CHEMISTRY THE CENTRAL SCIENCE
15 CHEMICAL EQUILIBRIUM
EXERCISES
VISUALIZING CONCEPTS
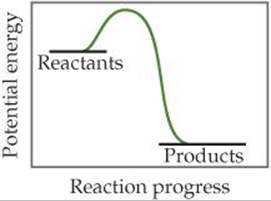
15.1 (a) Based on the following energy profile, predict whether kf > kr or kf < kr. (b) Using Equation 15.5, predict whether the equilibrium constant for the process is greater than 1 or less than 1. [Section 15.1]
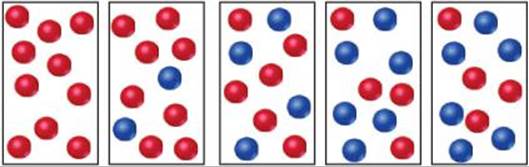
15.2 The following diagrams represent a hypothetical reaction A ![]() B, with A represented by red spheres and B represented by blue spheres. The sequence from left to right represents the system as time passes. Do the diagrams indicate that the system reaches an equilibrium state? Explain. [Section 15.1 and 15.2
B, with A represented by red spheres and B represented by blue spheres. The sequence from left to right represents the system as time passes. Do the diagrams indicate that the system reaches an equilibrium state? Explain. [Section 15.1 and 15.2
15.3 The following diagram represents an equilibrium mixture produced for a reaction of the type A + X ![]() AX. If the volume is 1 L, is K greater or smaller than 1? [Section 15.2]
AX. If the volume is 1 L, is K greater or smaller than 1? [Section 15.2]

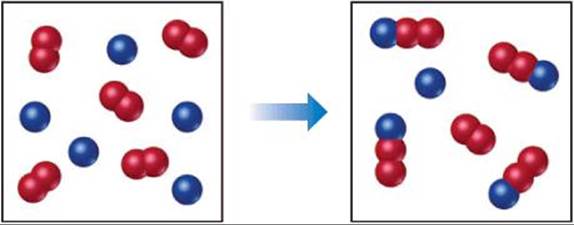
15.4 The following diagram represents a reaction shown going to completion. (a) Letting A = red spheres and B = blue spheres, write a balanced equation for the reaction. (b) Write the equilibrium-constant expression for the reaction. (c) Assuming that all of the molecules are in the gas phase, calculate Δn, the change in the number of gas molecules that accompanies the reaction. (d) How can you calculate Kp if you know Kc at a particular temperature? [Section 15.2]
15.5 A friend says that the faster the reaction, the larger the equilibrium constant. Is your friend correct? Why or why not? [Sections 15.1 and 15.2]
15.6 A certain chemical reaction has Kc = 1.5 × 106. Does this mean that at equilibrium there are 1.5 × 106 times as many product molecules as reactant molecules? Explain. [Sections 15.1 and 15.2]
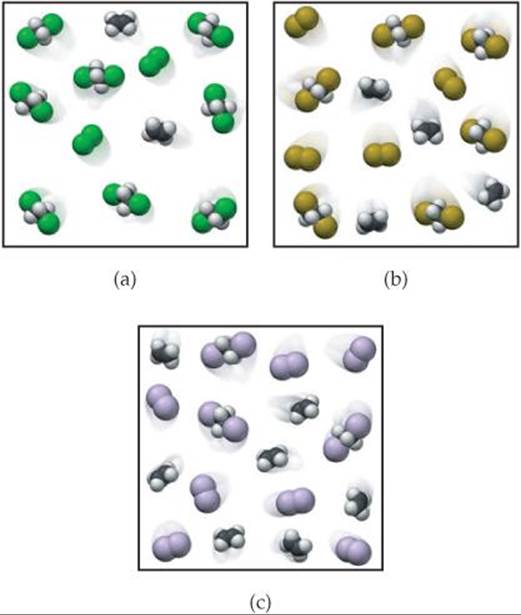
15.7 Ethene (C2H4) reacts with halogens (X2) by the following reaction:
![]()
The following figures represent the concentrations at equilibrium at the same temperature when X2 is Cl2 (green), Br2 (brown), and I2 (purple). List the equilibria from smallest to largest equilibrium constant. [Section 15.3]
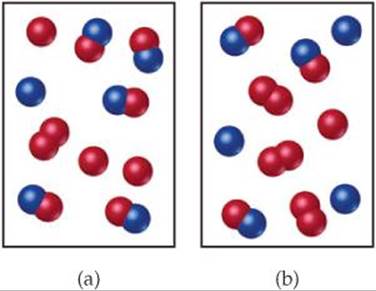
15.8 The reaction A2 + B2![]() 2 AB has an equilibrium constant Kc = 1.5. The following diagrams represent reaction mixtures containing A2 molecules (red), B2 molecules (blue), and AB molecules. (a) Which reaction mixture is at equilibrium? (b) For those mixtures that are not at equilibrium, how will the reaction proceed to reach equilibrium? [Sections 15.5 and 15.6]
2 AB has an equilibrium constant Kc = 1.5. The following diagrams represent reaction mixtures containing A2 molecules (red), B2 molecules (blue), and AB molecules. (a) Which reaction mixture is at equilibrium? (b) For those mixtures that are not at equilibrium, how will the reaction proceed to reach equilibrium? [Sections 15.5 and 15.6]
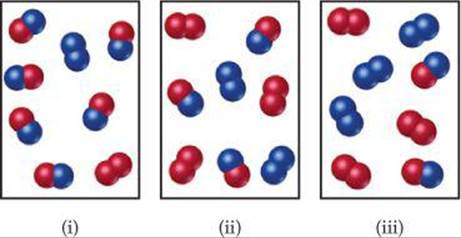
15.9 The reaction A2(g) + B(g) ![]() A(g) + AB(g) has an equilibrium constant of Kp = 2. The accompanying diagram shows a mixture containing A atoms (red), A2 molecules, and AB molecules (red and blue). How many B atoms should be added to the diagram to illustrate an equilibrium mixture? [Section 15.6]
A(g) + AB(g) has an equilibrium constant of Kp = 2. The accompanying diagram shows a mixture containing A atoms (red), A2 molecules, and AB molecules (red and blue). How many B atoms should be added to the diagram to illustrate an equilibrium mixture? [Section 15.6]
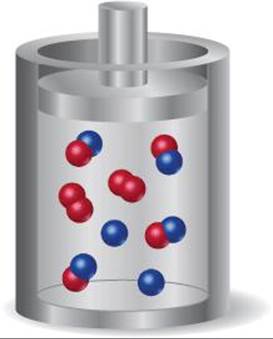
15.10 The diagram shown here represents the equilibrium state for the reaction A2(g) + 2 B(g) ![]() 2 AB(g). (a) Assuming the volume is 2 L, calculate the equilibrium constant Kc for the reaction. (b) If the volume of the equilibrium mixture is decreased, will the number of AB molecules increase or decrease? [Sections 15.5 and 15.7]
2 AB(g). (a) Assuming the volume is 2 L, calculate the equilibrium constant Kc for the reaction. (b) If the volume of the equilibrium mixture is decreased, will the number of AB molecules increase or decrease? [Sections 15.5 and 15.7]
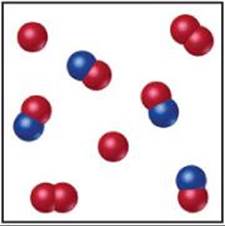
