Organic Chemistry: Concepts and Applications - Headley Allan D. 2020
Synthetic Polymers and Biopolymers
20.3 Anionic Polymerization of Alkenes
As pointed out earlier, the reaction of alkenes with anions results in carboanionic intermediates. Carboanions can be stabilized through resonance and hence alkenes that have electron-withdrawing groups that can stabilize anions result in alkene reactants that are ideal for the synthesis of polymers via an anionic intermediate pathway.
20.3.1 Anionic Polymerization of Vinylidene Cyanide
A classic example is the polymerization of 1,1-dicyanoethene, also known as vinylidene cyanide in the presence of a nucleophile, such as the hydroxide anion which forms a very strong glue. The reactions are shown below.
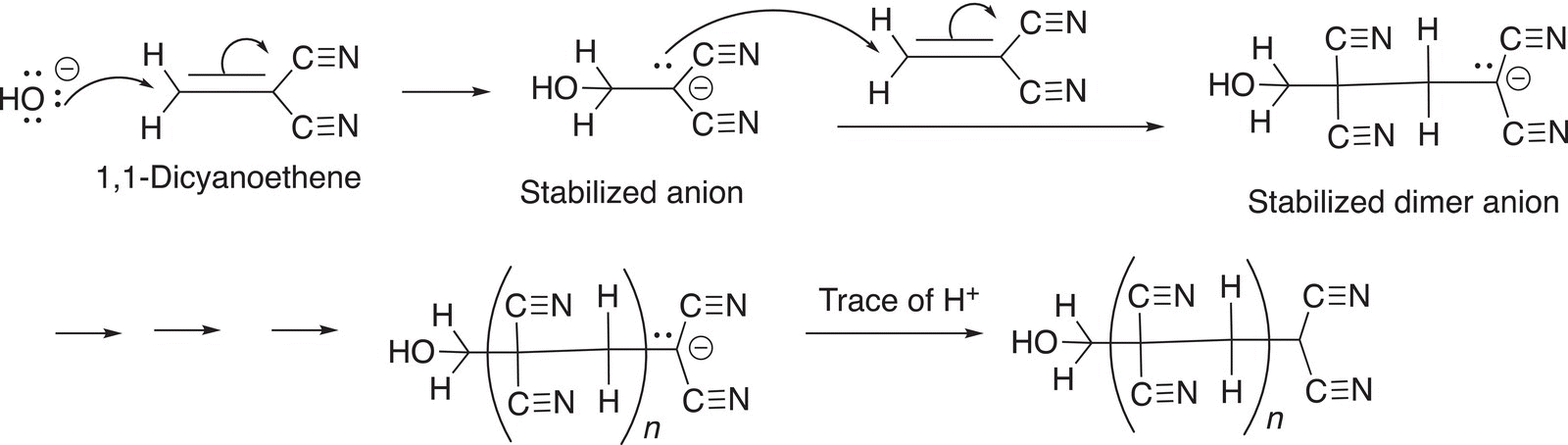
When the tube of this glue is opened, a trace of moisture, in which just a minute quantity of hydroxide anions is present starts the polymerization reaction. After the polymer is formed, a trace of protons terminates the reaction and the result is a solid polymer, which is used to join two surfaces together.