CHEMISTRY THE CENTRAL SCIENCE
9 MOLECULAR GEOMETRY AND BONDING THEORIES
EXERCISES
VISUALIZING CONCEPTS
9.1 A certain AB4 molecule has a “seesaw” shape:
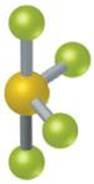
From which of the fundamental geometries shown in Figure 9.3 could you remove one or more atoms to create a molecule having this seesaw shape? [Section 9.1]
9.2 (a) If these three balloons are all the same size, what angle is formed between the red one and the green one? (b) If additional air is added to the blue balloon so that it gets larger, what happens to the angle between the red and green balloons? (c) What aspect of the VSEPR model is illustrated by part (b)? [Section 9.2]

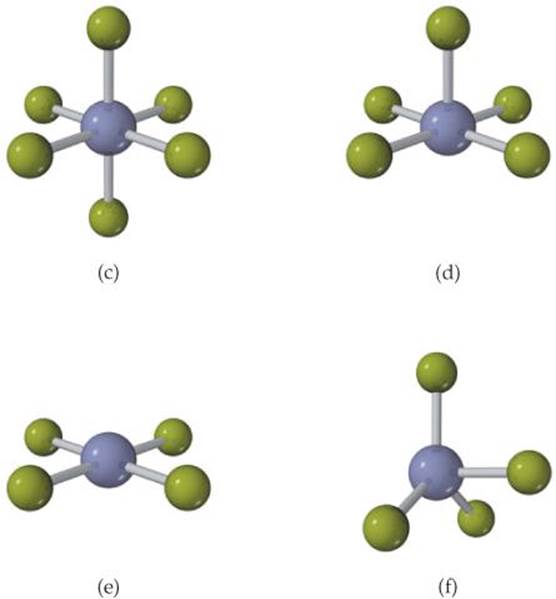
9.3 For each molecule (a)-(f), indicate how many different electron-domain geometries are consistent with the molecular geometry shown. [Section 9.2]

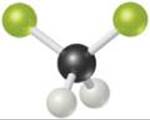
9.4 The molecule shown here is difluoromethane (CH2F2), which is used as a refrigerant called R-32. (a) Based on the structure, how many electron domains surround the C atom in this molecule? (b) Would the molecule have a nonzero dipole moment? (c) If the molecule is polar, in what direction will the overall dipole moment vector point in the molecule? [Sections 9.2 and 9.3]
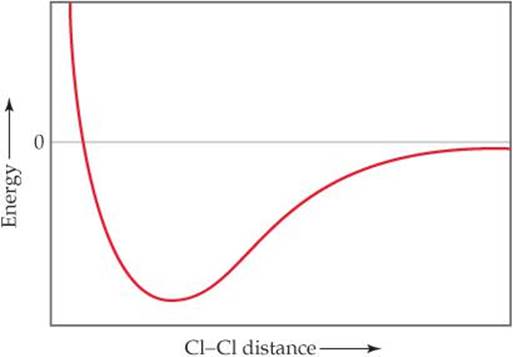
9.5 The following plot shows the potential energy of two Cl atoms as a function of the distance between them. (a) To what does an energy of zero correspond in this diagram? (b) According to the valence-bond model, why does the energy decrease as the Cl atoms move from a large separation to a smaller one? (c) What is the significance of the Cl—Cl distance at the minimum point in the plot? (d) Why does the energy rise at Cl—Cl distances less than that at the minimum point in the plot? (e) How can you estimate the bond strength of the Cl—Cl bond from the plot? [Section 9.4]
9.6 In the series SiF4, PF3, and SF2, estimate the F—X—F bond angle in each case and explain your rationale. [Section 9.2]
9.7 The orbital diagram that follows presents the final step in the formation of hybrid orbitals by a silicon atom. What type of hybrid orbital is produced in this hybridization? [Section 9.5]
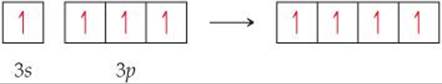
9.8 In the hydrocarbon
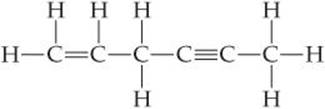
(a) What is the hybridization at each carbon atom in the molecule? (b) How many σ bonds are there in the molecule? (c) How many π bonds? (d) Identify all the 120° bond angles in the molecule. [Section 9.6]
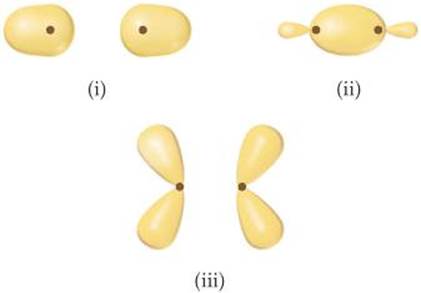
9.9 For each of these contour representations of molecular or-bitals, identify (a) the atomic orbitals (s or p) used to construct the MO (b) the type of MO (σ or π), (c) whether the MO is bonding or antibonding, and (d) the locations of nodal planes. [Sections 9.7 and 9.8]
9.10 The diagram that follows shows the highest-energy occupied MOs of a neutral molecule CX, where element X is in the same row of the periodic table as C. (a) Based on the number of electrons, can you determine the identity of X? (b) Would the molecule be diamagnetic or paramagnetic? (c) Consider the π2p MOs of the molecule. Would you expect them to have a greater atomic orbital contribution from C, have a greater atomic orbital contribution from X, or be an equal mixture of atomic orbitals from the two atoms? [Section 9.8]
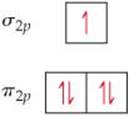
MOLECULAR SHAPES; THE VSEPR MODEL (sections 9.1 and 9.2)
9.11 An AB2 molecule is described as linear, and the A—B bond length is known. (a) Does this information completely describe the geometry of the molecule? (b) Can you tell how many nonbonding pairs of electrons are around the A atom from this information?
