Make: The Annotated Build-It-Yourself Science Laboratory (2015)
Part II. Physics
Chapter 6. Geology
Geology is often thought of as a field science. Many people feel that geology is limited to only what one can see and collect. To them geology is collecting interesting rocks and perhaps fossils.
Geology is not a spectator type of science. It is rather a participation type of science filled with nearly as much experimentation as any other branch of the physical sciences.
Geology is often defined as the study of the history of the Earth, what it is made of, and how it was formed. The branch of geology that deals with the study of rocks is called petrology. The branch that deals with minerals is called mineralogy.
Identification is important in either the study of rocks or minerals. The equipment needed to perform various identification tests is very simple and inexpensive. Most of the equipment has been described in other sections of this book, and if this is the case, the reader will be referred to the appropriate page.
General Equipment Needed for Geology
§ Beam balance (“Sensitive Gram Scale”)
§ Magnet (“Making a Magnet (or Recharging a Magnet)”)
§ Test tube holder (“Ring Support for Support Stand and Test Tube Holder”)
§ Alcohol lamp (“Alcohol Burner”)
§ Blowpipe (“Blowtorch Type of Alcohol Burner”)
Other equipment you will need:
§ Hand lens (tripod magnifying glass 5 to 10 power)
§ File
§ Test tube (closed tube)
§ Jackknife
Chemicals: 1
§ Borax
§ Ammonia (kitchen cupboard)
§ Sodium Bicarbonate (baking soda)
§ Hydrochloric acid-concentrated and dilute
§ Nitric acid-concentrated and dilute (optional)
Additional Equipment to Make or Improvise
§ Streak plate (unglazed tile)
§ Glass rod (seal both ends of a piece of glass tubing)
§ Nichrome wire loop (nichrome wire and glass tubing)
Rock and Mineral Worksheet
It is very helpful to make several copies of the following worksheet. This record sheet will aid you in your investigation of the specimen.
Test for Hardness
The standard test for hardness is the Mohs scale. This scale rates the minerals from 1 to 10, with ten being the hardest. Sample minerals for each degree of hardness on Mohs scale are:
|
Talc: 1 |
Orthoclase: 6 |
|
Gypsum: 2 |
Quartz: 7 |
|
Calcite: 3 |
Topaz: 8 |
|
Fluorite: 4 |
Corundum: 9 |
|
Apatite: 5 |
Diamond: 10 |

You may substitute many common materials for some of the minerals which you do not possess. Some common materials2 and their degrees of hardness are:
|
Fingernail: 2.5 |
Quartz/Flint: 7.0 |
|
Penny: 3 |
Emery paper: 8.0 to 9.0 |
|
Window glass/Knife blade: 5.5 to 6.0 |
Corundum paper: 9.0 |
|
Steel file: 6.5 to 7.0 |
Carborundum: 9.5 |
The hardness test is based on the fact that a harder material will scratch a softer piece of material. For instance, a penny will scratch a piece of talc (hardness 1) but not a piece of window glass (hardness 5.5). The penny is harder than 1 but softer than 5.5. By trying the penny on gypsum and fluorite we would find the penny would scratch the gypsum but not the fluorite. Therefore, the penny is somewhere between 2 and 4 on the hardness scale. By trying the penny on calcite we find it just about makes a faint mark on the calcite. We would conclude the hardness is just about that of calcite.
You must observe the mineral you are testing very closely to be sure that the surface you are testing is being marked. Many times a mineral of lesser hardness will leave a streak of its own on the mineral being tested. This is sometimes mistaken for a scratch.
Research suggestions:
1. Can you determine the hardness of various materials found around the house? Sample materials might include buttons, various metals, wood, ceramics, and others.
2. Can you determine the hardness of various rocks and minerals found around your neighborhood? You might compare the average hardness with that of rocks and minerals found in other locations.
3. Can you develop your own complete scale of hardness based on common materials instead of those suggested for Mohs scale?
Color Test
A careful visual examination of a specimen is usually sufficient to give you the correct natural color of the material. Be sure, however, that you have a fresh sample, as some specimens tarnish or change color from weathering.
Luster Test
Another characteristic or property of a mineral is the luster.3 Luster is the appearance of the mineral in ordinary light. Quartz and glass are vitreous or glassy in appearance. A diamond has adamantine luster.4 Stibnite has a metallic luster. Other terms are: earthy, dull, greasy, silky, pearly, and resinous.
Streak Test
The streak of a mineral is in some cases one of the most important tests. The streak is determined by any of three ways. First, the specimen is rubbed across a streak plate of unglazed porcelain (such as unglazed tile). The resulting color of the mark on the plate is the streak color. A second way streak can be determined is by scratching the mineral with a knife and observing the color of the powder. A third method for determining the streak is by observing the color of a crushed mineral.
If the mineral is harder than the streak plate, it will scratch the plate and leave a powder residue on the plate. If your streak is white, check the plate with your hand lens. The white mark may not be from the mineral. It may be caused by scratching the plate.
The streak is in many cases a different color from that of the mineral being tested and is more constant than color as a property of a mineral.
Fusibility Test
In order to test the fusibility of a mineral, a thin piece of the specimen is held in a pair of forceps or tweezers and placed in the flame of an alcohol burner or a blowpipe. If the mineral seems to melt, it is fusible at that temperature. A scale of fusibility is as follows:
