Microreactors in Organic Chemistry and Catalysis, Second Edition (2013)
11. Industrial Microreactor Process Development up to Production
11.4. Pilot Plants and Production
11.4.1 Hydrogen Peroxide Synthesis
![]()
The process developing Company UOP LLC in Des Plaines, IL, USA, searched for direct routes for the manufacture of hydrogen peroxide from elementary hydrogen and oxygen by microprocess technology that almost under all process conditions inevitably involves processing in the explosive area [50, 51] (Figure 11.11). Advantages of the direct over the indirect routes are better use of raw materials and less technical expenditure owing to process simplification. The few process windows out of the explosive range need large dilution and/or multistep processing with addition of intermediates as carriers for oxygen, such as the anthraquinone route.
Figure 11.11 Hydrogen peroxide synthesis pilot microprocess plant. Source: Courtesy of FMC Corporation, Princeton, NJ, USA, and Niyi Lawal, Stevens Institute [55].

On the contrary, operation in the explosive area (“New Process Windows”) benefits from higher space–time yield, since elevated temperatures, pressures, and reactant concentrations are utilized [50, 51]. This is enabled by the higher safety potential of microreactors, which are less accident-prone by thermal runaway due to enhanced heat transfer and also do not allow uncontrolled, ever-increasing radical chain propagation by providing an even high rate of radical quenching at the large surface area of the channel walls [52]. Due to the latter flame-arrestor effect that is intrinsic to the devices, the term “inherently safe” was assigned. However, this only refers to the device inner volume and is a theoretical term (e.g., walls between microchannels may break under very high pressure shock waves or other similar incidents, so a dimensional increase above the safety threshold may occur). Still, a practical safety evaluation needs to be done and safety issues are different in the interconnections and fluid distributions to the device and this holds even more in the plant peripherals. Thus, while the intrinsic safety of the microchannel on a device level in a theoretical manner can be assumed, that of the microprocess technology as a whole has to be verified and authorized for each chemical process. Even though, advantages remain due to faster safety approval.
Based on this information, a process flow was developed based on a mini-trickle bed operation where a premixed gas mixture of hydrogen and oxygen (without inert gas) was dispersed with an aqueous solution flowing through the interstices of a catalyst bed [50, 51]. Experimental investigations were first done at a laboratory level using slit-type high-pressure interdigital micromixer connected to a cartridge filled with catalyst. Special procedures and plant details were developed and added to have safety on a process level and to encounter typical failure situations such as total oxidation taking place outside the catalyst cartridge with water as heat absorbing medium.
The direct contact of hydrogen and oxygen offers other advantages besides having high concentrations and high reaction rates [50, 51]. Explosive regime mixing allows one to generate hydrogen/oxygen mixtures in a stable and reliable manner. This will transfer the noble metal catalyst into a partially oxidized state that then will generate hydrogen peroxide with high selectivity. Using the fully oxidized or fully reduced state of the catalyst, either only water is formed or no reaction takes place, respectively.
Operation of this process at only 20 bar and the use of oxygen/hydrogen ratios close to 1 : 1 were considerably improved [50, 51] compared to processes from the patent literature [53, 54]. For oxygen/hydrogen ratios of 1.5–3, a selectivity as high as 85% at 90% conversion and a space–time yield of 2 g hydrogen peroxide per (g catalyst h) were achieved, exceeding industrial benchmarks from the patent literature at that point of time. These laboratory experiments were followed by pilot tests that resulted in a basic engineering design for the production of about 150 000 t hydrogen peroxide per year.
FMC Corporation in Princeton, NJ, USA, one of the largest producers of hydrogen peroxide, works with Stevens Institute in Hoboken, NJ, USA, on a publicly funded project for the on-demand production of hydrogen peroxide in a microreactor at end-user sites [4, 55]. While today's hydrogen peroxide is mainly produced as a concentrated solution (70% in the anthraquinone process), many user applications demand solutions <15%, so dilution is common and this overrides the efforts made by the energy-consumptive distillation to yield concentrated solutions. FMC summarizes the prospects of a direct microreactor route for on-site production to reduce transportation and storage costs as well as “concentration–dilution” costs. Operating safely in the explosive regime is the target by designing a low-pressure and energy-efficient process. Solvent processing, gas recycling and treatment, and product purification are avoided as compared to the commercial anthraquinone process. The initial goal is to achieve the production of a 1% solution at laboratory scale and to transfer this to an on-site production scale within 5 years.
Hydrogen peroxide was generated in a microreactor in a gas–liquid–solid process with direct mixing of hydrogen and oxygen (using air), a liquid solvent, and a solid catalyst [55], which included operation in the explosive regime. The particles of the self-developed platinum group catalyst supported on an oxide were packed in the microchannel. In addition, sol–gel wall coatings were adhered to the microchannels and cellular structures were formed by different deposition routes involving closed-channel flow coating, open-channel surface-selective, and dip coating methods. Using moderate pressure and temperature, the residence time could be decreased by almost two orders of magnitude as compared to conventional reactors. The hydrogen peroxide concentrations were within the industrial relevant range. Based on the identification of the elemental reactions involved, a study of the reaction mechanism was done to determine the kinetics. Following the laboratory studies, an optimized process analysis, design, and simulation was developed for a pilot plant comprising a multichannel, multilayered microreactor system built by Chart Energy and Chemicals in The Woodlands, TX, USA. First runs proved the production of hydrogen peroxide in an industrial relevant range.
Recently, a new microreactor for the direct synthesis of hydrogen peroxide has been designed at the AIST, Tsukuba, and Mitsubishi Gas Chemical Inc., Tokyo. It operates at room temperature and 1 MPa. The reactor is made of glass and the catalyst is palladium on titania. The yield of 40% was reached, based on hydrogen, and the concentration was 10% [56].
As the potential of this technology is tremendous, companies are putting their effort in investigating the reaction kinetics and stability of hydrogen peroxide formation in a microreactor. Some such studies are performed at BASF Catalyst [57] and give a promising input for optimizing the reaction for more efficient industrial synthesis.
11.4.2 Phenylboronic Acid Synthesis
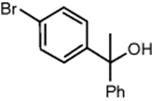
Clariant GmbH in Frankfurt, Germany, performed the synthesis of phenylboronic acid from phenylmagnesium bromide and boronic acid trimethyl ester on a pilot-scale level [58].

This reaction suffers, as many organometallic reactions, from insufficient mixing, since its reaction speed (at room temperature and below) is faster than the mixing times of many conventional mixer equipments. Thus, the reaction proceeds under nonstochiometric conditions with timely and spatially changing concentration profiles, which promotes consecutive reactions. In addition, too long processing times, quite typical for batch reactions, favor side reactions such as oxidations and hydrolysis. This is solved in conventional processing by slowing down the reaction speed by virtue of cryogenic conditions, which reduces the impact of mixing and expands the mixing timescale so that selectivity is sufficient. For this, cooling utilities need to be installed, which adds to the capital investment and makes the process energy consumptive.
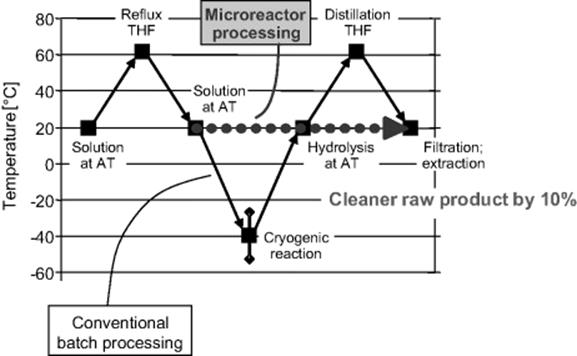
For the microreactor process, a micromixer–tube rig was used, with an interdigital mixer as the laboratory tool and a caterpillar mixer as the pilot tool [58]. The superior mixing of micromixers allows one to perform the reaction at much higher reaction speed, so a high selectivity of 90% could be demonstrated even at room temperature and above. This saves energy costs and reduces the respective CAPEX (capital-related) investments. The crude yield was about 25% higher than that for the industrial batch production. The purity of the crude product was increased by about 10%. This had an impact downstream the reaction processing. Purification was simplified and could be achieved by means of crystallization only, whereas in the current industrial process energy-consumptive distillation is required (see Figure 11.12).
Figure 11.12 Temperature profile of the phenylboronic acid synthesis along the major steps of the process flow scheme (AT, ambient temperature; THF, tetrahydrofuran). The difference in temperatures of the conventional batch and the microreactor processes is due to the reduction in energy consumption and respective heat transfer equipment when using the latter. Source: Courtesy of ACS [58].
11.4.3 Diverse Case Studies at Lonza
As microreaction technology becomes widely accepted, fine chemical and pharmaceutical companies frequently report the use of at least one continuous-flow step in their manufacturing campaigns and laboratory synthesis. Lonza Ltd in Visp, Switzerland, has developed at least 10 processes for continuous-flow production with micro- and other flow reactors. For an organolithium exchange reaction, up to 250 kg of product was obtained in a few weeks of operations using a Corning multi-injection reactor [59–62]. At the end of the campaign, the reactor was cleaned by standard procedure and used again for a new project. For an organolithium coupling reaction, several kilograms of product was obtained by continuous operation for 1 week. The Corning multi-injection reactor was used as well as the Corning single-injection reactor for validation [59]. An analysis of chemical production requirements in an exclusive synthesis market environment was performed and guided the development of this modular multi-injection reactor [59–62]. Other organometallic reactions were studied as well, with small modifications in the reactor setup. The reactor was then operated without interruption for seven consecutive days/24 hours per day producing several kilograms of product. The two production examples demonstrate easy cleaning, robustness, and modularity of the Corning microreactor and make it a valuable industrial reactor for the production of pharmaceuticals.
Using dedicated reactor technology, chlorination was carried out in a falling film reactor, which is a highly exothermic reaction (ΔTadiabatic > 250 °C) [46]. Dehydrogenations were done using a heterogeneous catalyst filled in a tubular packed bed at high temperatures above 500 °C. An organolithium coupling reaction was carried out in a small CSTR reactor to avoid reactor plugging.
The so-called continuous launch production unit R-01 was built with a capacity in the range of 150 kg/h and designed as a multipurpose system (see Figure 11.13) [46]. Conventional technology, such as static mixers, mini-heat exchangers, and so on, was used following experience with microprocess technology (Figure 11.14). Such devices are the mesoscale extension of microdevices, albeit not always using the same mixing and heat exchange principles. Several campaigns were performed with in-between cleaning for the following reactions. Tons of product was made by using the Simmons–Smith reaction and the production ran over weeks. A static mixer with conventional mini-tube heat exchangers was used. A similar amount of material was produced by an organolithium coupling reaction using a static mixer in an adiabatic regime.
Figure 11.13 Continuous launch production unit R-01 at Lonza. Source: Courtesy of D. Roberge/Lonza [46].