15.11 The following diagrams represent equilibrium mixtures for the reaction A2 + B ![]() A + AB at (a) 300 K and (b) 500 K. The A atoms are red, and the B atoms are blue. Is the reaction exothermic or endothermic? [Section 15.7]
A + AB at (a) 300 K and (b) 500 K. The A atoms are red, and the B atoms are blue. Is the reaction exothermic or endothermic? [Section 15.7]
15.12 The following graph represents the yield of the compound AB at equilibrium in the reaction A(g) + B(g) ![]() AB(g) at two different pressures, x andy, as a function of temperature. (a) Is this reaction exothermic or endothermic? (b) Is P = x greater or smaller than P = y? [Section 15.7]
AB(g) at two different pressures, x andy, as a function of temperature. (a) Is this reaction exothermic or endothermic? (b) Is P = x greater or smaller than P = y? [Section 15.7]
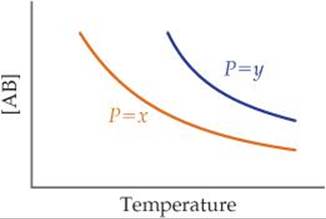
(a) Is this reaction exothermic or endothermic? (b) Is P = x greater or smaller than P = y? [Section 15.7]
EQUILIBRIUM; THE EQUILIBRIUM CONSTANT (sections 15.1, 15.2, 15.3, 15.4)
15.13 Suppose that the gas-phase reactions A ![]() B and B
B and B ![]() A are both elementary processes with rate constants of 4.7 × 10–3 s–1 and 5.8 × 10–3 s–1, respectively. (a) What is the value of the equilibrium constant for the equilibrium A(g)
A are both elementary processes with rate constants of 4.7 × 10–3 s–1 and 5.8 × 10–3 s–1, respectively. (a) What is the value of the equilibrium constant for the equilibrium A(g) ![]() B(g)? (b) Which is greater at equilibrium, the partial pressure of A or the partial pressure of B? Explain.
B(g)? (b) Which is greater at equilibrium, the partial pressure of A or the partial pressure of B? Explain.
15.14 Consider the reaction A + B ![]() C + D. Assume that both the forward reaction and the reverse reaction are elementary processes and that the value of the equilibrium constant is very large. (a) Which species predominate at equilibrium, reactants or products? (b) Which reaction has the larger rate constant, the forward or the reverse? Explain.
C + D. Assume that both the forward reaction and the reverse reaction are elementary processes and that the value of the equilibrium constant is very large. (a) Which species predominate at equilibrium, reactants or products? (b) Which reaction has the larger rate constant, the forward or the reverse? Explain.
______
15.15 Write the expression for Kc for the following reactions. In each case indicate whether the reaction is homogeneous or heterogeneous.
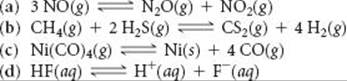
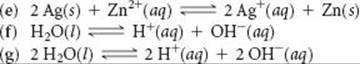
15.16 Write the expressions for Kc for the following reactions. In each case indicate whether the reaction is homogeneous or heterogeneous.
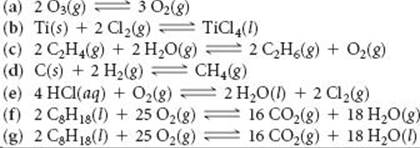
______
15.17 When the following reactions come to equilibrium, does the equilibrium mixture contain mostly reactants or mostly products?
![]()
15.18 Which of the following reactions lies to the right, favoring the formation of products, and which lies to the left, favoring formation of reactants?
![]()
______
15.19 Can the equilibrium constant ever be a negative number? Explain.
______
15.20 Can the equilibrium constant ever be zero? Explain.
15.21 If Kc = 0.042 for PCl3(g) + Cl2(g) ![]() PCl5(g) at 500 K, what is the value of Kp for this reaction at this temperature?
PCl5(g) at 500 K, what is the value of Kp for this reaction at this temperature?
15.22 Calculate Kc at 303 K for SO2(g) + Cl2(g) ![]() SO2Cl2(g) if Kp = 34.5 at this temperature.
SO2Cl2(g) if Kp = 34.5 at this temperature.
______
15.23 The equilibrium constant for the reaction
![]()
is Kc = 1.3 × 10–2 at 1000 K. (a) At this temperature does the equilibrium favor NO and Br2, or does it favor NOBr? (b) Calculate Kc for 2 NOBr(g) ![]() 2 NO(g) + Br2(g). (c) Calculate Kc for NOBr(g) → NO(g) +
2 NO(g) + Br2(g). (c) Calculate Kc for NOBr(g) → NO(g) + ![]() Br2(g).
Br2(g).
15.24 Consider the following equilibrium:
![]()
(a) Calculate Kp. (b) Does the equilibrium mixture contain mostly H2 and S2 or mostly H2S? (c) Calculate the values of Kc and Kp if you rewrote the balanced chemical equation with 1 mol of H2(g) instead of 2 mol.
______
15.25 At 1000 K, Kp = 1.85 for the reaction
![]()
(a) What is the value of Kp for the reaction ![]() (b) What is the value of Kp for the reaction 2 SO2(g) + O2(g)
(b) What is the value of Kp for the reaction 2 SO2(g) + O2(g) ![]() 2 SO3(g)? (c) What is the value of Kc for the reaction in part (b)?