1. Stibnite: fuses in the flame from an alcohol burner (980 °F)
2. Chalcopyrite: fuses easily in a blowpipe flame (1475 °F)
3. Almandine: fuses less easily in a blowpipe flame (1920 °F)
4. Actinolite: thin edges fuse in a blowpipe flame (2190 °F)
5. Orthoclase: thin edges fuse with difficulty (2374 °F)
6. Enstatite: thinnest edges fuse with a blowpipe (2550 °F)
7. Quartz: will not fuse in a blowpipe flame.
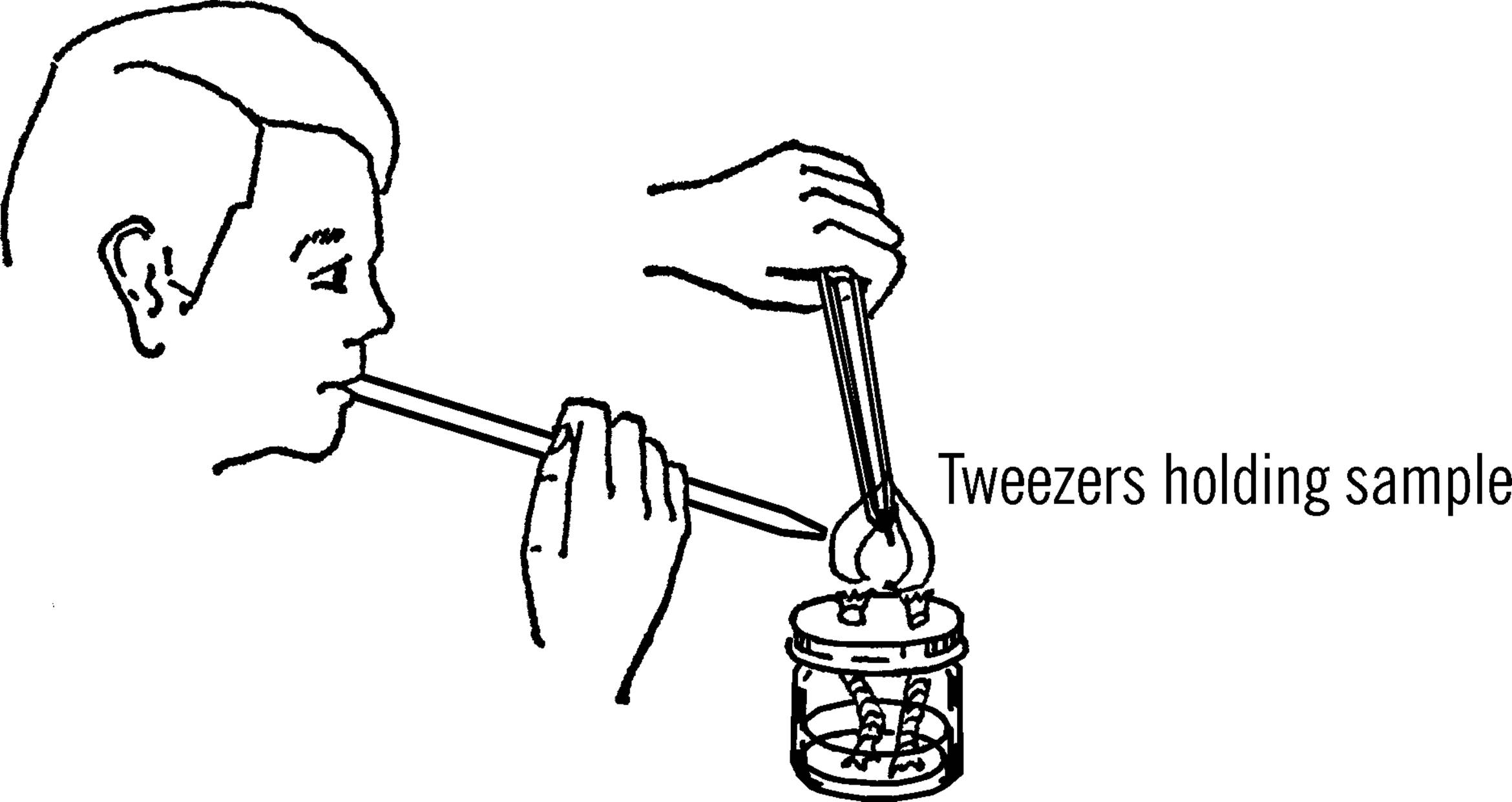
Modern Safety Practice
Follow the safety guidance given previously when using an open flame. See Note 3 in Appendix E. Wear your safety glasses!
Acid Test
For your acid bottle, use a small nose drop bottle or other bottle containing an eyedropper. This bottle should be labeled, “Dilute hydrochloric acid” (Dil. HCl). Your druggist will make up a dilute solution for you.5 You need the acid diluted in a ratio of one part acid and three parts water. If you are going to dilute the acid yourself, you must be sure to pour the acid into the water and not pour the water into the acid.
Be sure to carry this acid bottle with you in your field kit when you go exploring for rocks and minerals. This acid test offers one of the simplest means of determining carbonate rocks and minerals. If you place a small drop of dilute HCl on a specimen of a carbonate mineral, it will effervesce or give off bubbles.
Specific Gravity Test
The specific gravity of a mineral is its weight relative to the weight of an equal volume of water.
In the chemistry section you were given instructions for building a beam balance. You may use the same instrument for determining specific gravity if you make a few changes. Remove one of the paper cups and replace it with a small jar lid attached to the yardstick by string or thread as shown. Punch holes in the bottom of the jar lid with a nail. Place the beam balance on a stack of books or a block of wood about 4” high. The pan should be low enough so that it can be completely covered in the glass of water.