Figure 11.14 Lonza continuous small-scale plant with combined microreactor and conventional technologies (static mixers): process view (a) and service view (b). Source: Courtesy of PharmaChem/B5Srl [64].

Harsh reaction conditions in continuous-flow microreactors are also investigated on several Lonza's examples, including nitrations, organolithium and Grignard reactions, and DIBAL-H reduction [63]. Enhanced heat transfer for temperature control, good mixing properties including controlled backmixing, low reactor volume, and very rapid radical propagation in a confined volume enabled higher yields of nitro compounds when nitrations are performed in microreactors. A multi-injection principle was applied in a Grignard reaction, showing that a good thermal control in the microreactor can be obtained with increasing number of injection points. DIBAL-H reduction is performed at −60 °C in batch systems. Good mixing and heat transfer in microreactors allowed good selectivities at temperatures as high as −20 °C.
11.4.4 Alkylation Reactions Based on Butyllithium
Lonza Ltd in Visp, Switzerland, developed at the beginning of the 1990s a process for the halogen exchange of a brominated aromatic compound with butyllithium followed by the CߝC coupling to a ketone [64].
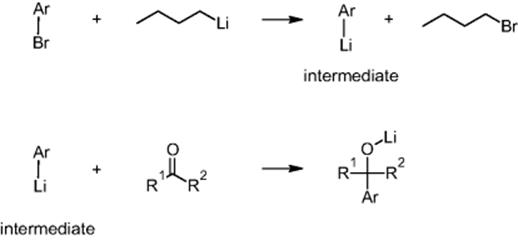
The lithium intermediate is unstable, even at temperatures as low as −60 °C. Only a continuous process with a short residence time between the two reactions allowed one to avoid decomposition and have sufficient selectivity for an economical process [64]. Clogging is a major issue for the first process. Due to heat release issues, high dilution is applied and the recycling of the solvent was an important issue that needed to be considered.
The first reaction is highly exothermic with an adiabatic temperature rise >100 °C (classified as Type A in Ref. [65]), while the second reaction is only slightly exothermic with an adiabatic temperature rise <30 °C (this follows a classification given in Ref. [65]: Type A and Type B, respectively) [44]. Despite the latter, the second reaction is accompanied by the formation of large amounts of side products at long residence times >1 min, which almost excludes batch processing here. A conventional continuous system, a static mixer with much shorter residence times, was successfully used. The first reaction can be performed batchwise, since the lithium intermediate is stable for a few hours. Still, a stop of production for several hours would lead to a significant loss of product.
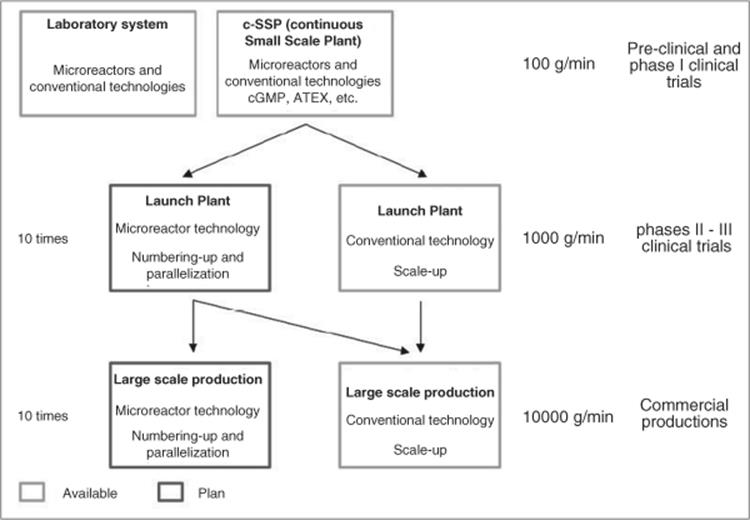
Thus, it is more flexible to run both reactions continuously [64]. A microreactor is used for the more demanding, highly exothermic reaction, whereas a static mixer is sufficient for the second reaction. After laboratory process development, a pilot phase in the so-called continuous small-scale production (c-SSP) comprising microreactor technology followed (see Figure 11.16). The c-SSP plant is a multipurpose and modular approach and can operate from cryogenic to high temperature.
The c-SSP plant typically operates at a total flow rate of 100 g/min that relates to 70 kg product/week of product [64]. This gives sufficient material for preclinical studies or phase I clinical trials during a typical pilot campaign cycle time, which is 1–2 weeks (see Figure 11.17). The c-SSP plant is qualified for c-GMP (GMP: good manufacturing practice; guidelines for quality management for production processes and environment in the fields of pharmaceutical and medicinal products, but also in food and animal feed industry) and meets the ATEX (“Atmosphère Explosible”; stands for two guidelines given by European Union in the field of explosion prevention) standards. The microreactor for cryogenic operation is insulated in the black box unit, shown in the upper image of Figure 11.15. The alkyl lithium process was performed at multiple kilogram scale, was safe, and led to an increase in yield.
Methodologies for scale-out to phase II–III clinical trials are also given in Figure 11.15 [64]. One approach would be to scale-up the c-SSP using static mixers and mini-heat exchangers, which are already in operation at Lonza. The possible flow rate of 1500 g/min is 10–15 times higher than that of the c-SSP. Another approach is the replication of the laboratory system in a few number of identical base units (numbering up). A maximum of 10 base units are estimated for sufficient throughput to supply phase II–III clinical trials or quantities in the range of a few tons. The technical transfer would be more rapid than done conventionally and scale-up issues would be avoided especially with exothermic reactions.
Figure 11.15 Scheme with typical process steps in the fine chemical and pharmaceutical industries and recommendation when to use microreactor technology or continuous processes based on a multipurpose approach. Source: Courtesy of PharmaChem/B5Srl [64].
For commercial production, other issues arise [64, 66]. Among these, the production cost is the key driver, as the last step in process development increasingly consumes large resources due to a number of additional tasks (scale-up, supply chain, waste management, ecology, location, etc.). The specific microreactor-related advantages such as speed in process R&D and avoidance of scale-up issues do not have the same importance here as in the clinical productions. Thus, microprocess technology is here in competition to other technologies and one advantage may not be enough, as a highly complex scenario has to be considered.
11.4.5 Microprocess Technology in Japan
The known industrial implementation of microprocess technology in Japan so far has been done in a consorted action [67]. The Ministry of Economy, Trade and Industry (METI) launched the first project cluster in 2002, entitled “High Efficiency Micro-Chemical Process Technology Project.” One year later, the focus of the associated works was more shifted to early industrial implementation and the project cluster name was changed to “Production, Analysis and Measurement System for Micro-Chemical Process Technologies.” The management of this project was done by the Association of Micro-Chemical Process Technology (MCPT), which is an umbrella for 30 chemical and instrument companies. The projects were organized in three groups covering different facets, which are the development of microchemical plant technology, the development of microchip technology, and systematization of microchemical process technology. In the Sections 11.4.6--11.4.14, developments done under the first issue will be reported with focus on the transfer of process developments into pilot plants. The researchers from 7 companies and 11 research groups at Kyoto University and 4 other universities were engaged in the latter action. The plan was to have 3-year fundamental development and then transfer it to pilot plants. Another project cluster was started in July 2006, named “Development of Microspace and Nanospace Reaction Environment Technology for Functional Materials.” This program was planned for 5 years and besides MCPT the National Institute of Advanced Industrial Science and Technology (AIST) was involved. Furthermore, seven universities carried out research, with focal points at Kyoto University and the Tsukuba Intensive Research Center.
11.4.6 Pilot Plant for Methyl Methacrylate Manufacture
Idemitsu Kosan in Chiba, Japan, operates a pilot plant for the free radical polymerization of methyl methacrylate (MMA) [68], following prior process development at Kyoto University [69].
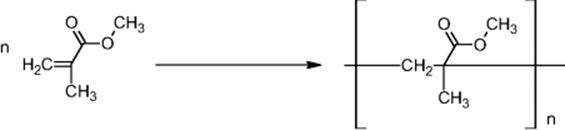
The plant has a capacity of 10 t/a (see Figure 11.16) [68]. Eight microreactor blocks form the reactor core and each comprises three tube reactors with micrometer-sized inner dimensions (500 μm internal diameter, 2 m length) in series. It could be shown that the numbering-up principle is valid; that is, relevant process figures such as the polydispersity index, yield, and average number-based molecular weight were similar for single and parallel tube operation.
Figure 11.16 Microprocess pilot plant for radical polymerization reaction. Source: Courtesy of ACS [68].