2 SO3(g)? (c) What is the value of Kc for the reaction in part (b)?
15.26 Consider the following equilibrium, for which Xp = 0.0752 at 480 °C:
![]()
(a) What is the value of Kp for the reaction 4 HCl(g) + O2(g) ![]() 2 Cl2(g) + 2 H2O(g)(b) What is the value of Kp for the reaction Cl2(g) + H2O(g)
2 Cl2(g) + 2 H2O(g)(b) What is the value of Kp for the reaction Cl2(g) + H2O(g) ![]() 2 HCl(g) +
2 HCl(g) + ![]() O2(g)? (c) What is the value of Kc for the reaction in part (b)?
O2(g)? (c) What is the value of Kc for the reaction in part (b)?
______
15.27 The following equilibria were attained at 823 K:
![]()
Based on these equilibria, calculate the equilibrium constant for H2(g) + CO2(g) ![]() CO(g) + H2O(g) at 823 K.
CO(g) + H2O(g) at 823 K.
15.28 Consider the equilibrium
![]()
Calculate the equilibrium constant Kp for this reaction, given the following information (at 298 K):
![]()
______
15.29 Explain why we normally exclude pure solids and liquids from equilibrium-constant expressions.
15.30 Explain why we normally exclude solvents from liquid-phase reactions in equilibrium-constant expressions.
______
15.31 Mercury(I) oxide decomposes into elemental mercury and elemental oxygen: 2 Hg2O(s) ![]() 4 Hg(ℓ) + O2(g). (a) Write the equilibrium-constant expression for this reaction in terms of partial pressures. (b) Suppose you run this reaction in a solvent that dissolves elemental mercury and elemental oxygen. Rewrite the equilibrium-constant expression in terms of molarities for the reaction, using (solv) to indicate solvation.
4 Hg(ℓ) + O2(g). (a) Write the equilibrium-constant expression for this reaction in terms of partial pressures. (b) Suppose you run this reaction in a solvent that dissolves elemental mercury and elemental oxygen. Rewrite the equilibrium-constant expression in terms of molarities for the reaction, using (solv) to indicate solvation.
15.32 Consider the equilibrium Na2O(s) + SO2(g) ![]() Na2SO3(s). (a) Write the equilibrium-constant expression for this reaction in terms of partial pressures. (b) All the compounds in this reaction are soluble in water. Rewrite the equilibrium-constant expression in terms of molarities for the aqueous reaction.
Na2SO3(s). (a) Write the equilibrium-constant expression for this reaction in terms of partial pressures. (b) All the compounds in this reaction are soluble in water. Rewrite the equilibrium-constant expression in terms of molarities for the aqueous reaction.
CALCULATING EQUILIBRIUM CONSTANTS (section 15.5)
15.33 Methanol (CH3OH) is produced commercially by the catalyzed reaction of carbon monoxide and hydrogen: CO(g) + 2 H2(g)![]() CH3OH(g). An equilibrium mixture in a 2.00-L vessel is found to contain 0.0406 mol CH3OH, 0.170 mol CO, and 0.302 mol H2 at 500 K. CalculateKc at this temperature.
CH3OH(g). An equilibrium mixture in a 2.00-L vessel is found to contain 0.0406 mol CH3OH, 0.170 mol CO, and 0.302 mol H2 at 500 K. CalculateKc at this temperature.
15.34 Gaseous hydrogen iodide is placed in a closed container at 425 °C, where it partially decomposes to hydrogen and iodine: 2 HI(g) ![]() H2(g) + I2(g). At equilibrium it is found that [HI] = 3.53 × 10–3M, [H2] = 4.79 × 10–4M, and [I2] = 4.79 × 10–4M. What is the value of Kc at this temperature?
H2(g) + I2(g). At equilibrium it is found that [HI] = 3.53 × 10–3M, [H2] = 4.79 × 10–4M, and [I2] = 4.79 × 10–4M. What is the value of Kc at this temperature?
______
15.35 The equilibrium 2 NO(g) + Cl2(g) ![]() 2 NOCl(g) is established at 500 K. An equilibrium mixture of the three gases has partial pressures of 0.095 atm, 0.171 atm, and 0.28 atm for NO, Cl2, and NOCl, respectively. (a) Calculate Kp for this reaction at 500.0 K. (b) If the vessel has a volume of 5.00 L, calculate Kc at this temperature.
2 NOCl(g) is established at 500 K. An equilibrium mixture of the three gases has partial pressures of 0.095 atm, 0.171 atm, and 0.28 atm for NO, Cl2, and NOCl, respectively. (a) Calculate Kp for this reaction at 500.0 K. (b) If the vessel has a volume of 5.00 L, calculate Kc at this temperature.
15.36 Phosphorus trichloride gas and chlorine gas react to form phosphorus pentachloride gas: PCl3(g) + Cl2(g) ![]() PCl5(g). A 7.5-L gas vessel is charged with a mixture of PCl3(g) and Cl2(g), which is allowed to equilibrate at 450 K. At equilibrium the partial pressures of the three gases are PPCl3 = 0.124 atm, PCl2 = 0.157 atm, and PPCl5 = 1.30 atm. (a) What is the value of Kp at this temperature? (b) Does the equilibrium favor reactants or products? (c) Calculate Kc for this reaction at 450 K.
PCl5(g). A 7.5-L gas vessel is charged with a mixture of PCl3(g) and Cl2(g), which is allowed to equilibrate at 450 K. At equilibrium the partial pressures of the three gases are PPCl3 = 0.124 atm, PCl2 = 0.157 atm, and PPCl5 = 1.30 atm. (a) What is the value of Kp at this temperature? (b) Does the equilibrium favor reactants or products? (c) Calculate Kc for this reaction at 450 K.
______
15.37 A mixture of 0.10 mol of NO, 0.050 mol of H2, and 0.10 mol of H2O is placed in a 1.0-L vessel at 300 K. The following equilibrium is established:
![]()
At equilibrium [NO] = 0.062 M. (a) Calculate the equilibrium concentrations of H2, N2, and H2O. (b) Calculate Kc.
15.38 A mixture of 1.374 g of H2 and 70.31 g of Br2 is heated in a 2.00-L vessel at 700 K. These substances react according to
![]()
At equilibrium the vessel is found to contain 0.566 g of H2. (a) Calculate the equilibrium concentrations of H2, Br2, and HBr. (b) Calculate Kc.
______
15.39 A mixture of 0.2000 mol of CO2, 0.1000 mol of H2, and 0.1600 mol of H2O is placed in a 2.000-L vessel. The following equilibrium is established at 500 K:
![]()
(a) Calculate the initial partial pressures of CO2, H2, and H2O. (b) At equilibrium PH2O = 3.51 atm. Calculate the equilibrium partial pressures of CO2, H2, and CO. (c) Calculate Kp for the reaction. (d) Calculate Kc for the reaction.