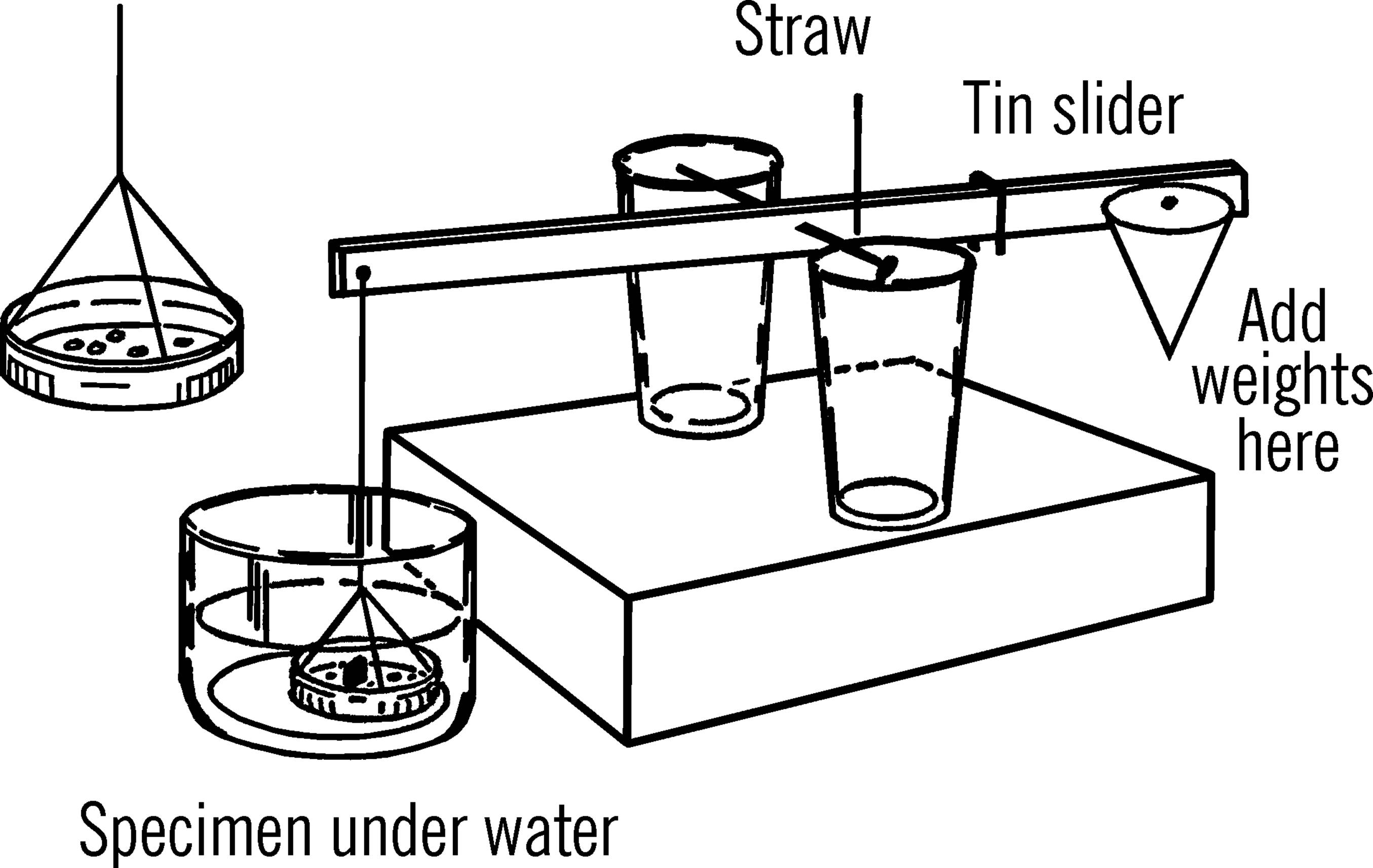
Procedure: Without the glass of water under the pan, balance the beam scale by using your slider on the right-hand side of the beam. Place a small piece of the specimen to be investigated on the pan and weigh it while it is dry. Record this weight.
Then, place the glass about half full of water under the left side of the beam and weigh the same specimen completely under water. Note this weight. Subtract the weight in water from the weight in air. This will give you the loss of weight, or the displacement in water. Then, divide the dry weight by the loss of weight, which will give the specific gravity.
The formula for specific gravity is:
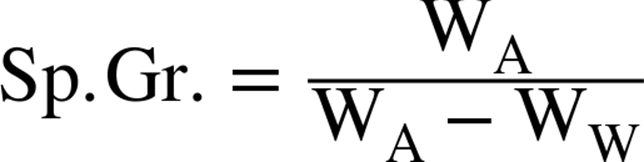
where WA is the weight in air, and WW is the weight in water.
Example: The weight of a specimen is 6 grams in air (its dry weight). In water the specimen weighed only 4 grams. The specific gravity would be determined as follows:
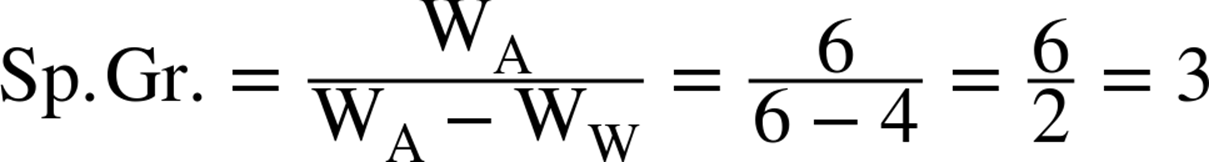
Bead Test
Purpose: Many minerals give off a characteristic color when placed in the tip of a very hot flame. The color in the flame is a clue to the chemical composition and thus to the type of mineral.
Materials: Nichrome wire about 3” long, glass tubing about 6” long, double wick alcohol burner, and a blowpipe.
What to Do: First make a loop about ⅛” in diameter in one end of the nichrome wire. You can form the loop around the tip of a pencil. Next, heat a 6” piece of glass tubing in your double wick alcohol burner until the glass becomes soft and begins to seal over. Push the straight end of the wire into the sealed end of the glass tube and then remove the tubing from the flame. The glass will then cool and harden around the wire.