9.12 (a) Methane (CH4) and the perchlorate ion (ClO4–) are both described as tetrahedral. What does this indicate about their bond angles? (b) The NH3 molecule is trigonal pyramidal, while BF3 is trigonal planar. Which of these molecules is flat?
______
9.13 How does a trigonal pyramid differ from a tetrahedron so far as molecular geometry is concerned?
9.14 Describe the bond angles to be found in each of the following molecular structures: (a) planar trigonal, (b) tetrahedral, (c) octahedral, (d) linear.
______
9.15 (a) What is meant by the term electron domain? (b) Explain in what way electron domains behave like the balloons in Figure 9.5. Why do they do so?
9.16 What property of the electron causes electron domains to have an effect on molecular shapes?
______
9.17 (a) How does one determine the number of electron domains in a molecule or ion? (b) What is the difference between a bonding electron domain and a nonbonding electron domain?
9.18 Would you expect the nonbonding electron-pair domain in NH3 to be greater or less in size than for the corresponding one in PH3? Explain.
______
9.19 In which of these molecules or ions does the presence of nonbonding electron pairs produce an effect on molecular shape, assuming they are all in the gaseous state? (a) SiH4, (b) PF3, (c) HBr, (d) HCN, (e) SO2.
9.20 In which of the following molecules can you confidently predict the bond angles about the central atom, and for which would you be a bit uncertain? Explain in each case. (a) H2S, (b) BCl3, (c) CH3I, (d) CBr4, (e) TeBr4.
______
9.21 How many nonbonding electron pairs are there in each of the following molecules: (a) (CH3)2S, (b) HCN, (c) H2C2, (d) CH3F?
9.22 Describe the characteristic electron-domain geometry of each of the following numbers of electron domains about a central atom: (a) 3, (b) 4, (c) 5, (d) 6.
______
9.23 What is the difference between the electron-domain geometry and the molecular geometry of a molecule? Use the water molecule as an example in your discussion. Why do we need to make this distinction?
9.24 An AB3 molecule is described as having a trigonal-bipyramidal electron-domain geometry. How many nonbonding domains are on atom A? Explain.
______
9.25 Give the electron-domain and molecular geometries of a molecule that has the following electron domains on its central atom: (a) four bonding domains and no nonbonding domains, (b) three bonding domains and two nonbonding domains, (c) five bonding domains and one nonbonding domain, (d) four bonding domains and two nonbonding domains.
9.26 What are the electron-domain and molecular geometries of a molecule that has the following electron domains on its central atom? (a) three bonding domains and no nonbonding domains, (b) three bonding domains and one nonbonding domain, (c) two bonding domains and two nonbonding domains.
______
9.27 Give the electron-domain and molecular geometries for the following molecules and ions: (a) HCN, (b) SO32–, (c) SF4, (d) PF6–, (e) NH3Cl+, (f) N3–.
9.28 Draw the Lewis structure for each of the following molecules or ions, and predict their electron-domain and molecular geometries: (a) AsF3, (b) CH3+, (c) BrF3, (d) ClO3–, (e) XeF2, (f) BrO2–.
______
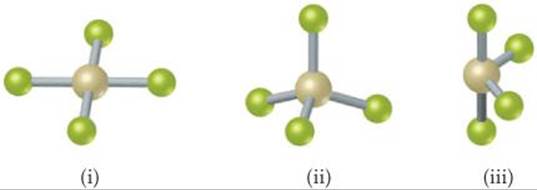
9.29 The figure that follows shows ball-and-stick drawings of three possible shapes of an AF3 molecule. (a) For each shape, give the electron-domain geometry on which the molecular geometry is based. (b) For each shape, how many nonbonding electron domains are there on atom A?(c) Which of the following elements will lead to an AF3 molecule with the shape in (ii): Li, B, N, Al, P, Cl? (d) Name an element A that is expected to lead to the AF3 structure shown in (iii). Explain your reasoning.
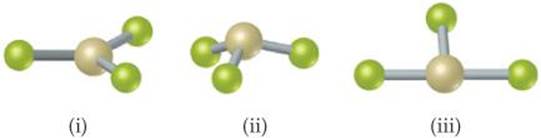
9.30 The figure that follows contains ball-and-stick drawings of three possible shapes of an AF4 molecule. (a) For each shape, give the electron-domain geometry on which the molecular geometry is based. (b) For each shape, how many nonbonding electron domains are there on atom A? (c) Which of the following elements will lead to an AF4 molecule with the shape in (iii): Be, C, S, Se, Si, Xe? (d) Name an element A that is expected to lead to the AF4 structure shown in (i).
9.31 Give the approximate values for the indicated bond angles in the following molecules:
9.32 Give approximate values for the indicated bond angles in the following molecules:

______
9.33 In which of the following AFn molecules or ions is there more than one F—A—F bond angle: SiF4, PF5, SF4, AsF3?
9.34 The three species NH2–, NH3, and NH4+ have H — N — H bond angles of 105°, 107°, and 109°, respectively. Explain this variation in bond angles.
______
9.35 (a) Explain why BrF4– is square planar, whereas BF4– is tetrahedral. (b) How would you expect the H—X—H bond angle to vary in the series H2O, H2S, H2Se? Explain. (Hint: The size of an electron pair domain depends in part on the electronegativity of the central atom.)
9.36 (a) Explain why the following ions have different bond angles: ClO2– and NO2–. Predict the bond angle in each case. (b) Explain why the XeF2 molecule is linear.