15.40 A flask is charged with 1.500 atm of N2O4(g) and 1.00 atm NO2(g) at 25 °C, and the following equilibrium is achieved:
![]()
After equilibrium is reached, the partial pressure of NO2 is 0.512 atm. (a) What is the equilibrium partial pressure of N2O4? (b) Calculate the value of Kp for the reaction. (c) Calculate Kc for the reaction.
______
15.41 Two different proteins X and Y are dissolved in aqueous solution at 37 °C. The proteins bind in a 1:1 ratio to form XY. A solution that is initially 1.00 mM in each protein is allowed to reach equilibrium. At equilibrium, 0.20 mM of free X and 0.20 mM of free Y remain. What isKc for the reaction?
[15.42] A chemist at a pharmaceutical company is measuring equilibrium constants for reactions in which drug candidate molecules bind to a protein involved in cancer. The drug molecules bind the protein in a 1:1 ratio to form a drug-protein complex. The protein concentration in aqueous solution at 25 °C is 1.50 × 10–6M. Drug A is introduced into the protein solution at an initial concentration of 2.00 × 10–6M. Drug B is introduced into a separate, identical protein solution at an initial concentration of 2.00 × 10–6M. At equilibrium, the drug A-protein solution has an A-protein complex concentration of 1.00 × 10–6M, and the drug B solution has a B-protein complex concentration of 1.40 × 10–6M. Calculate the Kc value for the A-protein binding reaction and for the B-protein binding reaction. Assuming that the drug that binds more strongly will be more effective, which drug is the better choice for further research?
APPLICATIONS OF EQUILIBRIUM CONSTANTS (section 15.6)
15.43 (a) How does a reaction quotient differ from an equilibrium constant? (b) If Qc < Kc, in which direction will a reaction proceed in order to reach equilibrium? (c) What condition must be satisfied so that Qc = K?
15.44 (a) How is a reaction quotient used to determine whether a system is at equilibrium? (b) If Qc > Kc, how must the reaction proceed to reach equilibrium? (c) At the start of a certain reaction, only reactants are present; no products have been formed. What is the value of Qc at this point in the reaction?
______
15.45 At 100 °C the equilibrium constant for the reaction COCl2(g) ![]() CO(g) + Cl2(g) has the value Kc = 2.19 × 10–10. Are the following mixtures of COCl2, CO, and Cl2 at 100 °C at equilibrium? If not, indicate the direction that the reaction must proceed to achieve equilibrium. (a)[COCl2] = 2.00 × 10–3M, [CO] = 3.3 × 10–6M, [Cl2] = 6.62 × 10–6M; (b) [COCl2] = 4.50 × 10–2M, [CO] = 1.1 × 10–7M, [Cl2] = 2.25 × 10–7M; (c) [COCl2] = 0.0100 M, [CO] = [Cl2] = 1.48 × 10–6M
CO(g) + Cl2(g) has the value Kc = 2.19 × 10–10. Are the following mixtures of COCl2, CO, and Cl2 at 100 °C at equilibrium? If not, indicate the direction that the reaction must proceed to achieve equilibrium. (a)[COCl2] = 2.00 × 10–3M, [CO] = 3.3 × 10–6M, [Cl2] = 6.62 × 10–6M; (b) [COCl2] = 4.50 × 10–2M, [CO] = 1.1 × 10–7M, [Cl2] = 2.25 × 10–7M; (c) [COCl2] = 0.0100 M, [CO] = [Cl2] = 1.48 × 10–6M
15.46 As shown in Table 15.2, Kp for the equilibrium
![]()
is 4.51 × 10–5 at 450 °C. For each of the mixtures listed here, indicate whether the mixture is at equilibrium at 450 °C. If it is not at equilibrium, indicate the direction (toward product or toward reactants) in which the mixture must shift to achieve equilibrium.

______
15.47 At 100 °C, Kc = 0.078 for the reaction
![]()
In an equilibrium mixture of the three gases, the concentrations of SO2Cl2 and SO2 are 0.108 M and 0.052 M, respectively. What is the partial pressure of Cl2 in the equilibrium mixture?
15.48 At 900 K the following reaction has Kp = 0.345:
![]()
In an equilibrium mixture the partial pressures of SO2 and O2 are 0.135 atm and 0.455 atm, respectively. What is the equilibrium partial pressure of SO3 in the mixture?
______
15.49 (a) At 1285 °C the equilibrium constant for the reaction Br2(g) ![]() 2 Br(g) is Kc = 1.04 × 10–3. A 0.200-L vessel containing an equilibrium mixture of the gases has 0.245 g Br2(g) in it. What is the mass of Br(g) in the vessel? (b) For the reaction H2(g) + I2(g)
2 Br(g) is Kc = 1.04 × 10–3. A 0.200-L vessel containing an equilibrium mixture of the gases has 0.245 g Br2(g) in it. What is the mass of Br(g) in the vessel? (b) For the reaction H2(g) + I2(g) ![]() 2 HI(g), Kc =55.3 at 700 K. In a 2.00-L flask containing an equilibrium mixture of the three gases, there are 0.056 g H2 and 4.36 g I2. What is the mass of HI in the flask?
2 HI(g), Kc =55.3 at 700 K. In a 2.00-L flask containing an equilibrium mixture of the three gases, there are 0.056 g H2 and 4.36 g I2. What is the mass of HI in the flask?
15.50 (a) At 800 K the equilibrium constant for I2(g) ![]() 2 I(g) is Kc = 3.1 × 10–5. If an equilibrium mixture in a 10.0-L vessel contains 2.67 × 10–2 g of I(g), how many grams of I2 are in the mixture? (b) For 2 SO2(g) + O2(g)
2 I(g) is Kc = 3.1 × 10–5. If an equilibrium mixture in a 10.0-L vessel contains 2.67 × 10–2 g of I(g), how many grams of I2 are in the mixture? (b) For 2 SO2(g) + O2(g) ![]() 2 SO3(g), Kp = 3.0 × 104 at 700 K. In a 2.00-L vessel the equilibrium mixture contains 1.17 g of SO3 and 0.105 g of O2. How many grams of SO2 are in the vessel?
2 SO3(g), Kp = 3.0 × 104 at 700 K. In a 2.00-L vessel the equilibrium mixture contains 1.17 g of SO3 and 0.105 g of O2. How many grams of SO2 are in the vessel?