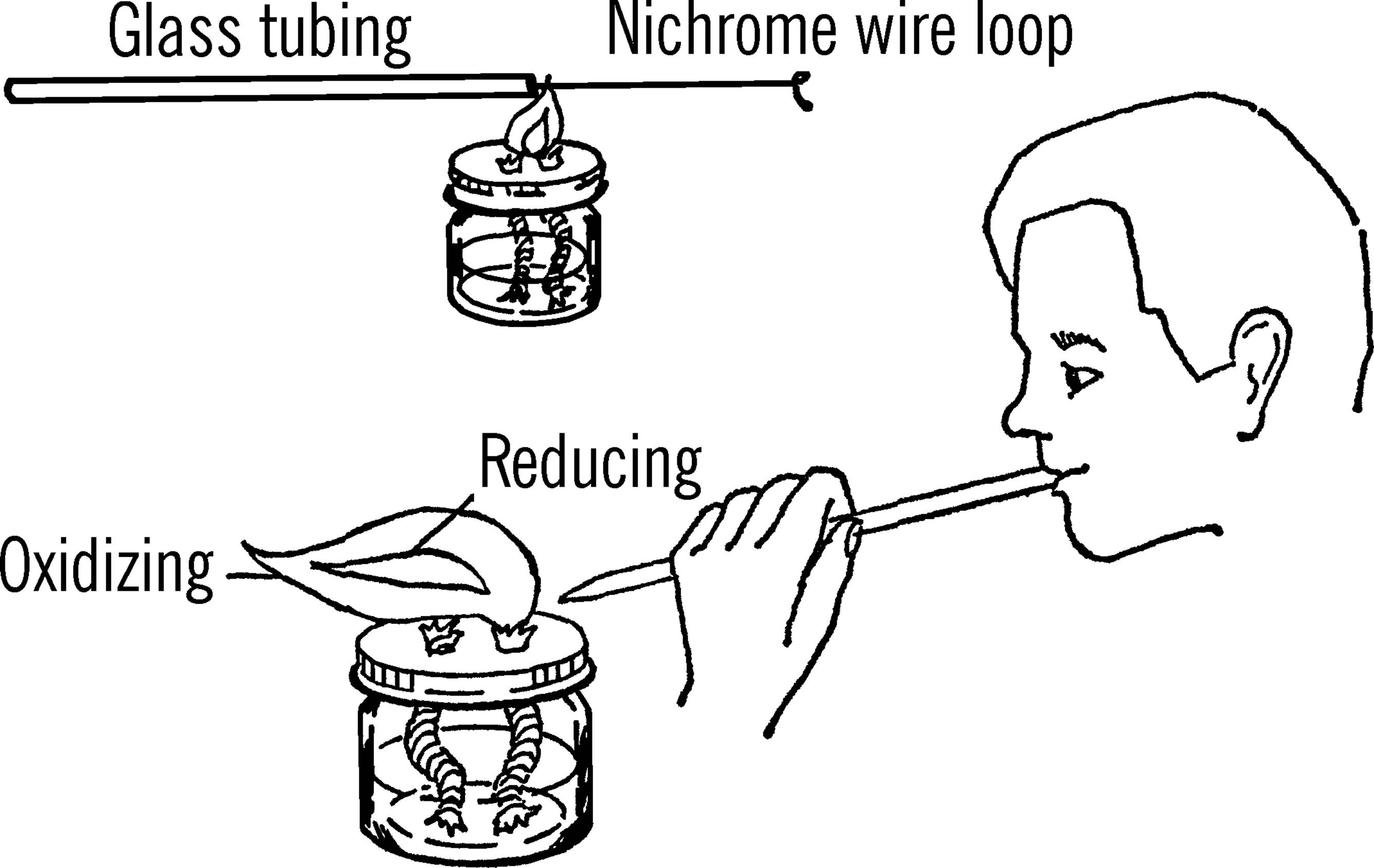
Operation of Equipment: Grind the mineral that you want to investigate into a fine powder. You can do this by breaking a small piece off the sample, covering the piece with a cloth so that the bits of rock and powder won’t scatter, and then hammering it into a powder.
Place the tip of the blowpipe in the edge of the flame of the alcohol burner. Blowing gently and continuously, you will produce a flame which is composed of two parts: the outer tip of the flame is called the oxidizing flame. The inner part is called the reducing flame.
With a little practice you will find that you can breathe in through your nose and blowout gently through the blowpipe at the same time. This produces a good continuous flame.6
Place the loop of wire in the oxidizing flame until it becomes red hot. Dip the loop quickly into powdered borax and pick up all that will stick to the wire. Place the loop with the borax back into the oxidizing flame until a bead is formed. This bead will be red hot. Pick up a small amount of the powdered mineral to be tested with the bead. Place the loop back into the oxidizing flame and note the color when hot. Then remove the loop from the flame and note the color when the bead is cold.
The following is a list of colors of the bead in the oxidizing flame and when cold.7
|
Substance |
Hot |
Cold |
|
Antimony (Stibnite) |
Yellow |
Colorless |
|
Copper (Azurite) |
Green |
Blue |
|
Iron (Limonite) |
Yellow |
Green |
Be sure the loop is clean before each test. Heat the bead in the blowpipe flame until the bead is red. Take the wire out of the flame and rap the bead sharply on a hard object to remove the bead. Dip the wire into HCl and then heat the wire in an alcohol flame. Repeat these directions until the flame is no longer colored.
Modern Safety Practice
1. When using an open flame, follow safety guidance in Note 3 of Appendix E.
Special Properties
Magnetism: Some of the iron minerals are naturally magnetic. Magnetite is one of these. Touch a magnet to a specimen of magnetite and you will find it attracts the specimen. Some specimens are very weakly magnetic. You can check these by bringing a compass near the specimen. If the rock contains iron, the compass needle is attracted or repelled. Some iron minerals become magnetic when heated. You will find this true of limonite, pyrrhotite, and pyrite.
Odor: Some minerals give off a characteristic odor. Pyrite, when heated, smells like sulfur. Arsenopyrite gives off sparks and a garlic odor when struck. Kaolinite has an earthy odor when breathed upon. Examine your specimens to see if other minerals give off odors.
Solubility: Minerals which dissolve in water are said to be water soluble. The following have this special property: halite, Epsom salt, potash, nitrates, borax, and soda.
Feel: Some minerals have a characteristic feel when rubbed with the fingers. Examples of this special property are:
§ Talc feels greasy
§ Kaolinite feels greasy
§ Graphite feels greasy
§ Meerschaum feels smooth
§ Molybdenite feels greasy
Chemical Tests: Some minerals are soluble (they dissolve) in hot or cold acids. As the mineral dissolves, it gives a characteristic color to the solution. If you then add some other chemical to the solution, the color may change again. These chemical tests are sometimes very helpful in the identification of a mineral.
In all chemical tests, try to keep the mineral being investigated as pure as possible. The associated minerals in a specimen might affect the weight and the chemical reactions of the specimen in question.