11.4.7 Grignard Exchange Reaction
Researchers at Kyoto University performed a few exchange reactions in microflow devices. One is a Grignard reaction of ethylmagnesium bromide and bromopentafluorobenzene to give pentafluorophenylmagnesium bromide [70]. Scale-out was done using small- and medium-scale microflow systems consisting of a micromixer and a microheat exchanger. The shell and tube microheat exchanger used for the medium-scale microflow systems could be operated at a high flow rate of 6 l/h. The heat exchanger consists of 55 microtubes (i.d. 490 μm × 200 mm) embedded in a shell (i.d. 16.7 mm × 200 mm). Water as coolant is circulated through the shell.
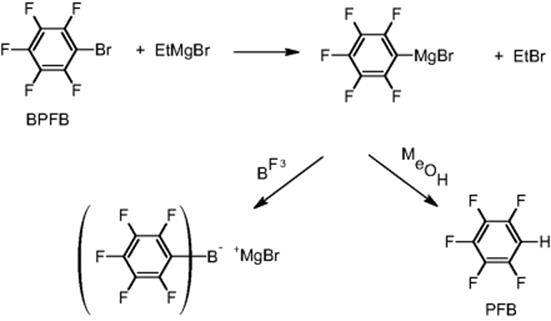
In a detailed process optimization study, the impact of the type of micromixers and process parameters was determined [70]. As a result, a pilot plant with a Toray Hi-mixer connected to a shell and tube microheat exchanger was constructed. Continuous operation for 24 h was carried out to obtain pentafluorobenzene (PFB) after protonation (92% yield). During this time, 14.7 kg of the product was produced, that is, about 5 t/a. Thus, the industrial-scale production carried out using a batch reactor (10 m3) can be replaced by adding only four microflow systems of the scale investigated. The pilot plant produces 0.5 kg in 6 h continuous operation, and thus about 730 kg/a (see Figure 11.17). The name of the industrial company was not disclosed.
Figure 11.17 Microprocess pilot plant for Grignard exchange reaction. Source: Courtesy of ACS [70].

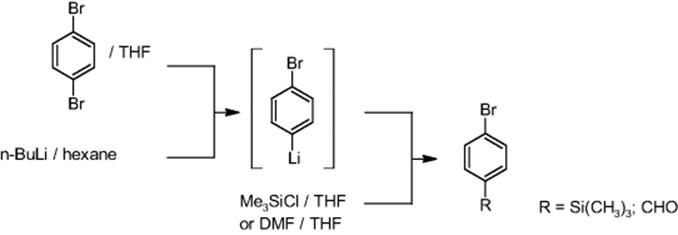
11.4.8 Halogen–Lithium Exchange Pilot Plant
The University of Kyoto, Japan, reports about a halogen–lithium exchange reaction of aryl bromides with butyllithium (see Figure 11.18) [67, 71, 72]. The intermediate was trapped with an electrophile. The whole process was performed under noncryogenic conditions at 0 °C (Table 11.4).
Figure 11.18 Halogen–lithium exchange reactions. Source: Courtesy of Wiley-VCH Verlag GmbH [71].
Table 11.4 Products synthesized in the microflow system from Ref. [72]
|
Electrophile |
Product |
Yield (%) |
|
MeI |
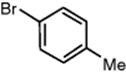
|
87 |
|
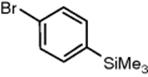
Me3SiCl |
|
85 |
|
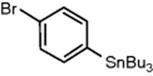
Bu3SnCl |
|
86 |
|
|
|
73 |
|
|
|
87 |
The pilot plant produces 0.5 kg in 6 h continuous operation, and thus about 730 kg/a [67, 71]. The name of the industrial company was not disclosed (see Figure 11.19).
Figure 11.19 Microprocess pilot plant for halogen–lithium exchange process. Source: Courtesy of Wiley-VCH Verlag GmbH [71].

Another example is a Br–Li exchange reaction of p-dibromobenzene with nBuLi [73]. The reaction is exothermic and requires very low temperature (< − 48 °C) in a batch system. With an integrated microflow system, reaction could be performed at 20 °C followed by reaction with an electrophile at 0 °C.
A variety of p-, m-, and o-disubstituted benzenes were synthesized in one flow at much higher temperatures than those required for conventional batch reactions. Industrial application of fine chemicals is envisaged after increasing productivity of this microflow system [72] (Figure 11.20).
Figure 11.20 The microflow system consisting of two micromixers and two microtube reactors [72].

11.4.9 Swern–Moffatt Oxidation Pilot Plant
Ube Industries Ltd in Yamaguchi, Japan, and Kyoto University investigated the Swern oxidation for pharmaceutical intermediates [71, 73]. In this reaction, alcohols are oxidized to carbonyl compounds using dimethyl sulfoxide. The reaction variant using dimethyl sulfoxide activated by trifluoroacetic anhydride (shown below) has found industrial application, but is limited to low-temperature operation (−50 °C or below) to avoid decomposition of an intermediate.
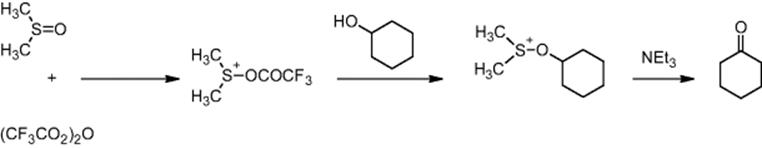
In a microscale tubular reactor, Swern oxidations were performed between 20 and 20 °C. Mixing was performed stagewise with a series of rapid mixing functions (see Figure 11.21) [71, 73]. First, dimethyl sulfoxide and trifluoroacetic anhydride were reacted in an interdigital micromixer followed by a stainless steel tube reactor R1. After addition of the alcohol and reaction in R2, the mixture was then contacted with a triethylamine solution and passed through two more reactors (R3 and R4) to complete the reaction.
Figure 11.21 Schematic diagram of the microscale flow system for the Swern oxidation. Source: Courtesy of Wiley-VCH Verlag GmbH [73].
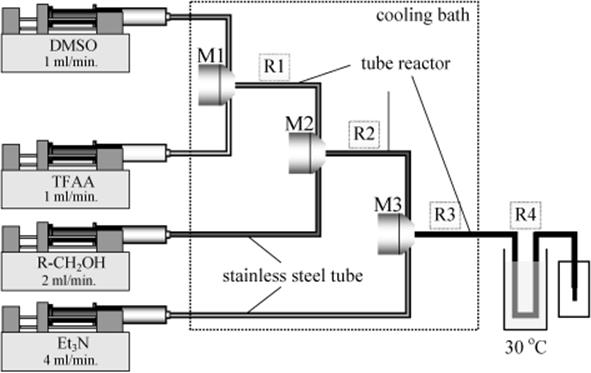
This microreactor system thus allows fast mixing, good temperature control, and changing process parameters at short residence times – the overall time to pass all reactors is between 8 and 11 s [71, 73]. The timescale between end and start of a new operation is 0.01 s in order to avoid significant decomposition. Thereby, conversions and yields were determined for primary, secondary, cyclic, and benzylic alcohols in the temperature range from −20 to 20 °C, which were equal to or better than those of lower temperature batch reactions. The oxidation of cyclohexanol was run for 3 h at 20 °C and a stable process in terms of conversion and selectivity was observed.
Then, a pilot plant with a capacity of 10 t/a was built for the Swern–Moffatt oxidation [71, 73]. The yield of the batch process is 83% at −70 °C, whereas the microchemical process achieved a yield of 88% at 20 °C. The pilot plant was operated under stable reaction conditions for a long run with similar product yields as in the laboratory experiment (see Figure 11.22).
Figure 11.22 Microprocess pilot plant for Swern–Moffatt reaction. Source: Courtesy of J.-I. Yoshida, University of Kyoto.