SHAPES AND POLARITY OF POLYATOMIC MOLECULES (section 9.3)
9.37 What is the distinction between a bond dipole and a molecular dipole moment?
9.38 Consider a molecule with formula AX3. Supposing the A—X bond is polar, how would you expect the dipole moment of the AX3 molecule to change as the X—A—X bond angle increases from 100° to 120°?
______
9.39 (a) Does SCl2 have a dipole moment? If so, in which direction does the net dipole point? (b) Does BeCl2 have a dipole moment? If so, in which direction does the net dipole point?
9.40 (a) The PH3 molecule is polar. Does this offer experimental proof that the molecule cannot be planar? Explain. (b) It turns out that ozone, O3, has a small dipole moment. How is this possible, given that all the atoms are the same?
______
9.41 (a) Consider the AF3 molecules in Exercise 9.29. Which of these will have a nonzero dipole moment? Explain. (b) Which of the AF4 molecules in Exercise 9.30 will have a zero dipole moment?
9.42 (a) What conditions must be met if a molecule with polar bonds is nonpolar? (b) What geometries will signify nonpolar molecules for AB2, AB3, and AB4 geometries?
______
9.43 Predict whether each of the following molecules is polar or nonpolar: (a) IF, (b) CS2, (c) SO3, (d) PCl3, (e) SF6, (f) IF5.
9.44 Predict whether each of the following molecules is polar or nonpolar: (a) CCl4, (b) NH3, (c) SF4, (d) XeF4, (e) CH3Br, (f) GaH3.
______
9.45 Dichloroethylene (C2H2Cl2) has three forms (isomers), each of which is a different substance. (a) Draw Lewis structures of the three isomers, all of which have a carbon–carbon double bond. (b) Which of these isomers has a zero dipole moment? (c) How many isomeric forms can chloroethylene, C2H3Cl, have? Would they be expected to have dipole moments?
9.46 Dichlorobenzene, C6H4Cl2, exists in three forms (isomers) called ortho, meta, and para:
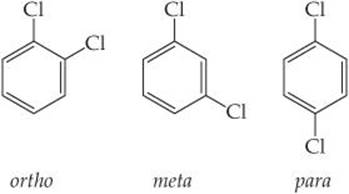
Which of these has a nonzero dipole moment? Explain.
ORBITAL OVERLAP; HYBRID ORBITALS (sections 9.4 and 9.5)
9.47 (a) What is meant by the term orbital overlap? (b) Describe what a chemical bond is in terms of electron density between two atoms.
9.48 Draw sketches illustrating the overlap between the following or-bitals on two atoms: (a) the 2s orbital on each atom, (b) the 2pz orbital on each atom (assume both atoms are on the z-axis), (c) the 2s orbital on one atom and the 2pz orbital on the other atom.
______
9.49 Consider the bonding in an MgH2 molecule. (a) Draw a Lewis structure for the molecule, and predict its molecular geometry. (b) What hybridization scheme is used in MgH2? (c) Sketch one of the two-electron bonds between an Mg hybrid orbital and an H 1s atomic orbital.
9.50 How would you expect the extent of overlap of the bonding atomic orbitals to vary in the series IF, ICl, IBr, and I2? Explain your answer.
______
9.51 Fill in the blank spaces in the following chart. If the molecule column is blank, find an example that fulfills the conditions of the rest of the row.
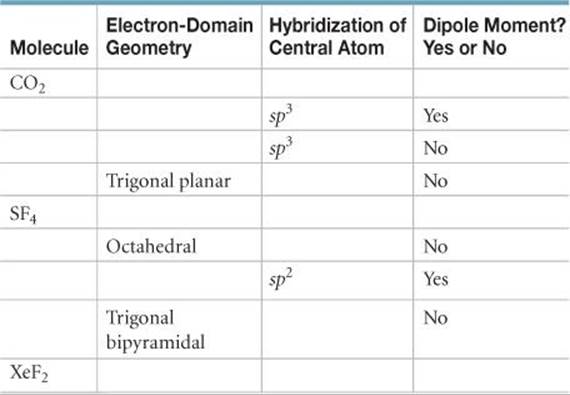
9.52 Why are there no sp4 or sp5 hybrid orbitals?
______
9.53 (a) Starting with the orbital diagram of a boron atom, describe the steps needed to construct hybrid orbitals appropriate to describe the bonding in BF3. (b) What is the name given to the hybrid orbitals constructed in (a)? (c) Sketch the large lobes of the hybrid orbitals constructed in part (a). (d) Are any valence atomic orbitals of B left unhybridized? If so, how are they oriented relative to the hybrid orbitals?
9.54 (a) Starting with the orbital diagram of a sulfur atom, describe the steps needed to construct hybrid orbitals appropriate to describe the bonding in SF2. (b) What is the name given to the hybrid orbitals constructed in (a)? (c) Sketch the large lobes of these hybrid orbitals. (d) Would the hybridization scheme in part (a) be appropriate for SF4? Explain.
______
9.55 Indicate the hybridization of the central atom in (a) BCl3, (b) AlCl4–, (c) CS2, (d) GeH4.
9.56 What is the hybridization of the central atom in (a) SiCl4, (b) HCN, (c) SO3, (d) TeCl2.
______
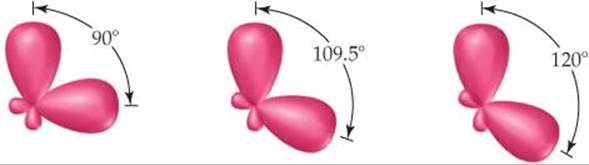
9.57 Shown here are three pairs of hybrid orbitals, with each set at a characteristic angle. For each pair, determine the type of hybridization, if any, that could lead to hybrid orbitals at the specified angle.