______
15.51 At 2000 °C the equilibrium constant for the reaction
![]()
is kc = 2.4 × 103. If the initial concentration of NO is 0.175 M what are the equilibrium concentrations of NO, N2, and O2?
15.52 For the equilibrium
![]()
at 400 K, Kc = 7.0. If 0.25 mol of Br2 and 0.55 mol of Cl2 are introduced into a 3.0-L container at 400 K, what will be the equilibrium concentrations of Br2, Cl2, and BrCl?
______
15.53 At 373 K, Xp = 0.416 for the equilibrium
![]()
If the pressures of NOBr(g) and NO(g) are equal, what is the equilibrium pressure of Br2(g)?
15.54 At 218 °C, Kc = 1.2 × 10–4 for the equilibrium
![]()
Calculate the equilibrium concentrations of NH3 and H2S if a sample of solid NH4SH is placed in a closed vessel at 218 °C and decomposes until equilibrium is reached.
______
15.55 Consider the reaction
![]()
At 25 °C the equilibrium constant is Kc = 2.4 × 10–5 for this reaction. (a) If excess CaSO4(s) is mixed with water at 25 °C to produce a saturated solution of CaSO4, what are the equilibrium concentrations of Ca2+ and SO42–? (b) If the resulting solution has a volume of 1.4 L, what is the minimum mass of CaSO4(s) needed to achieve equilibrium?
15.56 At 80 °C, Kc = 1.87 × 10–3 for the reaction
![]()
(a) Calculate the equilibrium concentrations of PH3 and BCl3 if a solid sample of PH3BCl3 is placed in a closed vessel at 80 °C and decomposes until equilibrium is reached. (b) If the flask has a volume of 0.250 L, what is the minimum mass of PH3BCl3(s) that must be added to the flask to achieve equilibrium?
______
15.57 For the reaction I2 + Br2(g) ![]() 2 IBr(g), Kc = 280 at 150 °C. Suppose that 0.500 mol IBr in a 2.00-L flask is allowed to reach equilibrium at 150 °C. What are the equilibrium concentrations of IBr, I2, and Br2?
2 IBr(g), Kc = 280 at 150 °C. Suppose that 0.500 mol IBr in a 2.00-L flask is allowed to reach equilibrium at 150 °C. What are the equilibrium concentrations of IBr, I2, and Br2?
15.58 At 25 °C the reaction
![]()
has an equilibrium constant Kc = 7.1 × 10–4. What are the equilibrium concentrations of Ca2+ and CrO42– in a saturated solution of CaCrO4?
______
15.59 Methane, CH4, reacts with I2 according to the reaction CH4(g) + l2(g) ![]() CH3l(g) + HI(g). At 630 K, Kp for this reaction is 2.26 × 10–4. A reaction was set up at 630 K with initial partial pressures of methane of 105.1 torr and of 7.96 torr for I2. Calculate the pressures, in torr, of all reactants and products at equilibrium.
CH3l(g) + HI(g). At 630 K, Kp for this reaction is 2.26 × 10–4. A reaction was set up at 630 K with initial partial pressures of methane of 105.1 torr and of 7.96 torr for I2. Calculate the pressures, in torr, of all reactants and products at equilibrium.
15.60 The reaction of an organic acid with an alcohol, in organic solvent, to produce an ester and water is commonly done in the pharmaceutical industry. This reaction is catalyzed by strong acid (usually H2SO4). A simple example is the reaction of acetic acid with ethyl alcohol to produce ethyl acetate and water:
![]()
where “(solv)” indicates that all reactants and products are in solution but not an aqueous solution. The equilibrium constant for this reaction at 55 °C is 6.68. A pharmaceutical chemist makes up 15.0 L of a solution that is initially 0.275 M in acetic acid and 3.85 M in ethanol. At equilibrium, how many grams of ethyl acetate are formed?
LE CHÂTELIER'S PRINCIPLE (section 15.7)
15.61 Consider the following equilibrium for which ΔH < 0
![]()
How will each of the following changes affect an equilibrium mixture of the three gases: (a) O2(g) is added to the system; (b) the reaction mixture is heated; (c) the volume of the reaction vessel is doubled; (d) a catalyst is added to the mixture; (e) the total pressure of the system is increased by adding a noble gas; (f) SO3(g) is removed from the system?
15.62 Consider 4 NH3(g) + 5 O2(g)![]() 4 NO(g) + 6 H2O(g), ΔH = –904.4 kJ. How does each of the following changes affect the yield of NO at equilibrium? Answer increase, decrease, or no change: (a) increase [NH3]; (b) increase [H2O]; (c) decrease [O2]; (d) decrease the volume of the container in which the reaction occurs; (e) add a catalyst; (f) increase temperature.
4 NO(g) + 6 H2O(g), ΔH = –904.4 kJ. How does each of the following changes affect the yield of NO at equilibrium? Answer increase, decrease, or no change: (a) increase [NH3]; (b) increase [H2O]; (c) decrease [O2]; (d) decrease the volume of the container in which the reaction occurs; (e) add a catalyst; (f) increase temperature.
______
15.63 How do the following changes affect the value of the equilibrium constant for a gas-phase exothermic reaction: (a) removal of a reactant (b) removal of a product, (c) decrease in the volume, (d) decrease in the temperature, (e) addition of a catalyst?
15.64 For a certain gas-phase reaction, the fraction of products in an equilibrium mixture is increased by either increasing the temperature or by increasing the volume of the reaction vessel. (a) Is the reaction exothermic or endothermic? (b) Does the balanced chemical equation have more molecules on the reactant side or product side?
______
15.65 Consider the following equilibrium between oxides of nitrogen
![]()
(a) Use data in Appendix C to calculate ΔH° for this reaction.
(b) Will the equilibrium constant for the reaction increase or decrease with increasing temperature? Explain. (c) At constant temperature, would a change in the volume of the container affect the fraction of products in the equilibrium mixture?
15.66 Methanol (CH3OH) can be made by the reaction of CO with H2:
![]()
(a) Use thermochemical data in Appendix C to calculate ΔH° for this reaction. (b) To maximize the equilibrium yield of methanol, would you use a high or low temperature? (c) To maximize the equilibrium yield of methanol, would you use a high or low pressure?
______
15.67 Ozone, O3, decomposes to molecular oxygen in the stratosphere according to the reaction 2 O3(g) —> 3 O2(g). Would an increase in pressure favor the formation of ozone or of oxygen?