Only a very small amount of the mineral sample is necessary for these tests. The reactions are highly sensitive so a few grains of the solid with a drop or two of liquid is all that is needed.
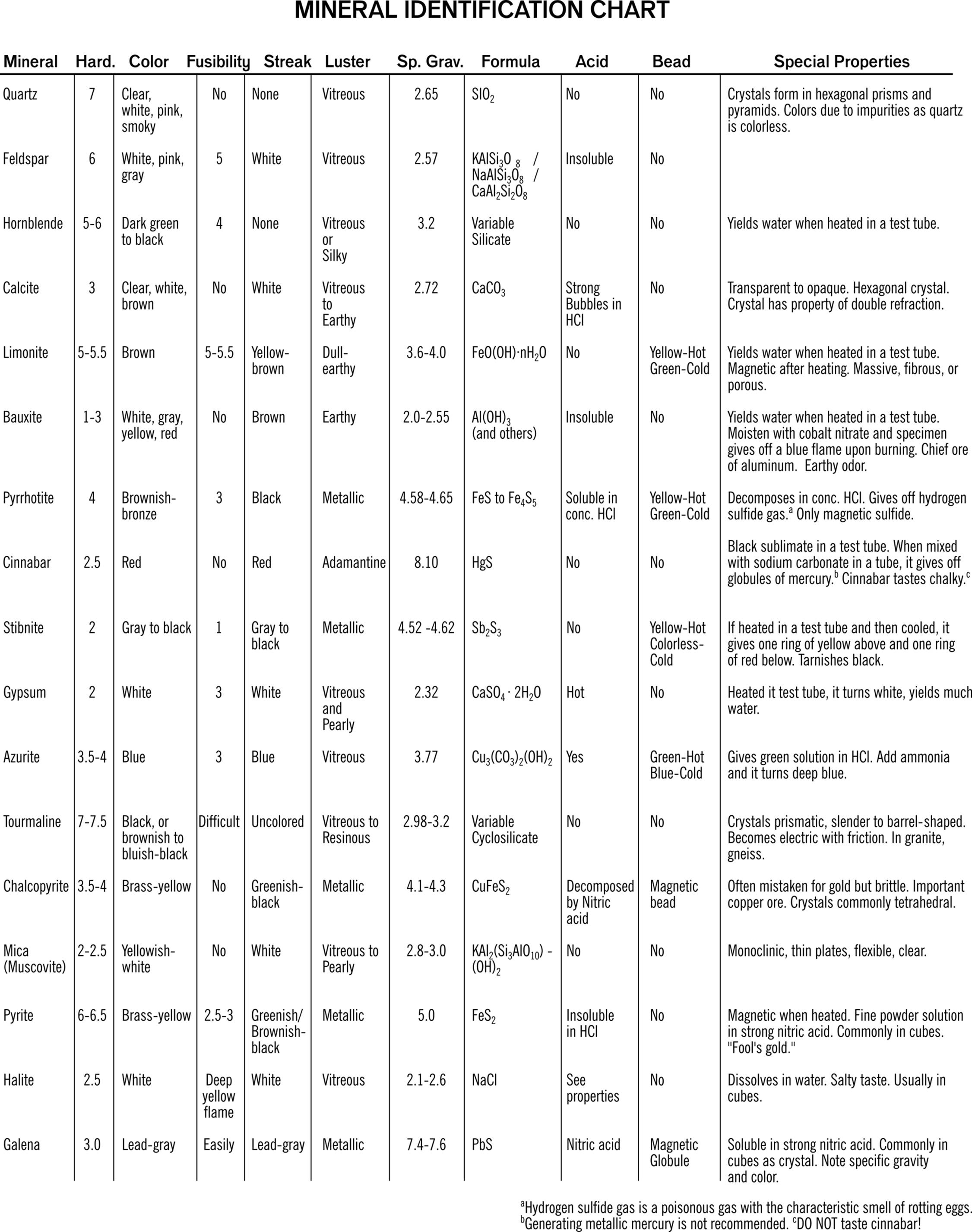
You can study the mineral identification chart and try some of these tests mentioned under the heading, “Special Properties.”
Seismograph
Purpose: seismograph is used to record shock waves from disturbances such as earthquakes. A modern use of seismographs is to detect nuclear and thermonuclear bomb tests. The seismograph operates on the principle of a stationary pendulum. A suspended pendulum remains motionless while the Earth moves beneath it. Since the recording drum is attached to the Earth, the drum moves back and forth and the record of the movement is inscribed by a pointer attached to the motionless pendulum.
Materials: The base is made of either a heavy 2” × 12” plank or a slab of concrete. Since the concrete is heavier, it will make a firmer support and will not vibrate with slight movements caused by local sources such as cars. You will also need a pipe flange, a ¾” pipe about three feet long, a metal rod about three feet in length, a steel wire about five feet in length, and an old alarm clock.8
What to Do: Drill a small dimple (the beginning of a hole) in the ¾” pipe as shown. Grind one end of the rod to a point. This point should fit in the dimple drilled in the pipe. Attach weights to the rod. Heavy bricks or iron can be wired onto the rod. Attach the pipe flange to the base with bolts. Screw the pipe into the flange. Run a wire from the top of the pipe to the end of the rod after the pointed end of the rod is placed in the dimple.