9.58 (a) Which geometry and central atom hybridization would you expect in the series BH4–, CH4, NH4+? (b) What would you expect for the magnitude and direction of the bond dipoles in this series? (c) Write the formulas for the analogous species of the elements of period 3; would you expect them to have the same hybridization at the central atom?
MULTIPLE BONDS (section 9.6)
9.59 (a) Draw a picture showing how two p orbitals on two different atoms can be combined to make a sigma bond. (b) Sketch a π bond that is constructed from p orbitals. (c) Which is generally stronger, a σ bond or a π bond? Explain. (d) Can two s orbitals combine to form a π bond? Explain.
9.60 (a) If the valence atomic orbitals of an atom are sp hybridized, how many unhybridized p orbitals remain in the valence shell? How many π bonds can the atom form? (b) Imagine that you could hold two atoms that are bonded together, twist them, and not change the bond length. Would it be easier to twist (rotate) around a single σ bond or around a double (σ plus π) bond, or would they be the same? Explain.
______
9.61 (a) Draw Lewis structures for ethane (C2H6), ethylene (C2H4), and acetylene (C2H2). (b) What is the hybridization of the carbon atoms in each molecule? (c) Predict which molecules, if any, are planar. (d) How many σ and π bonds are there in each molecule? (e) Suppose that silicon could form molecules that are precisely the analogs of ethane, ethylene, and acetylene. How would you describe the bonding about Si in terms of hydrid or-bitals? Silicon does not readily form some of the analogous compounds containing π bonds. Why might this be the case?
9.62 The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms in each molecule? (c) Which molecule has the stronger N—N bond?
______
9.63 Propylene, C3H6, is a gas that is used to form the important polymer called polypropylene. Its Lewis structure is
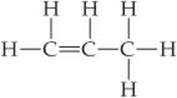
(a) What is the total number of valence electrons in the propylene molecule? (b) How many valence electrons are used to make σ bonds in the molecule? (c) How many valence electrons are used to make π bonds in the molecule? (d) How many valence electrons remain in nonbonding pairs in the molecule? (e) What is the hybridization at each carbon atom in the molecule?
9.64 Ethyl acetate, C4H8O2, is a fragrant substance used both as a solvent and as an aroma enhancer. Its Lewis structure is
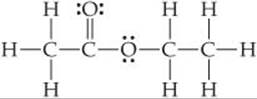
(a) What is the hybridization at each of the carbon atoms of the molecule? (b) What is the total number of valence electrons in ethyl acetate? (c) How many of the valence electrons are used to make σ bonds in the molecule? (d) How many valence electrons are used to make π bonds? (e) How many valence electrons remain in nonbonding pairs in the molecule?
______
9.65 Consider the Lewis structure for glycine, the simplest amino acid:
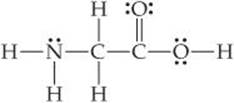
(a) What are the approximate bond angles about each of the two carbon atoms, and what are the hybridizations of the or-bitals on each of them? (b) What are the hybridizations of the orbitals on the two oxygens and the nitrogen atom, and what are the approximate bond angles at the nitrogen? (c) What is the total number of σ bonds in the entire molecule, and what is the total number of π bonds?
9.66 Acetylsalicylic acid, better known as aspirin, has the Lewis structure
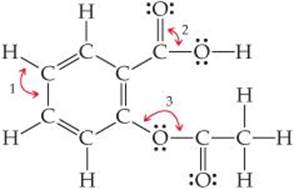
(a) What are the approximate values of the bond angles labeled 1, 2, and 3? (b) What hybrid orbitals are used about the central atom of each of these angles? (c) How many σ bonds are in the molecule?
______
9.67 (a) What is the difference between a localized π bond and a delocalized one? (b) How can you determine whether a molecule or ion will exhibit delocalized π bonding? (c) Is the π bond in NO2– localized or delocalized?
9.68 (a) Write a single Lewis structure for SO3, and determine the hybridization at the S atom. (b) Are there other equivalent Lewis structures for the molecule? (c) Would you expect SO3 to exhibit delocalized π bonding? Explain.
______
9.69 Predict the molecular geometry of each of the following molecules:
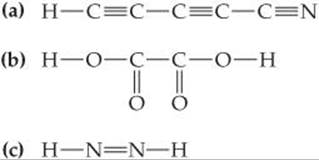
9.70 What hybridization do you expect for the atom indicated in red in each of the following species?
![]() ;
; ![]() ;
; ![]() ;
; ![]()
MOLECULAR ORBITALS AND SECOND-ROW DIATOMIC MOLECULES (sections 9.7 and 9.8)
9.71 (a) What is the difference between hybrid orbitals and molecular orbitals? (b) How many electrons can be placed into each MO of a molecule? (c) Can antibonding molecular orbitals have electrons in them?
9.72 (a) If you combine two atomic orbitals on two different atoms to make a new orbital, is this a hybrid orbital or a molecular orbital? (b) If you combine two atomic orbitals on one atom to make a new orbital, is this a hybrid orbital or a molecular orbital? (c) Does the Pauli exclusion principle (Section 6.7) apply to MOs? Explain.
______
9.73 Consider the H2+ ion. (a) Sketch the molecular orbitals of the ion and draw its energy-level diagram. (b) How many electrons are there in the H2+ ion? (c) Draw the electron configuration of the ion in terms of its MOs. (d) What is the bond order in H2+? (e) Suppose that the ion is excited by light so that an electron moves from a lower-energy to a higher-energy MO. Would you expect the excited-state H2+ ion to be stable or to fall apart? Explain.
9.74 (a) Sketch the molecular orbitals of the H2– ion and draw its energy-level diagram. (b) Write the electron configuration of the ion in terms of its MOs. (c) Calculate the bond order in H2–. (d) Suppose that the ion is excited by light, so that an electron moves from a lower-energy to a higher-energy molecular orbital. Would you expect the excited-state H2– ion to be stable? Explain.