11.4.10 Yellow Nano Pigment Plant
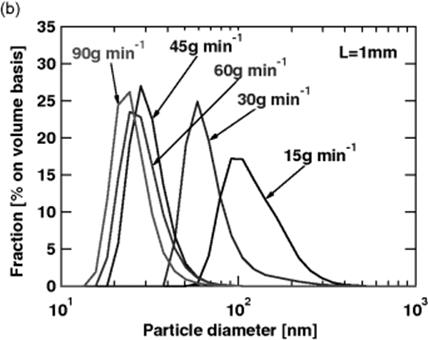
The University of Kyoto, Japan, and Fuji in Tokyo, Japan, developed a pilot plant for the production of a yellow nano pigment with a capacity of 70 t/a (see Figure 11.23) [74]. The particle size spectra show a clear impact of the flow conditions with smaller particles at higher flow rates.
Figure 11.23 (a) Yellow nano pigment pilot microprocess plant. (b) Particle size spectra at various volume flows. Source: Courtesy H. Maeta, Fuji [74].

11.4.11 Polycondensation
The MCPT research group investigated polycondensation reactions for the synthesis of highly heat-resistant polymers such as polyamide and polyimide [71]. These reactions are exothermic, which negatively impacts average molecular weights and the corresponding distributions when using conventional technology. A two-step reaction with polycondensation and terminal modification with NA was conducted in a microreactor as shown in the following scheme.
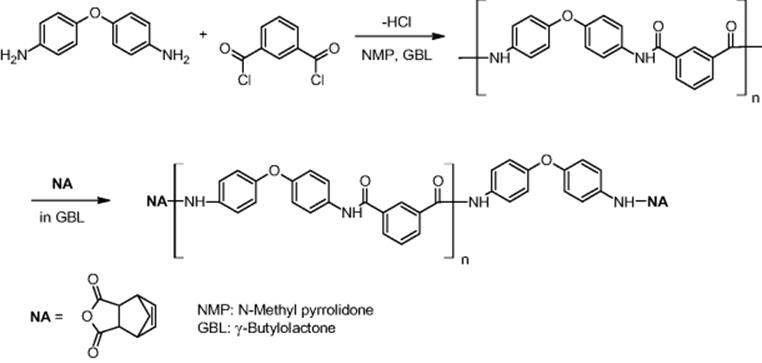
The reaction was faster in the microreactor than in the batch system, which is due to a mixing effect [71]. The molecular weight distribution obtained in the microreactor is slightly narrower than that of the batch reactor, which is attributed to the good thermal management of the microreactor.
11.4.12 H2O2-Based Oxidation to 2-Methyl-1,4-naphthoquinone
The MCPT research team investigated the synthesis of 2-methyl-1,4-naphthoquinone (Vitamin K3) by the oxidation of 2-methylnaphthalene [71].
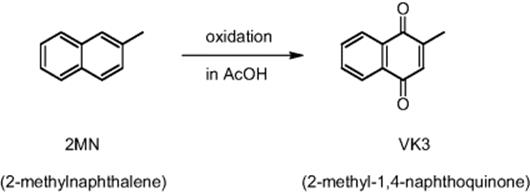
This is done in the commercial production with chromium trioxide and the aim was to test for possibility of substitution by using aqueous hydrogen peroxide (60%) as oxidant [71]. Hydrogen peroxide (H2O2) is a mild and environment-friendly oxidant. For better performance, the oxidation could be made faster by using higher temperatures and higher concentrations of the oxidant. This, however, is prohibited by side reactions such as overoxidation, side chain oxidation, and decomposition of the oxidant. Therefore, dropwise addition of hydrogen peroxide is common to achieve high selectivities, usually at low temperatures. This results in long reaction times.
Using a microreactor and a feed comprising also palladium acetate and sulfuric acid, the reaction time was shortened to 10 min at 70 °C as compared to the industrial semibatch process (see Figure 11.24) [71]. However, the selectivity was lower. This could be solved by process modification using peracetic acid, generated in situ from acetic acid and hydrogen peroxide.
Figure 11.24 Speeding up the oxidation of 2-methylnaphthalene (2MN) in a microreactor by virtue of faster hydrogen peroxide addition and higher temperatures. Source: Courtesy of Wiley-VCH Verlag GmbH [71].
11.4.13 Friedel–Crafts Alkylation
The MCPT research group investigated Friedel–Crafts alkylations, a widely used reaction in organic chemistry [71]. As an electron-donating substituent is introduced in the first reaction, the monoalkylated product is more reactive than the starting material and the second alkylation takes place more readily. This leads to by-products by dialkylation or even polyalkylation. This can be avoided by using a large excess of the starting material. However, a smart solution would be a selective monoalkylation using only one equivalent of an aromatic compound. The key to this may be micromixing, as respective impact on selectivity has been found in earlier investigations.
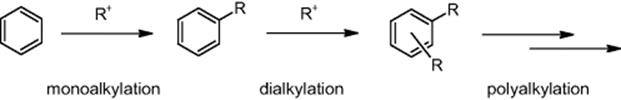
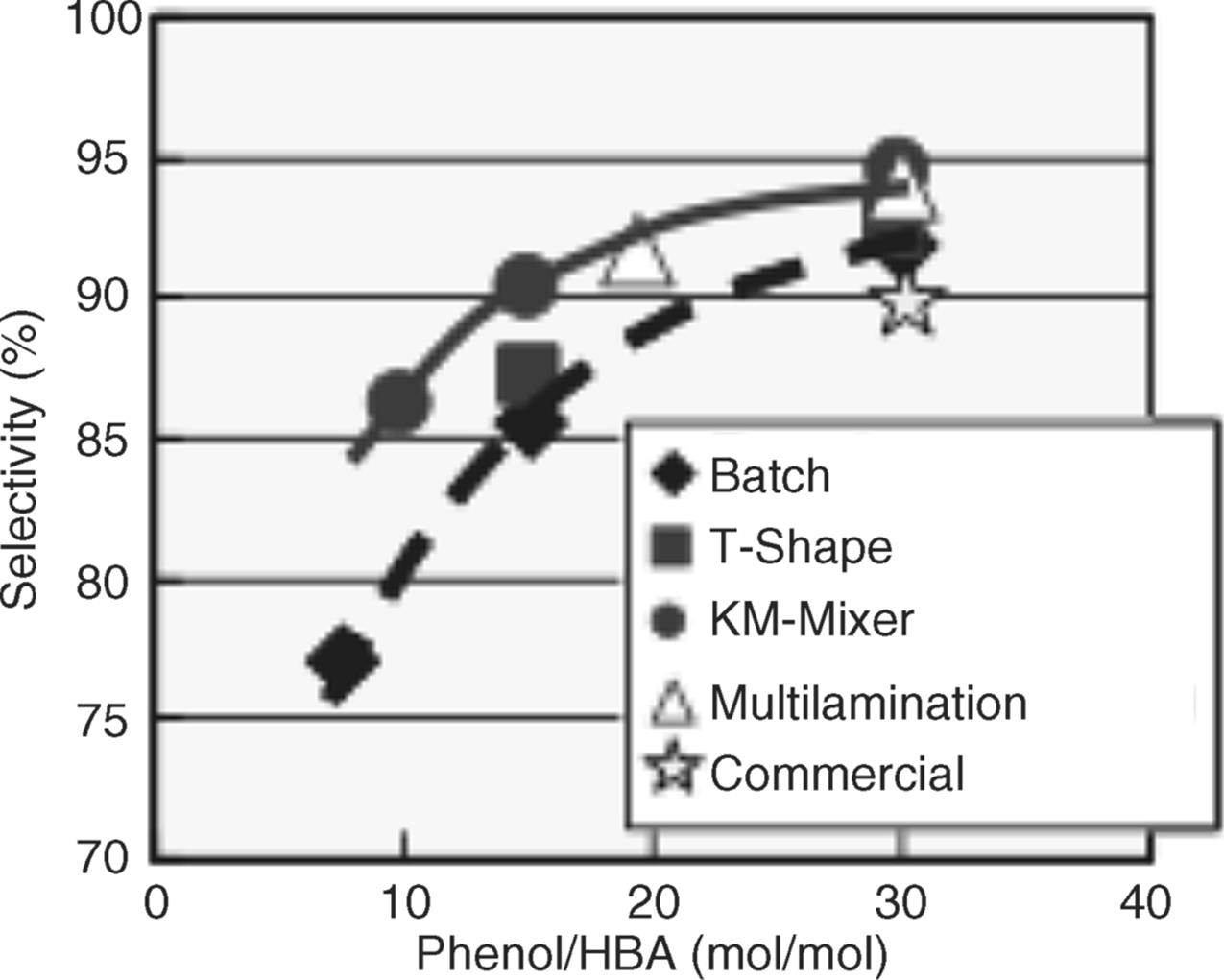
As industrially relevant Friedel–Crafts reaction, the synthesis of bisphenol F, a material for epoxy resin, from phenol and formaldehyde was chosen [71]. This reaction involves formation of higher order condensates such as trisphenols. To minimize the latter, the molar ratio of phenol to formaldehyde is set to a very high value (30–40), which is more than 15 times larger than the amount theoretically necessary. Three types of micromixers were used: a T-shaped mixer with 500 μm inner diameter, a multilaminating interdigital micromixer with 40 μm channels, and the so-called self-made K-M mixer with center collision mixing.