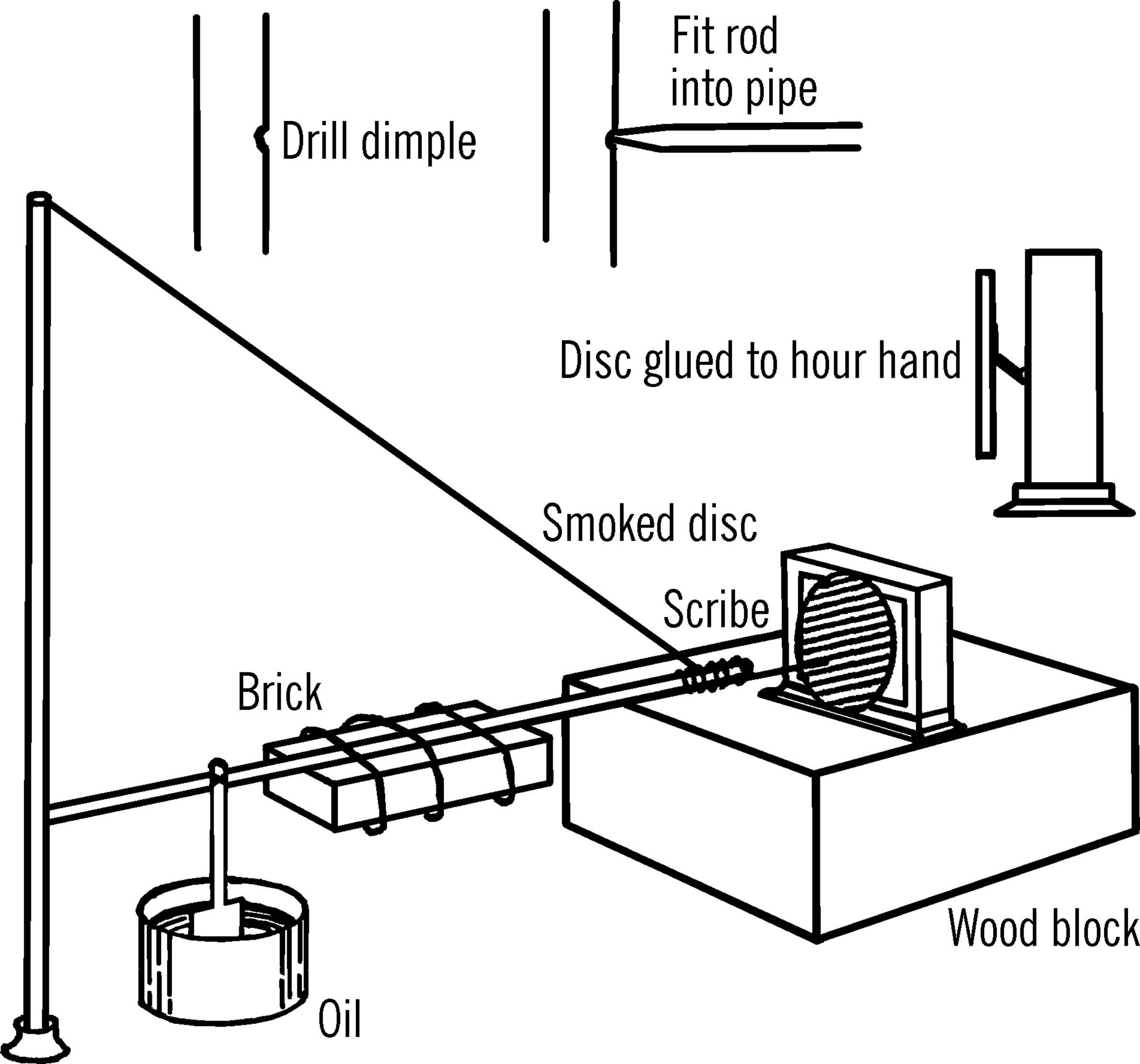
Bolt a stand supporting an alarm clock to the base in such a position that the end of the rod almost touches the outside edge of the clock dial. The minute hand of the clock should be removed. A thin cardboard disc is glued to the hour hand. The disc is smoked by holding it over a candle flame. The soot will blacken the face of the disc in a few minutes. A ball point pen or a stiff wire is fastened to the rod. This point makes a scratch mark in the black soot as the hour hand turns the disc.9
Operation of Equipment: When an earthquake or severe shock occurs, the base shakes. The pendulum point (end of rod with the ball point pen) does not move, and therefore the point of the pen moves back and forth across the dial. The greater the shock the farther the point will seem to move across the dial. If the outside of the dial is marked with the hours, the time of the shock can be read by close observation of the marks on the dial. Minor shocks can be eliminated by fastening a metal plate from the bottom of the rod so that the plate hangs vertically in a pan of oil. Then the oil will slow the movement of the rod (dampen it) and you will have one or two marks across the dial instead of a large number. You can determine the direction of the earthquake if you make two seismographs and have them face crossways to each other.
An earthquake sends out shock waves that travel at different speeds. The first wave to reach your seismograph is called a pressure wave. If you note the time that lapses between the first and second wave (shear wave), you can tell the approximate distance at which the earthquake occurred. The second wave travels about 5/9ths as fast as the first wave. Charts are available which give the time lapse and the distance for each time lapse.
Can You Work Like a Scientist?
1. A seismograph works much better if the base is fastened to bedrock (the solid layer of rock underneath the topsoil and subsoil). Can you find out how far down bedrock is in your neighborhood? Does the depth to bedrock vary from place to place?
2. Earthquakes occur more often in certain places around the Earth. Can you find out where the earthquake belts occur around the Earth?
3. The Earth is often likened to a huge ball filled with molten liquid rock and metals which surrounds a dense core. Fill a balloon with water. The outside skin of the balloon is similar to the crust of the Earth. What would happen if a hole were punched in the balloon? What occurs when this happens on the surface of the Earth?
4. Tie the neck of the water-filled balloon to a bent nail inserted in a hand drill. Turn the hand drill rapidly so you whirl the balloon. Can you see why, as the Earth was cooling, the equator bulged out and the poles of the Earth seemed to flatten? The Earth contained molten materials. These materials were thrown out away from the equator as the Earth rotated on its axis.
5. Earthquakes are often caused by a pressure or push somewhere else on Earth. Push on one side of the water balloon with your finger. Does this extra pressure affect the balloon in another place?