______
9.75 Draw a picture that shows all three 2p orbitals on one atom and all three 2p orbitals on another atom. (a) Imagine the atoms coming close together to bond. How many σ bonds can the two sets of 2p orbitals make with each other? (b) How many π bonds can the two sets of 2porbitals make with each other? (c) How many antibonding orbitals, and of what type, can be made from the two sets of 2p orbitals?
9.76 (a) What is the probability of finding an electron on the inter-nuclear axis if the electron occupies a π molecular orbital? (b) For a homonuclear diatomic molecule, what similarities and differences are there between the π2p MO made from the 2px atomic orbitals and the π2p MO made from the 2py atomic or-bitals? (c) How do the ![]() MOs formed from the 2px and 2py atomic orbitals differ from the π2p MOs in terms of energies and electron distributions?
MOs formed from the 2px and 2py atomic orbitals differ from the π2p MOs in terms of energies and electron distributions?
______
9.77 (a) What are the relationships among bond order, bond length, and bond energy? (b) According to molecular orbital theory, would either Be2 or Be2+ be expected to exist? Explain.
9.78 Explain the following: (a) The peroxide ion, O22–, has a longer bond length than the superoxide ion, O2–. (b) The magnetic properties of B2 are consistent with the π2p MOs being lower in energy than the σ2p MO. (c) The O22+ ion has a stronger O—O bond than O2 itself.
______
9.79 (a) What does the term diamagnetism mean? (b) How does a diamagnetic substance respond to a magnetic field? (c) Which of the following ions would you expect to be diamagnetic: N22–, O22–, Be22+, C2–?
9.80 (a) What does the term paramagnetism mean? (b) How can one determine experimentally whether a substance is paramagnetic? (c) Which of the following ions would you expect to be paramagnetic: O2+, N22–, Li2+, O22–? For those ions that are paramagnetic, determine the number of unpaired electrons.
______
9.81 Using Figures 9.35 and 9.43 as guides, draw the molecular orbital electron configuration for (a) B2+, (b) Li2+, (c) N2+, (d) Ne22+. In each case indicate whether the addition of an electron to the ion would increase or decrease the bond order of the species.
9.82 If we assume that the energy-level diagrams for homonuclear diatomic molecules shown in Figure 9.43 can be applied to heteronuclear diatomic molecules and ions, predict the bond order and magnetic behavior of (a) CO+, (b) NO–, (c) OF+, (d) NeF+.
______
9.83 Determine the electron configurations for CN+, CN, and CN–. (a) Which species has the strongest C—N bond? (b) Which species, if any, has unpaired electrons?
9.84 (a) The nitric oxide molecule, NO, readily loses one electron to form the NO+ ion. Why is this consistent with the electronic structure of NO? (b) Predict the order of the N—O bond strengths in NO, NO+, and NO–, and describe the magnetic properties of each. (c) With what neutral homonuclear diatomic molecules are the NO+ and NO– ions isoelectronic (same number of electrons)?
______
[9.85] Consider the molecular orbitals of the P2 molecule. Assume that the MOs of diatomics from the third row of the periodic table are analogous to those from the second row. (a) Which valence atomic orbitals of π are used to construct the MOs of P2? (b) The figure that follows shows a sketch of one of the MOs for P2. What is the label for this MO? (c) For the P2 molecule, how many electrons occupy the MO in the figure? (d) Is P2 expected to be diamagnetic or paramagnetic? Explain.

[9.86] The iodine bromide molecule, IBr, is an interhalogen compound. Assume that the molecular orbitals of IBr are analogous to the homonuclear diatomic molecule F2. (a) Which valence atomic orbitals of I and of Br are used to construct the MOs of IBr? (b) What is the bond order of the IBr molecule? (c) One of the valence MOs of IBr is sketched here. Why are the atomic orbital contributions to this MO different in size? (d) What is the label for the MO? (e) For the IBr molecule, how many electrons occupy the MO?
ADDITIONAL EXERCISES
9.87 (a) What is the physical basis for the VSEPR model? (b) When applying the VSEPR model, we count a double or triple bond as a single electron domain. Why is this justified?
9.88 What is the fundamental basis on which we assign electrons to electron domains in pairs and with their spins paired?
9.89 The molecules SiF4, SF4, and XeF4 have molecular formulas of the type AF4, but the molecules have different molecular geometries. Predict the shape of each molecule, and explain why the shapes differ.
9.90 Consider the molecule PF4Cl. (a) Draw a Lewis structure for the molecule, and predict its electron-domain geometry. (b) Which would you expect to take up more space, a P—F bond or a P—Cl bond? Explain. (c) Predict the molecular geometry of PF4Cl. How did your answer for part (b) influence your answer here in part (c)? (d) Would you expect the molecule to distort from its ideal electron-domain geometry? If so, how would it distort?
[9.91] The vertices of a tetrahedron correspond to four alternating corners of a cube. By using analytical geometry, demonstrate that the angle made by connecting two of the vertices to a point at the center of the cube is 109.5°, the characteristic angle for tetrahedral molecules.
9.92 From their Lewis structures, determine the number of σ and π bonds in each of the following molecules or ions: (a) CO2; (b) cyanogen, (CN)2; (c) formaldehyde, H2CO; (d) formic acid, HCOOH, which has one H and two O atoms attached to C.
9.93 The lactic acid molecule, CH3CH(OH)COOH, gives sour milk its unpleasant, sour taste. (a) Draw the Lewis structure for the molecule, assuming that carbon always forms four bonds in its stable compounds. (b) How many π and how many σ bonds are in the molecule? (c) Which CO bond is shortest in the molecule? (d) What is the hybridization of atomic orbitals around the carbon atom associated with that short bond? (e) What are the approximate bond angles around each carbon atom in the molecule?
