MCAT General Chemistry Review
Chapter 9: Solutions
Practice Questions
1. An aqueous solution was prepared by mixing 70 g of an unknown nondissociating solute into 100 g of water. The solution has a boiling point of 101.11°C. What is the molar mass of the solute?
(Note:)
![]()
1. ![]()
2. ![]()
3. ![]()
4. ![]()
2. Which phases of solvent and solute can form a solution?
1. Solid solvent, gaseous solute
2. Solid solvent, solid solute
3. Gaseous solvent, gaseous solute
1. I and II only
2. I and III only
3. II and III only
4. I, II, and III
3. Two organic liquids, pictured in the figure below, are combined to form a solution. Based on their structures, will the solution closely obey Raoult’s law?
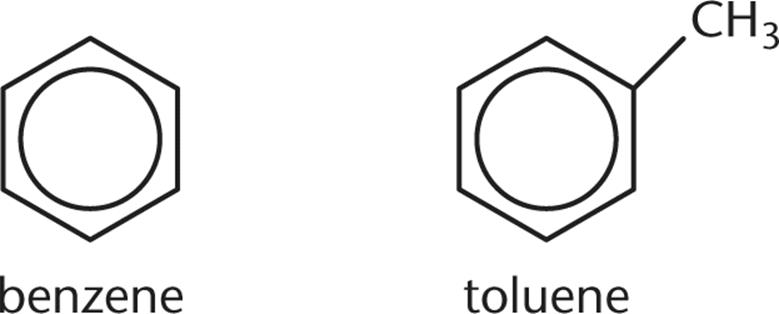
1. Yes; the liquids differ due to the additional methyl group on toluene and, therefore, will not deviate from Raoult’s law.
2. Yes; the liquids are very similar and, therefore, will not deviate from Raoult’s law.
3. No; the liquids differ due to the additional methyl group on toluene and, therefore, will deviate from Raoult’s law.
4. No; the liquids both contain benzene rings, which will interact with each other and cause deviation from Raoult’s law.
4. Which of the following explanations best describes the mechanism by which solute particles affect the melting point of ice?
1. Melting point is elevated because the kinetic energy of the substance increases.
2. Melting point is elevated because the kinetic energy of the substance decreases.
3. Melting point is depressed because solute particles interfere with lattice formation.
4. Melting point is depressed because solute particles enhance lattice formation.
5. The process of formation of a liquid solution can be better understood by breaking the process into three steps:
1. Breaking the solute into its individual components
2. Making room for the solute in the solvent by overcoming intermolecular forces in the solvent
3. Allowing solute–solvent interactions to occur to form the solution
Which of the following correctly lists the enthalpy changes for these three steps, respectively?
1. Endothermic, exothermic, endothermic
2. Exothermic, endothermic, endothermic
3. Exothermic, exothermic, endothermic
4. Endothermic, endothermic, exothermic
6. The entropy change when a solution forms can be expressed by the term ΔS°soln. When an ion dissolves and water molecules are ordered around it, the ordering would be expected to make a negative contribution to ΔS°soln. An ion that has more charge density will have a greater hydration effect, or ordering of water molecules. Based on this information, which of the following compounds will have the most negative ΔS°soln?
1. KCl
2. LiF
3. CaS
4. NaCl
7. When ammonia, NH3, is used as a solvent, it can form complex ions. For example, dissolving AgCl in NH3 will result in the complex ion [Ag(NH3)]2+. What effect would the formation of complex ions to have on the solubility of a compound like AgCl in NH3?
1. The solubility of AgCl will increase because complex ion formation will cause more ions to exist in solution, which interact with AgCl to cause it to dissociate.
2. The solubility of AgCl will increase because complex ion formation will consume Ag+ ions and cause the equilibrium to shift away from solid AgCl.
3. The solubility of AgCl will decrease because Ag+ ions are in complexes, and the Ag+ ions that are not complexed will want to associate with Cl− to form solid AgCl.
4. The solubility of AgCl will decrease because complex ion formation will consume Ag+ ions and cause the equilibrium to shift toward the solid AgCl.
8. One hundred grams of sucrose are dissolved in a cup of hot water at 80°C. The cup of water contains 300.00 mL of water. What is the percent composition by mass of sugar in the resulting solution?
(Note: Sucrose = C12H22O11, density of water at ![]() )
)
1. 25.0%
2. 25.5%
3. 33.3%
4. 34.2%
9. Which of the following combinations of liquids would be expected to have a vapor pressure higher than the vapor pressure that would be predicted by Raoult’s law?
1. Ethanol and hexane
2. Acetone and water
3. Isopropanol and methanol
4. Nitric acid and water
10.The salt KCl is dissolved in a beaker. To an observer holding the beaker, the solution begins to feel colder as the KCl dissolves. From this observation, one could conclude that:
1. ΔS°soln is large enough to overcome the unfavorable ΔH°soln.
2. KCl is mostly insoluble in water.
3. ΔS°soln must be negative when KCl dissolves.
4. boiling point depression will occur in this solution.
11.Which of the following will cause the greatest increase in the boiling point of water when it is dissolved in 1.00 kg H2O?
1. 0.46 mol calcium sulfate
2. 0.54 mol iron(III) nitrate
3. 1.09 mol acetic acid
4. 1.11 mol sucrose
12.At sea level and 25°C, the solubility of oxygen gas in water is 1.25 × 10−3 M. In Denver, a city in the United States that lies high above sea level, the atmospheric pressure is 0.800 atm. What is the solubility of oxygen in water in Denver?
1. 1.00 × 10−3 M
2. 1.05 × 10−3 M
3. 1.50 × 10−3 M
4. 2.56 × 10−3 M
13.Lead is a toxic element that can cause many symptoms, including mental retardation in children. If a body of water is polluted with lead ions at 30 ppb (parts per billion), what is the concentration of lead expressed as molarity? (Note: The density of water is ![]() and ppb = grams per 109 grams of solution)
and ppb = grams per 109 grams of solution)
1. 1.4 × 10−10 M Pb2+
2. 1.4 × 10−7 M Pb2+
3. 6.2 × 10−7 M Pb2+
4. 6.2 × 10−6 M Pb2+
14.A saturated solution of cobalt(III) hydroxide (Ksp = 1.6 × 10−44) is added to a saturated solution of thallium(III) hydroxide (Ksp = 6.3 × 10−46). What is likely to occur?
1. Both cobalt(III) hydroxide and thallium(III) hydroxide remain stable in solution.
2. Some cobalt(III) hydroxide precipitates and thallium(III) hydroxide remains stable in solution.
3. Some thallium(III) hydroxide precipitates and cobalt(III) hydroxide remains stable in solution.
4. Some of both cobalt(III) hydroxide and thallium(III) hydroxide precipitate.
15.The following equilibrium exists when AgBr (Ksp = 7.7 × 10−13) is in solution:
AgBr (s) ⇋ Ag+ (aq) + Br− (aq)
What is the solubility of AgBr in a solution of 0.0010 M NaBr?
1. ![]()
2. ![]()
3. ![]()
4. ![]()
PRACTICE QUESTIONS