15.68 Bioremediation is the use of microorganisms to degrade environmental pollutants. Many pollutants contain only carbon and hydrogen (oil being one example). The chemical reactions are complicated, but in general the microorganisms react the pollutant hydrocarbon with O2 to produce CO2 and other carbon-containing compounds that are incorporated into the organism's biomass. How would increasing levels of CO2 in the environment affect the bioremediation reaction?
ADDITIONAL EXERCISES
15.69 Both the forward reaction and the reverse reaction in the following equilibrium are believed to be elementary steps:
![]()
At 25 °C the rate constants for the forward and reverse reactions are 1.4 × 10–28M–1 s–1 and 9.3 × 1010M–1 s–1, respectively. (a) What is the value for the equilibrium constant at 25 °C? (b) Are reactants or products more plentiful at equilibrium? (c) What additional information would you need in order to decide whether the reaction as written is endothermic or exothermic?
15.70 If Kc = 1 for the equilibrium 2 A(g) ![]() B(g), what is the relationship between [A] and [B] at equilibrium?
B(g), what is the relationship between [A] and [B] at equilibrium?
15.71 A mixture of CH4 and H2O is passed over a nickel catalyst at 1000 K. The emerging gas is collected in a 5.00-L flask and is found to contain 8.62 g of CO, 2.60 g of H2,43.0 g of CH4, and 48.4 g of H2O. Assuming that equilibrium has been reached, calculate Kc and Kp for the reaction.
15.72 When 2.00 mol of SO2Cl2 is placed in a 2.00-L flask at 303 K, 56% of the SO2Cl2 decomposes to SO2 and Cl2:
![]()
(a) Calculate Kc for this reaction at this temperature. (b) Calculate Kp for this reaction at 303 K. (c) Repeat these calculations for 2.00 mol of SO2Cl2 in a 15.00-L vessel at 303 K.
15.73 A mixture of H2, S, and H2S is held in a 1.0-L vessel at 90 °C and reacts according to the equation:
![]()
At equilibrium the mixture contains 0.46 g of H2S and 0.40 g H2. (a) Write the equilibrium-constant expression for this reaction. (b) What is the value of Kc for the reaction at this temperature? (c) Why can we ignore the amount of S when doing the calculation in part (b)?
15.74 A sample of nitrosyl bromide (NOBr) decomposes according to the equation
![]()
An equilibrium mixture in a 5.00-L vessel at 100 °C contains 3.22 g of NOBr, 3.08 g of NO, and 4.19 g of Br2. (a) Calculate Kc. (b) What is the total pressure exerted by the mixture of gases? (c) What was the mass of the original sample of NOBr?
15.75 Consider the hypothetical reaction A(g) ![]() 2 B(g).A flask is charged with 0.75 atm of pure A, after which it is allowed to reach equilibrium at 0 °C. At equilibrium the partial pressure of A is 0.36 atm. (a) What is the total pressure in the flask at equilibrium? (b) What is the value of Kp? (c) What could we do to maximize the yield of B?
2 B(g).A flask is charged with 0.75 atm of pure A, after which it is allowed to reach equilibrium at 0 °C. At equilibrium the partial pressure of A is 0.36 atm. (a) What is the total pressure in the flask at equilibrium? (b) What is the value of Kp? (c) What could we do to maximize the yield of B?
15.76 As shown in Table 15.2, the equilibrium constant for the reaction N2(g) + 3 H2(g) ![]() 2 NH3(g) is Kp = 4.34 × 10–3 at 300 °C. Pure NH3 is placed in a 1.00-L flask and allowed to reach equilibrium at this temperature. There are 1.05 g NH3 in the equilibrium mixture. (a) What are the masses of N2 and H2 in the equilibrium mixture? (b) What was the initial mass of ammonia placed in the vessel? (c) What is the total pressure in the vessel?
2 NH3(g) is Kp = 4.34 × 10–3 at 300 °C. Pure NH3 is placed in a 1.00-L flask and allowed to reach equilibrium at this temperature. There are 1.05 g NH3 in the equilibrium mixture. (a) What are the masses of N2 and H2 in the equilibrium mixture? (b) What was the initial mass of ammonia placed in the vessel? (c) What is the total pressure in the vessel?
15.77 For the equilibrium
![]()
Kp = 8.5 × 10–3 at 150 °C. If 0.025 atm of IBr is placed in a 2.0-L container, what is the partial pressure of all substances after equilibrium is reached?
15.78 For the equilibrium
![]()
Kp = 0.052 at 60 °C. (a) Calculate Kc. (b) After 3.00 g of solid PH3BCl3 is added to a closed 1.500-L vessel at 60 °C, the vessel is charged with 0.0500 g of BCl3(g). What is the equilibrium concentration of PH3?
[15.79] Solid NH4SH is introduced into an evacuated flask at 24 °C. The following reaction takes place:
![]()
At equilibrium the total pressure (for NH3 and H2S taken together) is 0.614 atm. What is Kp for this equilibrium at 24 °C?
[15.80] A 0.831-g sample of SO3 is placed in a 1.00-L container and heated to 1100 K. The SO3 decomposes to SO2 and O2:
![]()
At equilibrium the total pressure in the container is 1.300 atm. Find the values of Kp and Kc for this reaction at 1100 K.
15.81 Nitric oxide (NO) reacts readily with chlorine gas as follows:
![]()
At 700 K the equilibrium constant Kp for this reaction is 0.26. Predict the behavior of each of the following mixtures at this temperature and indicate whether or not the mixtures are at equilibrium. If not, state whether the mixture will need to produce more products or reactants to reach equilibrium. (a) PNO = 0.15 atm, PCl2 = 0.31 atm, and PNOCl = 0.11 atm; (b) PNO = 0.12 atm,PCl2 = 0.10 atm, and PNOCl = 0.050 atm; (c) PNO = 0.15 atm, PCl2 = 0.20 atm, and PNOCl = 5.10 × 10–3 atm.
15.82 At 900 °C, Kc = 0.0108 for the reaction
![]()
A mixture of CaCO3, CaO, and CO2 is placed in a 10.0-L vessel at 900 °C. For the following mixtures, will the amount of CaCO3 increase, decrease, or remain the same as the system approaches equilibrium?

15.83 When 1.50 mol CO2 and 1.50 mol H2 are placed in a 3.00-L container at 395 °C, the following reaction occurs: CO2(g) + H2(g) ![]() CO(g) + H2O(g). If KC = 0.802, what are the concentrations of each substance in the equilibrium mixture?
CO(g) + H2O(g). If KC = 0.802, what are the concentrations of each substance in the equilibrium mixture?