9.94 The PF3 molecule has a dipole moment of 1.03 D, but BF3 has a dipole moment of zero. How can you explain the difference?
9.95 An AB5 molecule adopts the geometry shown here. (a) What is the name of this geometry? (b) Do you think there are any non-bonding electron pairs on atom A? Why or why not? (c) Suppose the atoms B are halogen atoms. Can you determine uniquely to which group in the periodic table atom A belongs?
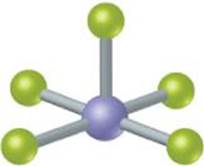
9.96 There are two compounds of the formula Pt(NH3)2Cl2:
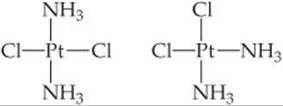
The compound on the right, cisplatin, is used in cancer therapy. The compound on the left, transplatin, is ineffective for cancer therapy. Both compounds have a square-planar geometry. (a) Which compound has a nonzero dipole moment? (b) The reason cisplatin is a good anticancer drug is that it binds tightly to DNA. Cancer cells are rapidly dividing, producing a lot of DNA. Consequently, cisplatin kills cancer cells at a faster rate than normal cells. However, since normal cells also are making DNA, cisplatin also attacks healthy cells, which leads to unpleasant side effects. The way both molecules bind to DNA involves the Cl– ions leaving the Pt ion, to be replaced by two nitrogens in DNA. Draw a picture in which a long vertical line represents a piece of DNA. Draw the Pt(NH3)2 fragments of cisplatin and transplatin with the proper shape. Also draw them attaching to your DNA line. Can you explain from your drawing why the shape of the cisplatin causes it to bind to DNA more effectively than transplatin?
[9.97] The O—H bond lengths in the water molecule (H2O) are 0.96 Å, and the H—O—H angle is 104.5°. The dipole moment of the water molecule is 1.85 D. (a) In what directions do the bond dipoles of the O—H bonds point? In what direction does the dipole moment vector of the water molecule point? (b) Calculate the magnitude of the bond dipole of the O—H bonds. (Note: You will need to use vector addition to do this.) (c) Compare your answer from part (b) to the dipole moments of the hydrogen halides (Table 8.3). Is your answer in accord with the relative electronegativity of oxygen?
[9.98] The reaction of three molecules of fluorine gas with a Xe atom produces the substance xenon hexafluoride, XeF6:
Xe(g) + 3 F2(g) → XeF6(s)
(a) Draw a Lewis structure for XeF6. (b) If you try to use the VSEPR model to predict the molecular geometry of XeF6, you run into a problem. What is it? (c) What could you do to resolve the difficulty in part (b)? (d) The molecule IF7 has a pentagonal-bipyramidal structure (five equatorial fluorine atoms at the vertices of a regular pentagon and two axial fluorine atoms). Based on the structure of IF7, suggest a structure for XeF6.
[9.99] The Lewis structure for allene is

Make a sketch of the structure of this molecule that is analogous to Figure 9.25. In addition, answer the following three questions: (a) Is the molecule planar? (b) Does it have a nonzero dipole moment? (c) Would the bonding in allene be described as delocalized? Explain.
[9.100] The azide ion, N3–, is linear with two N—N bonds of equal length, 1.16 Å. (a) Draw a Lewis structure for the azide ion. (b) With reference to Table 8.5, is the observed N—N bond length consistent with your Lewis structure? (c) What hybridization scheme would you expect at each of the nitrogen atoms in N3–? (d) Show which hybridized and unhybridized orbitals are involved in the formation of σ and π bonds in N3–. (e) It is often observed that σ bonds that involve an sp2 hybrid orbital are shorter than those that involve only sp2 or sp3 hybrid orbitals. Can you propose a reason for this? Is this observation applicable to the observed bond lengths in N3–?
[9.101] In ozone, O3, the two oxygen atoms on the ends of the molecule are equivalent to one another. (a) What is the best choice of hybridization scheme for the atoms of ozone? (b) For one of the resonance forms of ozone, which of the orbitals are used to make bonds and which are used to hold nonbonding pairs of electrons? (c) Which of the orbitals can be used to delocalize the π electrons? (d) How many electrons are delocalized in the π system of ozone?
9.102 Butadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:
![]()
(a) Predict the bond angles around each of the carbon atoms and sketch the molecule.
(b) Compare the bond lengths to the average bond lengths listed in Table 8.5. Can you explain any differences?
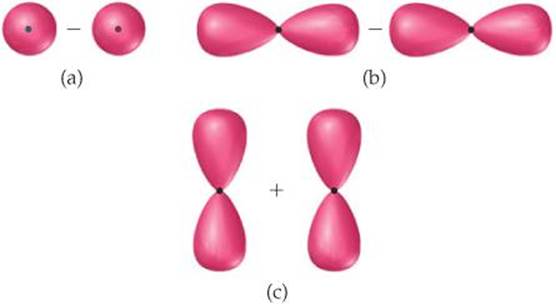
[9.103] The following sketches show the atomic orbital wave functions (with phases) used to construct some of the MOs of a homonuclear diatomic molecule. For each sketch, determine the type of MO that will result from mixing the atomic orbital wave functions as drawn. Use the same labels for the MOs as in the “Closer Look” box on phases.
9.104 Write the electron configuration for the first excited state for N2—that is, the state with the highest-energy electron moved to the next available energy level. (a) Is the nitrogen in its first excited state diamagnetic or paramagnetic? (b) Is the N—N bond strength in the first excited state stronger or weaker compared to that in the ground state? Explain.