The selectivity increased with increasing phenol/hydroxybenzyl alcohol ratio (see Figure 11.25) [71]. Best performance was achieved by the K-M mixer. The phenol/hydroxybenzyl alcohol ratio was decreased to half in this way.
Figure 11.25 Impact of choice of micromixer on selectivity when varying the phenol/hydroxybenzyl alcohol molar ratio. Source: Courtesy of Wiley-VCH Verlag GmbH [71].
11.4.14 Diverse Studies from Japanese Project Cluster
11.4.14.1 Synthesis of Photochromic Diarylethenes
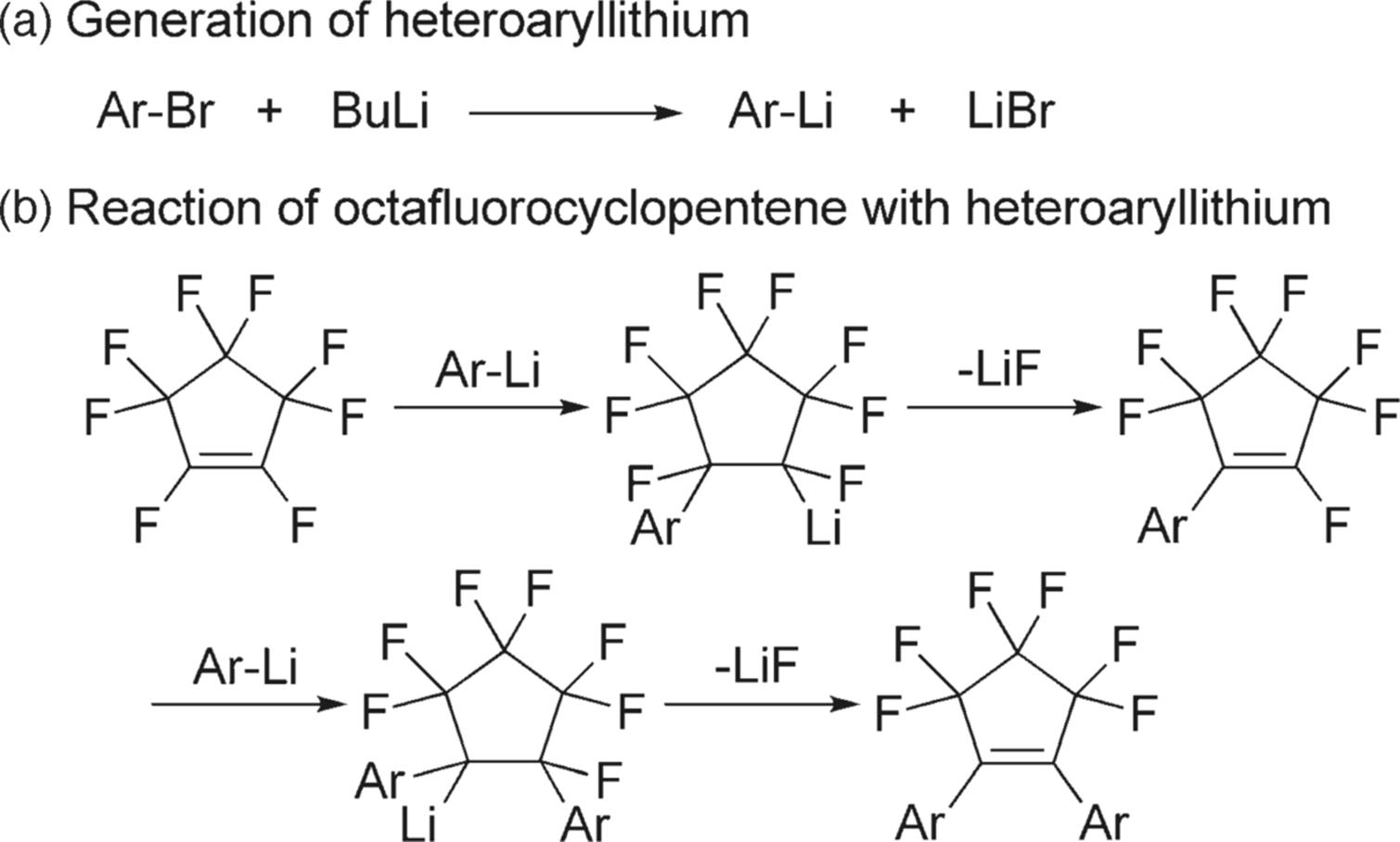
The University of Kyoto, the MCPT research group, and Yamada Chemical developed an effective method for the synthesis of photochromic diarylethenes through the generation of heteroaryllithiums and subsequent reaction with octafluorocyclopentene by using integrated flow microreactor systems [75] (Figure 11.26). The reactions are performed at very low temperatures (<− 78 °C) in batch devices. Reactions can be conducted without using cryogenic conditions by virtue of effective temperature and residence time control. Moreover, the synthesis of unsymmetrical diarylethenes, which is difficult to achieve when using conventional batch macrosystems, has been accomplished based on the selective introduction of one aryl group to give arylheptafluorocyclopentene followed by the introduction of another aryl group. The productivity of the laboratory-scale system is approximately 0.5 mmol/min. Therefore, the present integrated flow microreactor method serves as a practical way of synthesizing various photochromic diarylethene derivatives.
Figure 11.26 Synthesis scheme of photochromic diarylethenes. (a) Generation of heteroaryllithiums through the Br–Li exchange reaction of heteroaryl bromides and (b) the reaction of octafluorocyclopentene with heteroaryllithium.
11.4.14.2 Cross-Coupling in a Flow Microreactor
The MCPT research group and the University of Kyoto investigated the Murahashi coupling in a flow microreactor system [76] (Figure 11.27). Integration with the Br–Li exchange of ArBr with BuLi in a microreactor was enabled by the use of palladium catalysts bearing a carbene ligand.
Figure 11.27 Microreactor system for Murahashi coupling.
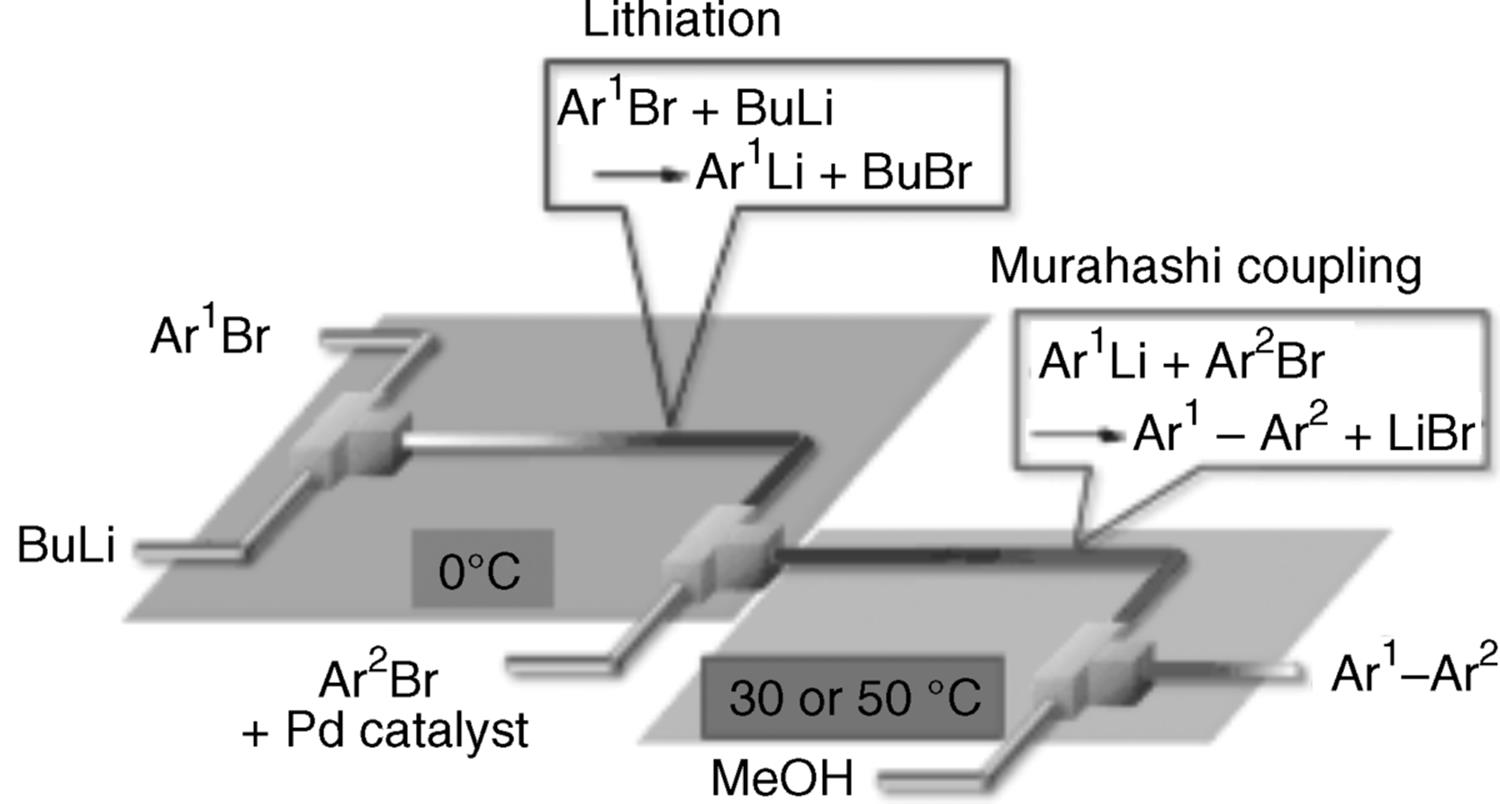
A cross-coupling of two different aryl bromides occurred within a minute without necessitating low temperatures (−78 °C) needed in a conventional process.
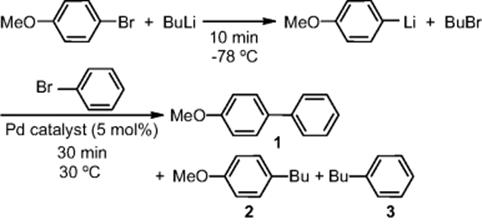
In the case of heteroaromatic compounds, the use of H–Li exchange enabled cross-coupling of ArH and ArBr. Hence, the method greatly enhances the synthetic utility of organolithium compounds and adds a new dimension to the chemistry of cross-coupling. Wider scope and limitations of this useful transformation are under investigation.
11.4.15 Direct Fluorination of Ethyl 3-Oxobutanoate
Following extensive laboratory studies at the University of Durham, UK, on the direct fluorination of ethyl 3-oxobutanoate in formic acid, scale-out was done from a three- to a nine-channel microstructured reactor, in cooperation with Asahi Glass Co. in Yokohama, Japan [77].
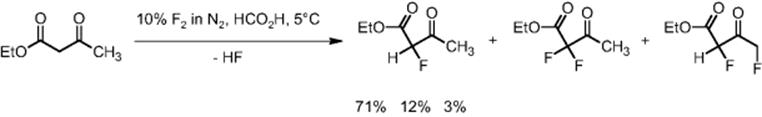
The process was performed for many months yielding 700 g of monofluorinated product with the nine-channel microstructured reactor [77]. A continuous 150 h operation was performed without decline of yield or conversion. Even in the scale-out to a 30-channel reactor (see Figure 11.28), no loss in performance was noticed. A single feed system distributed the reactants and reagents to the various microchannels.
Figure 11.28 A 30-channel microstructured reactor for the direct fluorination of ethyl 3-oxobutanoate. Source: Courtesy of the Royal Society of Chemistry [77].