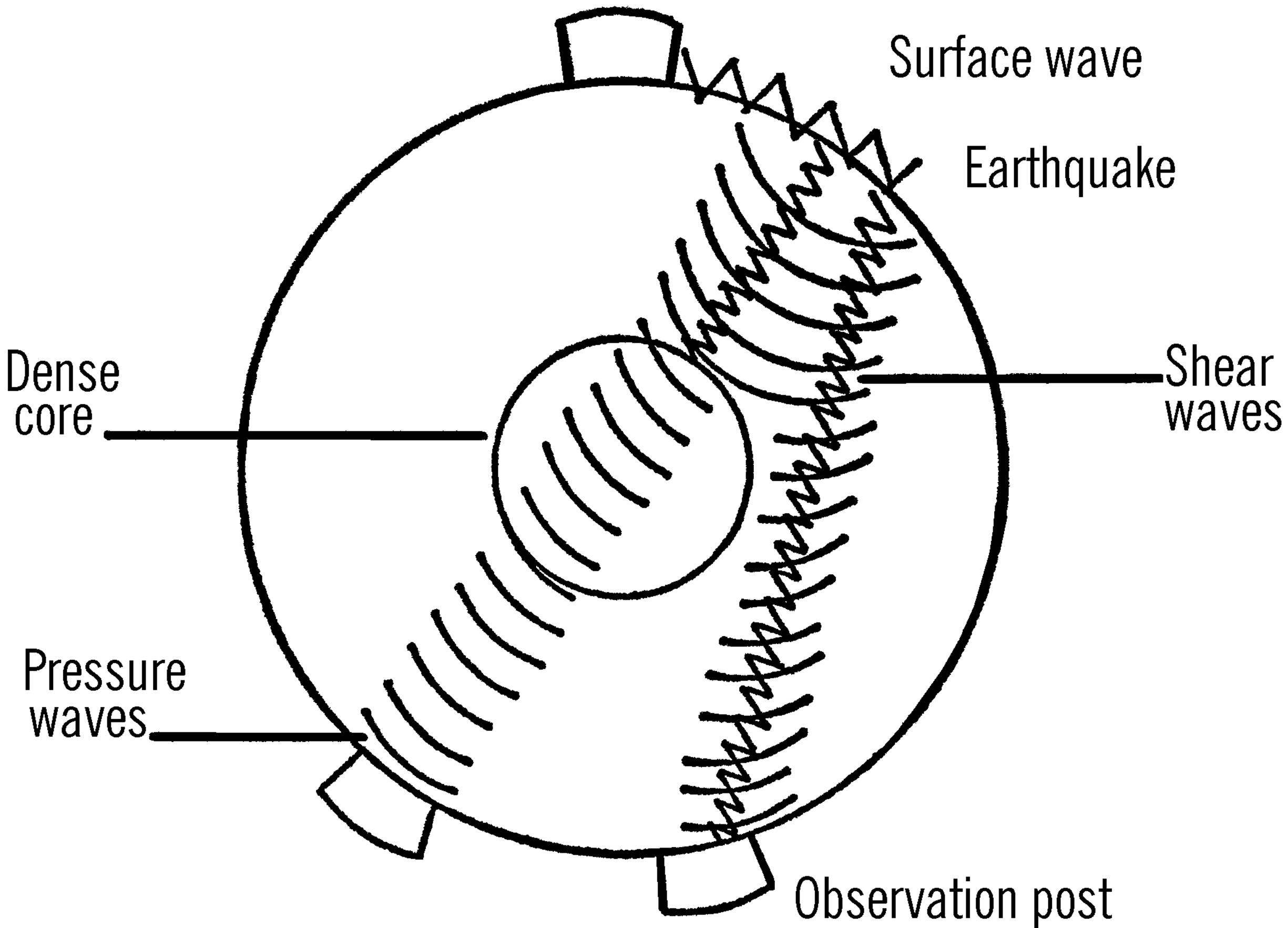
6. Earthquakes help the geologist to determine what is inside the Earth and the layers of the interior part of the Earth. There are observation posts all around the Earth. When an earthquake occurs, it sends out three different types of waves. The shear waves go through the molten rock but not through the dense core of the Earth. From the drawing can you see how reports from different observation points would help pinpoint the location of the dense core?
7. Get several knitting needles (or wires) and an avocado. Stick the knitting needles through the avocado from different positions. Can you locate the core without seeing it?
8. The pressure of the Earth often pushes layers of rocks together. Since the rocks can’t compress, they fold either up or down. Open a book. Push on the top and bottom of the page. Can you see an example of an upfold? A downfold? Can you find examples of these Earth activities around where you live?
9. Mix several sheets of colored paper together. These represent layers of Earth. Cut the stack and let one pile drop about a quarter of an inch. Can you see how a sudden dropping of a section of the Earth can be detected by observing rock layers? This dropping is called faulting. There are many such weak areas around the Earth where faulting occurs. Can you find where the major fault lines run in your area? Faults send out waves, which are called earthquakes.