9.105 Azo dyes are organic dyes that are used for many applications, such as the coloring of fabrics. Many azo dyes are derivatives of the organic substance azobenzene, C12H10N2. A closely related substance is hydrazobenzene, C12H12N2. The Lewis structures of these two substances are
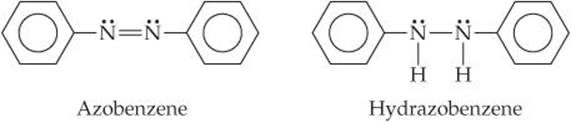
(Recall the shorthand notation used for benzene.) (a) What is the hybridization at the N atom in each of the substances? (b) How many unhybridized atomic orbitals are there on the N and the C atoms in each of the substances? (c) Predict the N—N—C angles in each of the substances. (d) Azobenzene is said to have greater delocalization of its π electrons than hydrazobenzene. Discuss this statement in light of your answers to (a) and (b). (e) All the atoms of azobenzene lie in one plane, whereas those of hydrazobenzene do not. Is this observation consistent with the statement in part (d)? (f) Azobenzene is an intense red-orange color, whereas hydrazobenzene is nearly colorless. Which molecule would be a better one to use in a solar energy conversion device? (See the “Chemistry Put to Work” box for more information about solar cells.)
[9.106] (a) Using only the valence atomic orbitals of a hydrogen atom and a fluorine atom, and following the model of Figure 9.46, how many MOs would you expect for the HF molecule? (b) How many of the MOs from part (a) would be occupied by electrons? (c) It turns out that the difference in energies between the valence atomic orbitals of H and F are sufficiently different that we can neglect the interaction of the 1s orbital of hydrogen with the 2s orbital of fluorine. The 1s orbital of hydrogen will mix only with one 2p orbital of fluorine. Draw pictures showing the proper orientation of all three 2p orbitals on F interacting with a 1s orbital on H. Which of the 2p or-bitals can actually make a bond with a 1s orbital, assuming that the atoms lie on the z-axis? (d) In the most accepted picture of HF, all the other atomic orbitals on fluorine move over at the same energy into the molecular orbital energy-level diagram for HF. These are called “nonbonding orbitals.” Sketch the energy-level diagram for HF using this information and calculate the bond order. (Nonbonding electrons do not contribute to bond order.) (e) Look at the Lewis structure for HF. Where are the nonbonding electrons?
[9.107] Carbon monoxide, CO, is isoelectronic to N2. (a) Draw a Lewis structure for CO that satisfies the octet rule. (b) Assume that the diagram in Figure 9.46 can be used to describe the MOs of CO. What is the predicted bond order for CO? Is this answer in accord with the Lewis structure you drew in part (a)? (c) Experimentally, it is found that the highest-energy electrons in CO reside in a σ-type MO. Is that observation consistent with Figure 9.46? If not, what modification needs to be made to the diagram? How does this modification relate to Figure 9.43? (d) Would you expect the π2p MOs of CO to have equal atomic orbital contributions from the C and O atoms? If not, which atom would have the greater contribution?
9.108 The energy-level diagram in Figure 9.36 shows that the sideways overlap of a pair of p orbitals produces two molecular orbitals, one bonding and one antibonding. In ethylene there is a pair of electrons in the bonding π orbital between the two carbons. Absorption of a photon of the appropriate wavelength can result in promotion of one of the bonding electrons from the π2p to the ![]() molecular orbital. (a) What would you expect this electronic transition to do to the carbon–carbon bond order in ethylene? (b) How does this relate to the fact that absorption of a photon of appropriate wavelength can cause ready rotation about the carbon–carbon bond, as described in the “Chemistry and Life” box and shown in Figure 9.30?
molecular orbital. (a) What would you expect this electronic transition to do to the carbon–carbon bond order in ethylene? (b) How does this relate to the fact that absorption of a photon of appropriate wavelength can cause ready rotation about the carbon–carbon bond, as described in the “Chemistry and Life” box and shown in Figure 9.30?
INTEGRATIVE EXERCISES
9.109 A compound composed of 2.1% H, 29.8% N, and 68.1% O has a molar mass of approximately 50 g/mol. (a) What is the molecular formula of the compound? (b) What is its Lewis structure if H is bonded to O? (c) What is the geometry of the molecule? (d) What is the hybridization of the orbitals around the N atom? (e) How many σ and how many π bonds are there in the molecule?
9.110 Sulfur tetrafluoride (SF4) reacts slowly with O2 to form sulfur tetrafluoride monoxide (OSF4) according to the following unbalanced reaction:
![]()
The O atom and the four F atoms in OSF4 are bonded to a central S atom. (a) Balance the equation. (b) Write a Lewis structure of OSF4 in which the formal charges of all atoms are zero. (c) Use average bond enthalpies (Table 8.4) to estimate the enthalpy of the reaction. Is it endothermic or exothermic? (d) Determine the electron-domain geometry of OSF4, and write two possible molecular geometries for the molecule based on this electron-domain geometry. (e) Which of the molecular geometries in part (d) is more likely to be observed for the molecule? Explain.
[9.111] The phosphorus trihalides (PX3) show the following variation in the bond angle X—P—X: PF3, 96.3°; PCl3, 100.3°; PBr3, 101.0°; PI3, 102.0°. The trend is generally attributed to the change in the electronegativity of the halogen. (a) Assuming that all electron domains are the same size, what value of the X — P — X angle is predicted by the VSEPR model? (b) What is the general trend in the X — P — X angle as the halide electronegativity increases? (c) Using the VSEPR model, explain the observed trend in X — P — X angle as the electronegativity of X changes. (d) Based on your answer to part (c), predict the structure of PBrCl4.