If the results of the first pilot studies are extrapolated to large-scale synthesis, a two-sided 30-channel device with 60 channels would be capable of synthesizing about 300 g product per day [77]. By external numbering to 10 reactors, about 3 kg of product per day would result. Such a pilot plant benefits from low expenditure and having exactly the same operating conditions as given for the laboratory processing. In addition, convenient maintenance and purification by distillation at improved safety features are predicted as major advantages.
11.4.16 Deoxofluorination of a Steroid
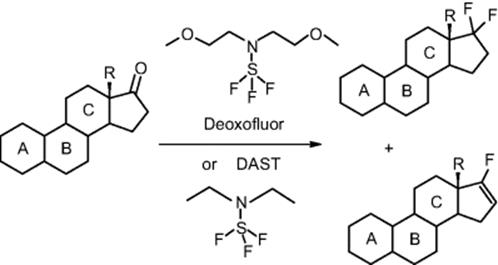
Bayer in Berlin, Germany, developed a continuous process for the fluorination of a steroid derivative with bis(methoxyethyl)aminosulfur trifluoride to produce geminal difluoride [78]. The goal was maximization of conversion and the selection of optimal residence time, temperature, and reagent amount. The continuous reaction was performed on a laboratory scale, in a 3 mm inner diameter stainless steel tube. As an important reaction in medicinal chemistry, further investigations about a larger scale process are on the way.
11.4.17 Microprocess Technology in the United States
Novartis–MIT Center for continuous manufacturing is 10-year research collaboration between Novartis and Massachusetts Institute of Technology, started in 2007. The research is aimed at transforming pharmaceutical production. Combining the industrial expertise of Novartis with MIT's scientific and technological leadership, the Center develops new technologies to replace the pharmaceutical industry's conventional batch-based system with a continuous manufacturing process. It is aimed to bring benefits to patients, healthcare providers, and the pharmaceutical industry by accelerating the introduction of new drugs through efficient production processes, minimizing waste, energy consumption, and raw material use, monitoring drug quality on a continuous basis rather than through postproduction, batch-based testing, and enhancing process reliability and flexibility to respond to market needs.
Initial research is conducted at MIT laboratories, while Novartis applies the research to industrial-scale projects and pilots new manufacturing processes using its own pharmaceutical products. The company invested $65 million in this research [79].
To demonstrate the system, the researchers built a prototype that produces tablets of a specific drug manufactured by Novartis (see Figure 11.29). However, the system is designed so that components can be swapped in and out to create different drugs.
Figure 11.29 Continuous drug manufacturing prototype in an MIT chemical engineering laboratory [80].

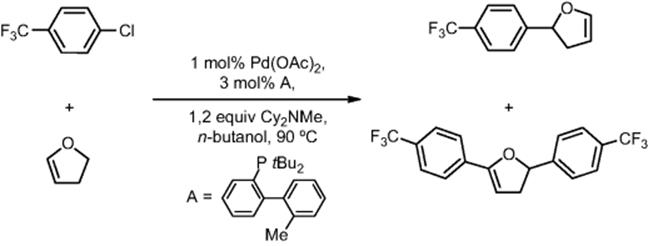
The research in the field of microreactor-based continuous processes is a large portion of their research. In one of the projects, researchers from Novartis–MIT Center for Continuous Manufacturing developed a self-optimizing microreactor with a feedback reaction that uses previous reaction data to efficiently select future experiments. They maximized the yield of a Heck reaction of 4-chlorobenzotrifluoride and 2,3-dihydrofuran by adjusting the equivalents of the alkene and the residence time [81]. Optimal reaction conditions in the microreactor were determined after 19 automated experiments and required a relatively small amount of starting material. The reaction was then successfully scaled up 50-fold using the optimal conditions determined by the microreactor system.
More papers from Klavs Jensen's group depict their collaboration with companies such as Dow Chemicals where they explore silicon-on-insulator (SOI)-supported microdevice for hydrogen purification for industrial applications [82]. Hydrogen purification using palladium–silver membranes was possible at higher pressures than in previous devices. Thin (200 nm) palladium–silver membranes yield high permeation rates of up to 50 mol/(m2 s) at 350 °C. An analysis of the transport resistances controlling hydrogen permeation in the micromembrane system suggested that the process was limited by gas-phase diffusion and that even larger fluxes would be possible with improved flow channel designs.
In collaboration with DuPont, a direct multistep synthesis of sodium nitrotetrazolate was investigated in a modular silicon micromixer. The mixer is operated by splitting two inlet flows into a large number of channels, interdigitating them, and maintaining the laminated flow to create submicrometer diffusion lengths. Successful production of 4.4 g/h of nitrotetrazolate in solution was ultimately achieved, verifying the potential for scaling to typical production amounts. Minimizing concentration and temperature gradients and applying continuous flow enabled safe, room-temperature, continuous synthesis of nitrotetrazolate, greatly simplifying and accelerating the process by an order of magnitude compared to the traditional batch method. The reaction setup for determining kinetics is shown in Figure 11.30.
Figure 11.30 Reaction setup for multistep synthesis of sodium nitrotetrazolate.
11.4.18 Propene Oxide Formation
The propene oxidation process is globally conducted on a 5 million t/a scale. A new gas-phase oxidation process of propene to propene oxide using hydrogen peroxide would avoid use of solvents and is expected to have higher selectivity.
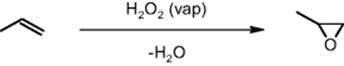
Microstructured reactors have the potential to perform a key step here to thermally treat concentrated hydrogen peroxide solutions for vaporization, which would be dangerous using conventional technology due to handling of the combustible hydrogen peroxide. Thus, large microreactor units need to be built that are capable of safe operation of the hydrogen peroxide route. In addition, these units need to transfer the large reaction heats produced in the exothermic propylene oxide reaction.
Following these ideas, Degussa in Hanau, Germany, developed and built, together with the large-scale plant manufacturer Uhde GmbH in Dortmund, Germany, and other university and institute partners in a publicly funded project (DEMiS), a 6 m long, two-storey-high pilot-scale microstructured reactor by numbering up of meter-long and -wide plates brought to micrometer distance (see Figure 11.31) [83]. The lower part of the reactor comprises the vaporizer and in the longer upper part the reaction plates are positioned. The construction uses a simplified, robust approach with no real walls for laterally defining microchannels, but the few fins only have mechanical function and the flow dimension in lateral direction is virtually infinite compared to typical microchannels. This simplification in reactor design has to be paid by development of a catalyst coating technique for such extended dimensions. Typically, the small dimensions in microchannels help in having good mechanical adhesion to the substrate and also promote uniform coating due to surface forces. Initial results are very stable and reproducible and Degussa considers this as encouraging.
Figure 11.31 (a) DEMiS pilot reactor for propylene oxide formation at Degussa site, superposed by schematic of the reactor construction. (b) Assembly of the reactor internals. Source: Courtesy of Wiley-VCH Verlag GmbH [83].