Volcano
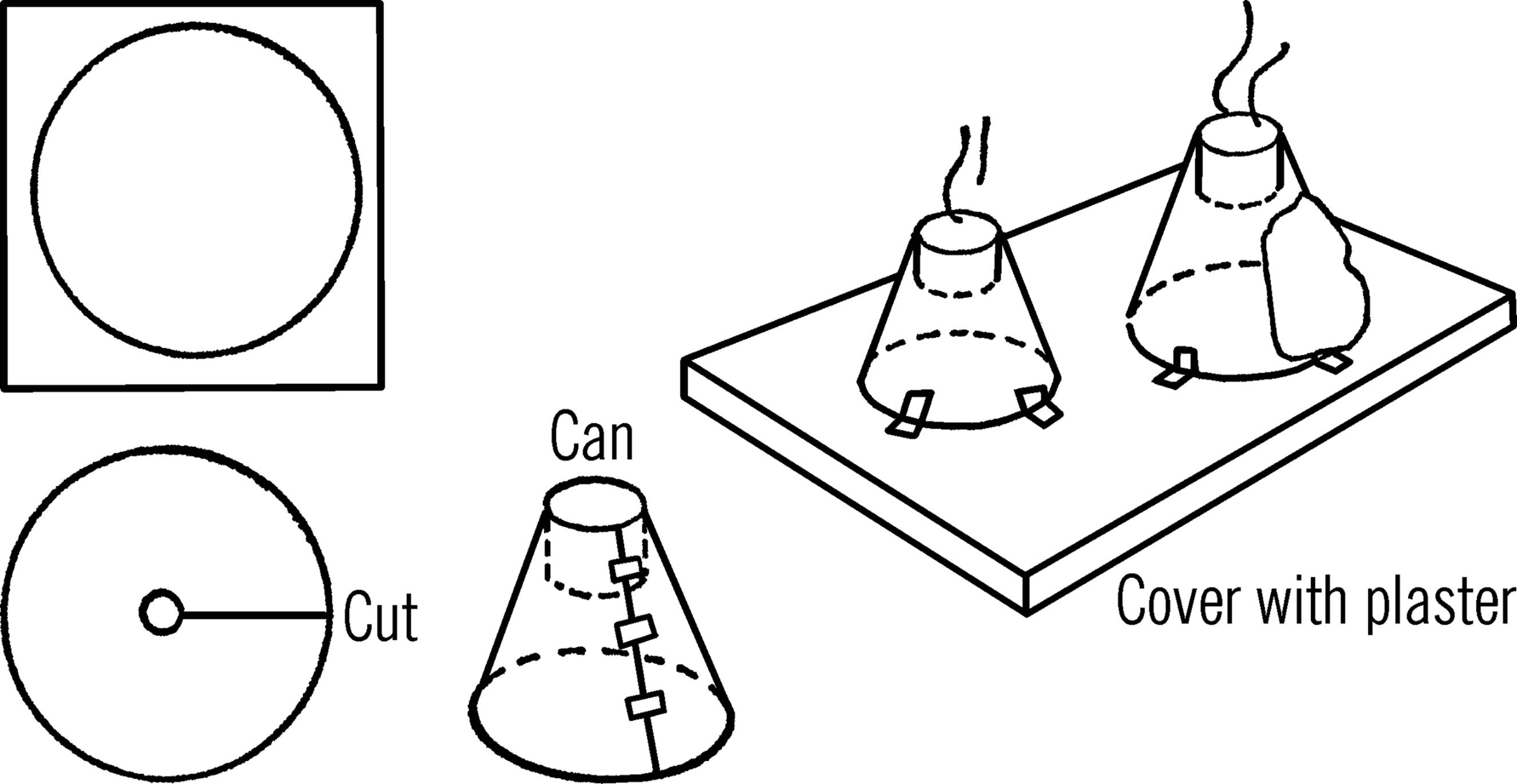
Purpose: This model volcano erupts and demonstrates the building of mountains.
Materials: Two pieces of cardboard (the larger the pieces the bigger the volcanoes), two small cans (baby food or fruit juice),10 and plaster of Paris.
Safety Tips
1. Don’t handle the chemicals with your bare hands. Some people are allergic to certain chemicals of this type. You should never directly handle any chemicals. Use spoons, etc.
2. Don’t inhale the fumes. You should never inhale gas fumes of any type.
3. Don’t try to plug the volcano to get an explosion. Any gas fumes that are trapped build up pressure. If these fumes can’t get out, they explode. The baby food can would burst, and it could be dangerous. There is no danger at all if you follow these simple precautions.11
Modern Safety Practice
This project has chemicals, fire, flying sparks, and toxic fumes. If at all possible, do it outdoors in a place where sparks and fumes won’t be a problem.
1. Follow safe-handling precautions for chemicals (Note 17 in Appendix E), including the use of protective gloves so that you do not handle the chemicals directly.
2. There will be fire. Awesome, fun fire, but fire nonetheless. Follow fire safety practice as described in Note 3 of Appendix E.
3. The fumes are dangerous, so excellent ventilation is required. Ammonium dichromate is both toxic and a carcinogen. Again, it is best to perform this experiment outdoors.
What to Do: Make as large a circle as possible on the cardboard. Cut the circle out. Cut the cardboard as shown. Make the hole in the center so that the cardboard will fit tightly around the baby food can. Bend the cardboard into a cone and insert the baby food can. Hold the cardboard together with tape or staples. Make a second, similar cone. Fasten them down to a wooden base with masking tape. Mix up plaster of Paris into a thick paste. Cover the cones with plaster of Paris. Shape the outside of the cones like two mountain peaks, making a rough finish. Put some plaster of Paris inside the cans so the opening in the can is only about an inch or an inch and a half deep. Allow the plaster to dry and then paint the volcano with a mixture of colors.
Operation of Equipment: Fill the opening of the volcanoes with about two spoonfuls of a chemical, ammonium dichromate. This chemical is used for photography and is available in photographic supply houses.12 It can also be purchased from scientific supply houses. Lay a lighted match on the top of the ammonium dichromate. The chemical will ignite, and the volcano will start to erupt. Bits of magnesium powder, added to the ammonium dichromate, gives a more spectacular effect.
Can You Work Like a Scientist?
1. Does the volcanic ash go way up in the air? How high does the ash go on a real volcano? Find out about the volcano that erupted on the island of Krakatoa in the South Pacific.
2. Could such action of a volcano have caused the start of the ice ages?
3. Can you find the cinder cone? Does your mountain grow? Where is the lava?
1 Cobalt nitrate was originally in this list, but may be omitted from a standard kit. Certain substances become colored when moistened with a cobalt nitrate solution and heated.
2 Quartz may be found as common agate rocks. Corundum paper is more often labeled as aluminum oxide sandpaper. Carborundum is the abrasive on silicon carbide sandpaper. Most “wet or dry” sandpaper is silicon carbide.
3 Note that the word is more commonly spelled lustre, in the context of mineralogy
4 Adamantine luster means diamondlike: as in, transparent and sparkly.
5 No longer; see Note 16 in Appendix E about acids.
6 If you do this, be sure to practice without the flame!
7 How can you find the bead color for other materials?
8 Specifically, a clock with moving hands. A clock that runs on wall power (one driven by an internal AC motor) is ideal. An alarm function is not actually needed.
9 There is probably not enough pressure to write on paper with a ballpoint pen, but a rollerball pen might work on plain (unsmoked) paper or cardboard. Would a felt-tip pen work instead? Why or why not?
10 Cat food or tuna cans might be good choices as well.
11 Correct in spirit. However, think it through as well: There is no danger other than the fire-spitting pile of flaming poisonous chemicals.
12 That was true back in the days before digital photography! See Appendix A for current sources.