of its elevation above sea level. There is less oxygen available for breathing as elevation increases. Because this meet is so important, Jose plans to arrive in Denver several weeks before the meet. Why? Would you expect Jose's red blood cell count to be higher or lower than normal when he returns home to New York City, which is at sea level?
3. Indira is a 25-year-old woman who has uterine polyps that cause heavy vaginal bleeding. She complains that she tires easily with physical activity and always feels fatigued. The doctor orders that a blood test be done to determine what percentage of her blood is red blood cells. Why? Would you expect the percentage to be higher or lower than normal? The doctor suggests that she take iron supplements. Why?
4. Raul is in a car accident and is taken to the emergency room. He has type AB blood. Which blood types can he receive?
5. Elizabeth has type Rh-negative blood. She is pregnant for the second time. What information would the doctor want to know about the first and second children to know whether to expect a problem with this pregnancy?
6. Sarala was given an antibiotic that caused her platelet count to fall. What symptoms would be expected?
Becoming Information Literate
The umbilical cord is the lifeline for a growing fetus that connects the fetus to the mother through the placenta. Within the umbilical blood (and blood from the placenta) are blood stem cells that retain the ability to develop into red blood cells, white blood cells, and platelets. There is a growing interest in collecting umbilical cord blood when a baby is born and storing it for future use.
Use at least three reliable sources (books, journals, websites) to answer the following questions. Cite your sources, and explain why you chose them.
1. What are potential uses for stem cells from umbilical cord blood?
2. What are advantages of using umbilical cord blood over a bone marrow transplant as a source of stem cells?
3. Based on what you have learned about umbilical cord blood, do you think that there should be umbilical cord blood banks? Why? If so, do you think use of the cord blood should be restricted to the family whose infant donated the blood, or should it be made available to anyone?