11.4.19 Diverse Industrial Pilot-Oriented Involvements
Bayer Company operates several microprocess plants. Clariant GmbH has formed a competence center on microreaction technology in early 2004 to offer the technology in its custom synthesis business. Lonza has also used microreaction technology over years for pharmaceutical customers. The pharmaceutical company Bristol-Myers Squibb, Lucent Technologies, and the Stevens Institute work on a public funded project on hydrogenation reactions [84]. They started with a model reaction, the hydrogenation of o-nitroanisole to o-anisidine, and will move to proprietary reactions.
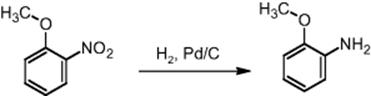
The reaction engineering uses the innovative approach of fixing single Pd/C catalyst particles in thousands of small microstructured traps [84]. This is a mixed approach relying on traditional catalysts and using the innovative flow features in microchannels. In this way, higher specific surfaces between gas, liquid, and catalyst are ensured than possible by using fixed beds. Compared to wall coatings, it is expected that the trapped catalyst provides a more reliable and potentially higher catalyst coating.
Bristol-Myers Squibb has filed patents for making glycosides using noncryogenic processes that involve the lithiation of aromatic compounds [84]. These aryllithium derivatives react with carbonyl groups of other reactants to form a glycoside. Both steps are highly exothermic and rapid, which reveal the advantages of microprocess technology in terms of selectivity and reduced manufacturing cost.
The Syrris Company, a system manufacturer, and GlaxoSmithKline developed an automated flow system AFRICA (automated flow reaction incubation and control apparatus) where a sequence of reactions can be run under several process conditions using the advantages of miniature-scale flow [84]. Attached to the system is an online high-pressure liquid chromatograph for immediate product separation to analyze the performance of process optimization. As further scientific support, Syrris has entered into collaboration with S.V. Ley at Cambridge University who studies flow-through liquid reactions with immobilized reagents. Pfizer has some high-throughput oriented research and development work with a microreactor combined to an automated multiple reactant feed upstream and UV detection downstream.
Dow Chemical in Midland, MI, USA, the microprocess technologist Velocys in Plain City, OH, USA, and PNNL in Richland, WA, USA, as research institute in microreactor technology work on a public funded project on high-intensity production of ethylene and other olefins by oxidation such as the formation of ethylene from ethane [4]. A two-step reactor engineering is performed, starting with a bench-scale reactor with microchannel dimensions equal to the later commercial unit and followed by numbering to the latter. An economic analysis with focus on reactor costs and energy consumption completes the project.
At Sigma-Aldrich, a pilot study was performed for the transfer of microreactor technology into large-scale production for the synthesis of 2-benzoylpyridine through a Grignard reaction [85]. They transferred the small-scale 2 ml microreactor processes to production in a stainless steel Alfa Laval Art® plate reactor and achieved rapid flow rates and continuous production of 200–300 kg of 2-benzoylpyridine per day.
There have been also advancements in the pharmaceutical industry. Thus, DSM designed a microreactor system to produce a few hundred tons of naproxcinod per year [86].
Recent patents from Velocys, Uhde GmbH, Lonza, AstraZeneca, and other companies justify the increasing industrial interest in gas–liquid–solid processes in microreactors [87].
The field of mobile gas-to-liquid conversion has recently also seen the developments toward continuous microflow processing. Velocys has signed joint agreement with Petróleo Brasileiro S.A. (Petrobras) [88]. The goal is the operation of an integrated gas-to-liquid demonstration plant based on Velocys' catalyst and microchannel reactor technology. The plant will be operated for 9 months and total costs are estimated at tens of millions of dollars. The technology is to be applied on floating production storage and offloading (FPSO) vessels used by the offshore industry for the processing of hydrocarbons and for storage of oil [6].
11.4.20 Production of Polymer Intermediates
DSM Fine Chemicals GmbH in Linz, Austria, installed a microstructured reactor into an existing production plant for the Ritter reaction in a retrofit manner (see Figure 11.32). A high-value intermediate, not disclosed by chemical formula, for the polymer industry is produced [89]. This approach, under involvement of the product team right from the start, stands for a plant philosophy that may be best paraphrased by “minimal invasive plant surgery,” different from some holistic approaches for a total change in plant design. This enabled the replacement of a central reaction route in a very large reactor tank encasing several tons of explosive and corrosive chemicals. Design and fabrication of this reactor was done at the Institut für Mikroverfahrenstechnik (IMVT) in the Forschungszentrum Karlsruhe (FZK), Germany.
Figure 11.32 Production-type microstructured reactor for throughput at 1700 kg/h and transfer of a power of 100 kW. This apparatus was used for manufacture of a high-value product for plastics industry at DSM in Linz, Austria. Source: Courtesy of Wiley-VCH Verlag GmbH [90].

During a 10-week production campaign, over 300 tons of the polymer product was produced [89]. The microstructured reactor (65 cm long; 290 kg heavy, special nickel alloy, several ten thousands of microchannels) was operated at a throughput of 1700 kg liquid chemicals per hour. A critical issue was the removal of the reaction heat, which was accomplished within seconds. The yield exceeds that of the former route, albeit it was not detailed. It was also found that the process safety for handling the corrosive chemicals was higher for the microreactor process. The use of raw materials and the waste streams were reduced, improving the cost and efficiency of the process.
11.4.21 Synthesis of Diazo Pigments
Clariant carried out diazo coupling for pigment synthesis. Laboratory developments were transferred to pilot scale, reported in the literature, and a further transfer to production scale was announced at conferences [91].
![]()
Particle synthesis is known to be highly sensitive to mixing, as this controls seed formation and crystal growth. With the Clariant and later works, it became evident that by use of micromixing technology particles with more uniform size, defined morphology, or chemical selectivity can be prepared, that is, with considerably improved product qualities, which is the main driver for such investigations. The motivation to use microreactors also stems from the benefits of developing a continuous process, for example, the production of flexible quantities and eliminating the need of refining such as milling the pigment at the end of the production line, as typically done for batch processes. A further argument for microreaction technology in the case of diazonium salt-based synthesis comes from the hazardous potential of that intermediate. A major hurdle is to find processing solutions, which allow one to handle solids generated in microchannels.
Two commercial azo pigments, one yellow and one red, were made based on azo benzene coupling; the nature of the substituents was not disclosed [91]. Diazotization was performed in batchwise manner. A CPC microreactor with multilamination mixer (lamellae <100 μm) was used. The two azo pigments had a color strength of 119 and 139%, five and six times glossier brightness, and five and six steps higher transparency as compared to the same products made by batch processing (see Table 11.5), respectively [91]. This originated from the formation of smaller particles with more narrow size distribution (microreactor: D50 = 250 nm, s = 1.5; batch: D50 = 600 nm, s = 2.0).
Table 11.5 Coloristic properties of pigments synthesized in two different microreactors compared to the batch standard.
|
Microreactor pigment 1 |
Microreactor pigment 2 |
|
|
Color strength |
119% |
139% |
|
Brightness |
5 steps glossier |
6 steps glossier |
|
Transparency |
5 steps more transparent |
6 steps more transparent |
|
Source: From Ref. [91]. |
||
The same features were found for pilot-size microreactor operation (see Figure 11.33). Brightness and transparency were the same, and the color strength could even be increased to 149% [91]. The mean particle size was even smaller as compared to the laboratory-scale microreactor processing (microreactor: D50 = 90 nm, s = 1.5; batch: D50 = 600 nm, s = 2.0), probably due to process optimization.
Figure 11.33 Size characterization of an azo pigment, produced in the pilot plant microreactor, compared to the batch standard. (a) Volume density distribution and (b) TEM magnification. Source: Courtesy of Wiley-VCH Verlag GmbH [92].
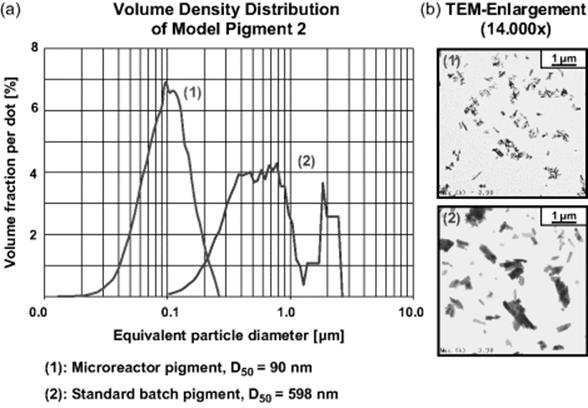
Pilot-size microreactor operation was performed using a flow of 500 ml/h, which is equivalent to a production in the range of 10 t/a when accounting for 8000 h annual running time [91]. The increase in throughput compared to laboratory-scale microreactors used earlier (1 t/a; 20–80 ml/h) was achieved by both internal and external numbering up accompanied by a slight scale-up of internal dimensions. More reaction plates were assembled in parallel within one device; in addition, three such devices were connected in parallel. Furthermore, slightly larger microchannels were used, still ensuring laminar flow.
To test for fouling, a 24 h run of a pilot-scale microreactor for azo pigment production was performed using a diazo suspension [91]. At the end of this period, the pressure loss of the microreactor increased exponentially. Special means were developed to prevent clogging and unstable operation. By partial removal of the deposits, the pressure loss was brought back to normal.
Continuous production of a diazo compound in microreactors separated by a hydrophobic membrane was also patented by DSM IP Assets, The Netherlands, and DSM Fine Chemicals, Austria [93]. Continuous supply, mixing the precursor of a diazo compound, the water-miscible solvent, the base, and water to generate a diazo compound, and continuous removing from micro- or millireactors are involved in the preparation of diazomethane and diazoethane.
11.4.22 Selective Nitration for Pharmaceutical Production
DSM and Corning Incorporated have collaborated to develop and pilot a new process to perform a selective nitration for pharmaceutical production under c-GMP conditions [94]. DSM developed and intensified a process to utilize the capabilities of the Corning's reactor technology. Twelve glass microstructures enabling the mass and heat transfer functions were required. The complete reactor made by assembly of the glass microstructures with other parts through appropriate connections, frames, and piping is shown in Figure 11.34a.
Figure 11.34 (a) Reactor made by assembly of the glass microstructures. (b) Production bank with four identical reactors and distribution system.