[9.112] The molecule 2-butene, C4H8, can undergo a geometric change called cis-trans isomerization:
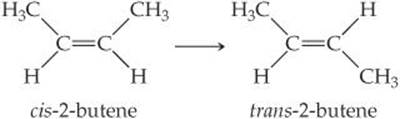
As discussed in the “Chemistry and Life” box on the chemistry of vision, such transformations can be induced by light and are the key to human vision. (a) What is the hybridization at the two central carbon atoms of 2-butene? (b) The isomerization occurs by rotation about the central C—C bond. With reference to Figure 9.30, explain why the π bond between the two central carbon atoms is destroyed halfway through the rotation from cis- to trans-2-butene. (c) Based on average bond enthalpies (Table 8.4), how much energy per molecule must be supplied to break the C — Cπ bond? (d) What is the longest wavelength of light that will provide photons of sufficient energy to break the C — Cπ bond and cause the isomerization? (e) Is the wavelength in your answer to part (d) in the visible portion of the electromagnetic spectrum? Comment on the importance of this result for human vision.
9.113 (a) Compare the bond enthalpies (Table 8.4) of the carbon-carbon single, double, and triple bonds to deduce an average π-bond contribution to the enthalpy. What fraction of a single bond does this quantity represent? (b) Make a similar comparison of nitrogen-nitrogen bonds. What do you observe? (c) Write Lewis structures of N2H4, N2H2, and N2, and determine the hybridization around nitrogen in each case. (d) Propose a reason for the large difference in your observations of parts (a) and (b).
9.114 Use average bond enthalpies (Table 8.4) to estimate ΔH for the atomization of benzene, C6H6:
![]()
Compare the value to that obtained by using ![]() data given in Appendix C and Hess's law. To what do you attribute the large discrepancy in the two values?
data given in Appendix C and Hess's law. To what do you attribute the large discrepancy in the two values?
[9.115] Many compounds of the transition-metal elements contain direct bonds between metal atoms. We will assume that the z-axis is defined as the metal-metal bond axis. (a) Which of the 3d orbitals (Figure 6.23) can be used to make a s bond between metal atoms? (b) Sketch the σ3dbonding and ![]() antibonding MOs. (c) With reference to the “Closer Look” box on the phases of orbitals, explain why a node is generated in the
antibonding MOs. (c) With reference to the “Closer Look” box on the phases of orbitals, explain why a node is generated in the ![]() MO. (d) Sketch the energy-level diagram for the Sc2 molecule, assuming that only the 3d orbital from part (a) is important. (e)What is the bond order in Sc2?
MO. (d) Sketch the energy-level diagram for the Sc2 molecule, assuming that only the 3d orbital from part (a) is important. (e)What is the bond order in Sc2?
[9.116] The organic molecules shown here are derivatives of benzene in which six-membered rings are “fused” at the edges of the hexagons.

(a) Determine the empirical formula of benzene and of these three compounds. (b) Suppose you are given a sample of one of the compounds. Could combustion analysis be used to determine unambiguously which of the three it is? (c) Naphthalene, the active ingredient in mothballs, is a white solid. Write a balanced equation for the combustion of naphthalene to CO2(g) and H2O(g). (d) Using the Lewis structure for naphthalene and the average bond enthalpies in Table 8.4, estimate the heat of combustion of naphthalene in kJ/mol. (e) Would you expect naphthalene, anthracene, and tetracene to have multiple resonance structures? If so, draw the additional resonance structures for naphthalene. (f) Benzene, naphthalene, and anthracene are colorless, but tetracene is orange. What does this imply about the relative HOMO–LUMO energy gaps in these molecules? See the “Chemistry Put to Work” box on orbitals and energy.
[9.117] Antibonding molecular orbitals can be used to make bonds to other atoms in a molecule. For example, metal atoms can use appropriate d orbitals to overlap with the ![]() orbitals of the carbon monoxide molecule. This is called d–π backbonding. (a) Draw a coordinate axis system in which the y-axis is vertical in the plane of the paper and the x-axis horizontal. Write “M” at the origin to denote a metal atom. (b) Now, on the x-axis to the right of M, draw the Lewis structure of a CO molecule, with the carbon nearest the M. The CO bond axis should be on the x-axis. (c) Draw the CO
orbitals of the carbon monoxide molecule. This is called d–π backbonding. (a) Draw a coordinate axis system in which the y-axis is vertical in the plane of the paper and the x-axis horizontal. Write “M” at the origin to denote a metal atom. (b) Now, on the x-axis to the right of M, draw the Lewis structure of a CO molecule, with the carbon nearest the M. The CO bond axis should be on the x-axis. (c) Draw the CO ![]() orbital, with phases (see the “Closer Look” box on phases) in the plane of the paper. Two lobes should be pointing toward M. (d) Now draw the dxy orbital of M, with phases. Can you see how they will overlap with the
orbital, with phases (see the “Closer Look” box on phases) in the plane of the paper. Two lobes should be pointing toward M. (d) Now draw the dxy orbital of M, with phases. Can you see how they will overlap with the ![]() orbital of CO? (e) What kind of bond is being made with the orbitals between M and C, σ or π? (f) Predict what will happen to the strength of the CO bond in a metal–CO complex compared to CO alone.
orbital of CO? (e) What kind of bond is being made with the orbitals between M and C, σ or π? (f) Predict what will happen to the strength of the CO bond in a metal–CO complex compared to CO alone.
9.118 Methyl isocyanate, CH3NCO, was made infamous in 1984 when an accidental leakage of this compound from a storage tank in Bhopal, India, resulted in the deaths of about 3,800 people and severe and lasting injury to many thousands more. (a) Draw a Lewis structure for methyl isocyanate. (b) Draw a ball-and-stick model of the structure, including estimates of all the bond angles in the compound. (c) Predict all the bond distances in the molecule. (d) Do you predict that the molecule will have a dipole moment? Explain.