15.84 The equilibrium constant Kc for C(s) + CO2(g) ![]() 2 CO(g) is 1.9 at 1000 K and 0.133 at 298 K. (a) If excess C is allowed to react with 25.0 g of CO2 in a 3.00-L vessel at 1000 K, how many grams of CO are produced? (b) How many grams of C are consumed? (c) If a smaller vessel is used for the reaction, will the yield of CO be greater or smaller? (d) Is the reaction endothermic or exothermic?
2 CO(g) is 1.9 at 1000 K and 0.133 at 298 K. (a) If excess C is allowed to react with 25.0 g of CO2 in a 3.00-L vessel at 1000 K, how many grams of CO are produced? (b) How many grams of C are consumed? (c) If a smaller vessel is used for the reaction, will the yield of CO be greater or smaller? (d) Is the reaction endothermic or exothermic?
15.85 NiO is to be reduced to nickel metal in an industrial process by use of the reaction
![]()
At 1600 K the equilibrium constant for the reaction is Kp = 6.0 × 102. If a CO pressure of 150 torr is to be employed in the furnace and total pressure never exceeds 760 torr, will reduction occur?
15.86 Le Châtelier noted that many industrial processes of his time could be improved by an understanding of chemical equilibria. For example, the reaction of iron oxide with carbon monoxide was used to produce elemental iron and CO2 according to the reaction
![]()
Even in Le Châtelier's time, it was noted that a great deal of CO was wasted, expelled through the chimneys over the furnaces. Le Châtelier wrote, “Because this incomplete reaction was thought to be due to an insufficiently prolonged contact between carbon monoxide and the iron ore [oxide], the dimensions of the furnaces have been increased. In England they have been made as high as thirty meters. But the proportion of carbon monoxide escaping has not diminished, thus demonstrating, by an experiment costing several hundred thousand francs, that the reduction of iron oxide by carbon monoxide is a limited reaction. Acquaintance with the laws of chemical equilibrium would have permitted the same conclusion to be reached more rapidly and far more economically.” What does this anecdote tell us about the equilibrium constant for this reaction?
[15.87] At 700 K the equilibrium constant for the reaction
![]()
is Kp = 0.76. A flask is charged with 2.00 atm of CCl4, which then reaches equilibrium at 700 K. (a) What fraction of the CCl4 is converted into C and Cl2? (b) What are the partial pressures of CCl4 and Cl2 at equilibrium?
[15.88] The reaction PCl3(g) + Cl2(g) ![]() PCl5(g) has Kp = 0.0870 at 300 °C. A flask is charged with 0.50 atm PCl3, 0.50 atm Cl2, and 0.20 atm PCl5 at this temperature. (a) Use the reaction quotient to determine the direction the reaction must proceed to reach equilibrium. (b)Calculate the equilibrium partial pressures of the gases. (c) What effect will increasing the volume of the system have on the mole fraction of Cl2 in the equilibrium mixture? (d) The reaction is exothermic. What effect will increasing the temperature of the system have on the mole fraction of Cl2 in the equilibrium mixture?
PCl5(g) has Kp = 0.0870 at 300 °C. A flask is charged with 0.50 atm PCl3, 0.50 atm Cl2, and 0.20 atm PCl5 at this temperature. (a) Use the reaction quotient to determine the direction the reaction must proceed to reach equilibrium. (b)Calculate the equilibrium partial pressures of the gases. (c) What effect will increasing the volume of the system have on the mole fraction of Cl2 in the equilibrium mixture? (d) The reaction is exothermic. What effect will increasing the temperature of the system have on the mole fraction of Cl2 in the equilibrium mixture?
[15.89] An equilibrium mixture of H2, I2, and HI at 458 °C contains 0.112 mol H2, 0.112 mol I2, and 0.775 mol HI in a 5.00-L vessel. What are the equilibrium partial pressures when equilibrium is reestablished following the addition of 0.200 mol of HI?
[15.90] Consider the hypothetical reaction A(g) + 2 B(g) ![]() 2 C(g), for which Kc = 0.25 at a certain temperature. A 1.00-L reaction vessel is loaded with 1.00 mol of compound C, which is allowed to reach equilibrium. Let the variable x represent the number of mol/L of compound A present at equilibrium. (a) In terms of x, what are the equilibrium concentrations of compounds B and C? (b) What limits must be placed on the value of x so that all concentrations are positive? (c) By putting the equilibrium concentrations (in terms of x) into the equilibrium-constant expression, derive an equation that can be solved for x. (d) The equation from part (c) is a cubic equation (one that has the form ax3 + bx2 + cx + d = 0). In general, cubic equations cannot be solved in closed form. However, you can estimate the solution by plotting the cubic equation in the allowed range of x that you specified in part (b). The point at which the cubic equation crosses the x-axis is the solution. (e) From the plot in part (d), estimate the equilibrium concentrations of A, B, and C. (Hint: You can check the accuracy of your answer by substituting these concentrations into the equilibrium expression.)
2 C(g), for which Kc = 0.25 at a certain temperature. A 1.00-L reaction vessel is loaded with 1.00 mol of compound C, which is allowed to reach equilibrium. Let the variable x represent the number of mol/L of compound A present at equilibrium. (a) In terms of x, what are the equilibrium concentrations of compounds B and C? (b) What limits must be placed on the value of x so that all concentrations are positive? (c) By putting the equilibrium concentrations (in terms of x) into the equilibrium-constant expression, derive an equation that can be solved for x. (d) The equation from part (c) is a cubic equation (one that has the form ax3 + bx2 + cx + d = 0). In general, cubic equations cannot be solved in closed form. However, you can estimate the solution by plotting the cubic equation in the allowed range of x that you specified in part (b). The point at which the cubic equation crosses the x-axis is the solution. (e) From the plot in part (d), estimate the equilibrium concentrations of A, B, and C. (Hint: You can check the accuracy of your answer by substituting these concentrations into the equilibrium expression.)
15.91 At 1200 K, the approximate temperature of automobile exhaust gases (Figure 15.15), Kp for the reaction
![]()
is about 1 × 10–13. Assuming that the exhaust gas (total pressure 1 atm) contains 0.2% CO, 12% CO2, and 3% O2 by volume, is the system at equilibrium with respect to the CO2 reaction? Based on your conclusion, would the CO concentration in the exhaust be decreased or increased by a catalyst that speeds up the CO2 reaction?