This combination has resulted in a highly efficient process, which was quickly and easily transferred from the laboratory to production, resulting in the processing of more than 25 metric tons of material.
The transfer to a production unit required no scale-up work – parameters obtained on one reactor were applied to the production unit. The unit is shown in Figure 11.34b.
The production unit has been implemented at DSM into a standard industrial production facility, connected with conventional pumping systems and plant utilities. The total flow processed by the production unit was approximately 100 kg/h, which corresponds to approximately 800 tons per year of the desired quality product under c-GMP conditions, based on 8000 h of continuous operation.
11.4.23 Nitroglycerine Production
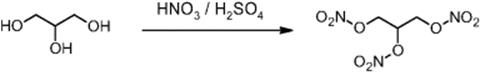
The Xi'an Huian Industrial Group in Xi'an, China, operates a nitroglycerine plant with at 15 kg/h production, which has been developed and installed by IMM (see Figure 11.35) [86].
Figure 11.35 Microprocess production plant for pharmaceutical nitroglycerine. Source: Courtesy of H. Löwe, IMM.

The nitroglycerine is of pharmaceutical grade and used as medicine for acute cardiac infarction. This demands high selectivity and low levels of impurity. First manual plant start-up tests demonstrated that this can be achieved by performing the reaction in a microreactor. Safe operation was found during the first runs. In the second step, full automation of the plant is planned. The extension of the process chain is another future issue, adding a purification unit for washing and drying and finally a unit for formulation and packaging to encapsulate the nitroglycerine drug safely in tablets. Advanced wastewater treatment and a closed water cycle should lead to an environmentally clean process.
11.4.24 Fine Chemical Production Process
Microinnova KEG, Graz, Austria, carried out process development and the installation of a StarLam 3000 microstructured mixer in an existing production plant of an undisclosed customer and for an undisclosed chemical process (see Figure 11.36) [95]. The aim was to double the capacity of a running two-step batch process. This was achieved by installing the microreactor for the first reaction step. A higher reaction rate made it possible to reach overall throughputs of 3.6 tons per hour. In addition, energy savings were achieved. The microstructured mixer is running in production for more than a year now.
Figure 11.36 StarLam 3000 microstructured mixer retrofitted to existing plant peripherals and tank reactor. Source: Courtesy of Wiley-VCH Verlag GmbH [95].

The application refers to the production of fine chemicals [95]. A 10 m3 batch vessel was used to perform the two-step chemical process. The first strongly exothermic reaction step needed cooling due to the volatility of one of the starting materials. The reaction was complete when all of the volatile reactant had reacted. The second step was endothermic and the batch vessel had to be heated for some hours to complete the reaction. It took several hours to perform these two steps and a throughput of about 1800 kg/h was achieved.
In laboratory-scale investigations, the reactor and attached tube reactor were kept at temperatures of about 150 °C [95]. The experiments showed that in the microprocess laboratory plant the first reaction step can be completed in less than 60 s, whereas it takes about 4 h to perform it in the cooled batch vessel of the production plant.
The inlet pipes of the two starting reactants to the batch vessel were simply connected to the StarLam mixer [95]. The only difference to the previous feed lines was the installation of filter cartridges before the entries to the microstructured mixer, necessary to avoid blocking of the reactor. The pressure drop in the lines was lower than 3 bar, so it was possible to install the pumps early in the plant. At the outlet of the reactor, a tube reactor was installed. During optimization it was found that it is sufficient to insulate this tube to reach the temperature needed to complete the reaction. The pipe ended directly in the batch vessel where the second endothermic reaction step was carried out as before.
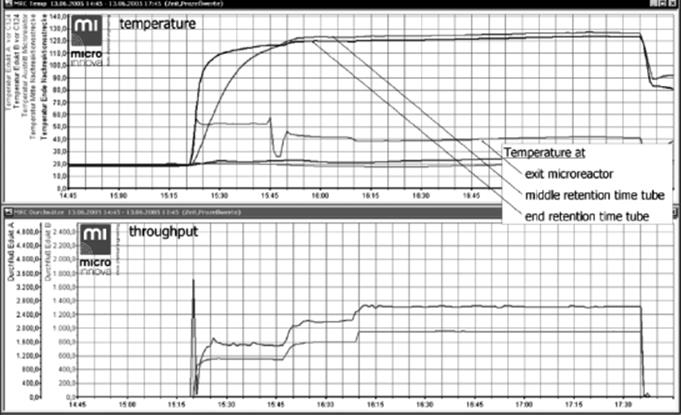
The first start-up of the plant was in June 2005 [95]. A temperature diagram shows that most of the heat is released in the retention time tube (see Figure 11.37). The temperature measured directly at the outlet of the microstructured mixer–reactor StarLam 3000 was below 50 °C even at higher throughputs. During the retention time, tube temperatures of up to about 130 °C were reached. The throughputs for this first test run were increased in three steps to up to 3600 kg/h.
Figure 11.37 Temperature diagram of the microreactor-assisted processing. Source: Courtesy of Wiley-VCH Verlag GmbH [95].
11.4.25 Grignard-Based Enolate Formation
Merck KGaA in Darmstadt, Germany, investigated a Grignard reaction with a high reaction enthalpy of 300 kJ/mol and high reaction speed so that heat transfer limitations result for conventional technology [96]. The Grignard reactant having a long alkyl chain was added to a keto compound; the substituents remain disclosed. An enolate was formed that was further reacted in the frame of a multistage fine chemical industrial process.

A yield of 95% was obtained by a micromixer-based process (<10 s, at −10 °C), while the industrial batch process (6 m3 stirred vessel) gave only 72% yield (5 h, at −20 °C) [96]. The laboratory-scale batch process (0.5 l flask; 0.5 h, at −40 °C) gave 88% yield (Table 11.6). Pilot-scale studies followed with a home-built minimixer for reasons of clogging, which was not decisive at the laboratory scale. With one minimixer at the pilot scale, a yield of 92% was obtained (<10 s, at −10 °C). The validity of the numbering-up concept was proven by using five minimixers of the same type, which gave a yield of 92% (<10 s, at −10 °C). This was the central part of the actual production process, running for more than 3 years until the life cycle of the commercial product of the corresponding multistage process ran out (see Figure 11.38).
Figure 11.38 Merck production plant for Grignard-based enolate formation. Source: Courtesy of Springer [96].

Table 11.6 Comparison of reaction time and yield for different reactor types.
Source: From Ref. [96].

Meanwhile, Merck has reported to have 20 microreactor plants under operation for diverse reactions [91]. The production costs are typically reduced by 20% as compared to prior conventional technology. The throughputs range from 50 g/h to 4 kg/h, which corresponds to 146 kg/a and 11.7 t/a, respectively.