15.92 Suppose that you worked at the U.S. Patent Office and a patent application came across your desk claiming that a newly developed catalyst was much superior to the Haber catalyst for ammonia synthesis because the catalyst led to much greater equilibrium conversion of N2 and H2 into NH3 than the Haber catalyst under the same conditions. What would be your response?
INTEGRATIVE EXERCISES
15.93 Consider the reaction IO4–(aq) + 2 H2O(l) ![]() H4IO6(aq); Kc = 3.5 × 10–2. If you start with 25.0 mL of a 0.905 M solution of NaIO4, and then dilute it with water to 500.0 mL, what is the concentration of H4IO6– at equilibrium?
H4IO6(aq); Kc = 3.5 × 10–2. If you start with 25.0 mL of a 0.905 M solution of NaIO4, and then dilute it with water to 500.0 mL, what is the concentration of H4IO6– at equilibrium?
[15.94] Silver chloride, AgCl(s), is an “insoluble” strong electrolyte. (a) Write the equation for the dissolution of AgCl(s) in H2O(l). (b) Write the expression for Kc for the reaction in part (a). (c) Based on the thermochemical data in Appendix C and Le Châtelier's principle, predict whether the solubility of AgCl in H2O increases or decreases with increasing temperature. (d) The equilibrium constant for the dissolution of AgCl in water is 1.6 × 10–10 at 25 °C. In addition, Ag (aq) can react with Cl (aq) according to the reaction
![]()
where Kc = 1.8 × 105 at 25 °C. Although AgCl is “not soluble” in water, the complex AgCl2– is soluble. At 25 °C, is the solubility of AgCl in a 0.100 M NaCl solution greater than the solubility of AgCl in pure water, due to the formation of soluble AgCl2– ions? Or is the AgCl solubility in 0.100 M NaCl less than in pure water because of a Le Châtelier-type argument? Justify your answer with calculations. (Hint: Any form in which silver is in solution counts as “solubility.”)
[15.95] Consider the equilibrium A ![]() B in which both the forward and reverse reactions are elementary (single-step) reactions. Assume that the only effect of a catalyst on the reaction is to lower the activation energies of the forward and reverse reactions, as shown in Figure 15.14. Using the Arrhenius equation (Section 14.5), prove that the equilibrium constant is the same for the catalyzed reaction as for the uncatalyzed one.
B in which both the forward and reverse reactions are elementary (single-step) reactions. Assume that the only effect of a catalyst on the reaction is to lower the activation energies of the forward and reverse reactions, as shown in Figure 15.14. Using the Arrhenius equation (Section 14.5), prove that the equilibrium constant is the same for the catalyzed reaction as for the uncatalyzed one.
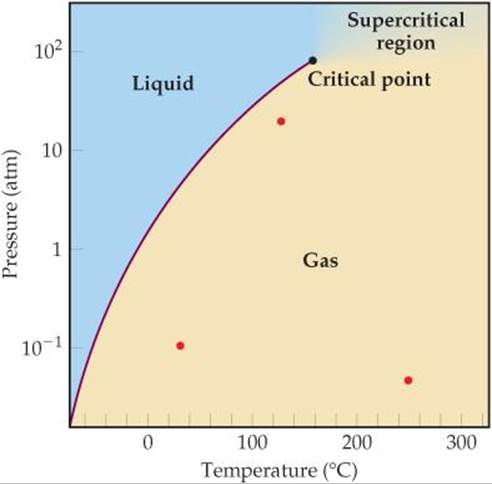
[15.96] The phase diagram for SO2 is shown here. (a) What does this diagram tell you about the enthalpy change in the reaction SO2(ℓ) ![]() SO2(g)? (b) Calculate the equilibrium constant for this reaction at 100 °C and at 0 °C. (c) Why is it not possible to calculate an equilibrium constant between the gas and liquid phases in the supercritical region? (d) At which of the three points marked in red does SO2(g) most closely approach ideal-gas behavior? (e) At which of the three red points does SO2(g) behave least ideally?
SO2(g)? (b) Calculate the equilibrium constant for this reaction at 100 °C and at 0 °C. (c) Why is it not possible to calculate an equilibrium constant between the gas and liquid phases in the supercritical region? (d) At which of the three points marked in red does SO2(g) most closely approach ideal-gas behavior? (e) At which of the three red points does SO2(g) behave least ideally?
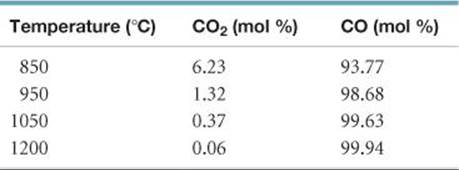
[15.97] Write the equilibrium-constant expression for the equilibrium
C(s) + CO2(g) ![]() 2 CO(g)
2 CO(g)
The table that follows shows the relative mole percentages of CO2(g) and CO(g) at a total pressure of 1 atm for several temperatures. Calculate the value of Kp at each temperature. Is the reaction exothermic or endothermic? Explain.
15.98 In Section 11.5 we defined the vapor pressure of a liquid in terms of an equilibrium. (a) Write the equation representing the equilibrium between liquid water and water vapor and the corresponding expression for Kp. (b) By using data in Appendix B, give the value of Kp for this reaction at 30 °C. (c) What is the value of Kp for any liquid in equilibrium with its vapor at the normal boiling point of the liquid?
15.99 Water molecules in the atmosphere can form hydrogen-bonded dimers, (H2O)2. The presence of these dimers is thought to be important in the nucleation of ice crystals in the atmosphere and in the formation of acid rain. (a) Using VSEPR theory, draw the structure of a water dimer, using dashed lines to indicate intermolecular interactions. (b) What kind of intermolecular forces are involved in water dimer formation? (c) The Kp for water dimer formation in the gas phase is 0.050 at 300 K and 0.020 at 350 K. Is water dimer formation endothermic or exothermic?
15.100 The protein hemoglobin (Hb) transports O2 in mammalian blood. Each Hb can bind 4 O2 molecules. The equilibrium constant for the O2-binding reaction is higher in fetal hemoglobin than in adult hemoglobin. In discussing protein oxygenbinding capacity, biochemists use a measure called the P50 value, defined as the partial pressure of oxygen at which 50% of the protein is saturated. Fetal hemoglobin has a P50 value of 19 torr, and adult hemoglobin has a P50 value of 26.8 torr. Use these data to estimate how much larger Kc is for the aqueous reaction 4 O2(g) + Hb(aq) ![]() [Hb(O2)4(aq)].
[Hb(O2)4(aq)].