Cracking the AP Biology Exam
2
The Chemistry of Life
PROTEINS
Amino acids are organic molecules that serve as the building blocks of proteins. They contain carbon, hydrogen, oxygen, and nitrogen atoms. There are 20 different amino acids commonly found in proteins. Fortunately, you don’t have to memorize the 20 amino acids. But you do have to remember that every amino acid has four important parts: an amino group (–NH2), a carboxyl group (–COOH), a hydrogen, and an R group.
Here’s a typical amino acid:
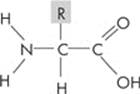
Amino acids differ only in the R group, which is also called the side chain. The R group associated with an amino acid could be as simple as a hydrogen atom (as in the amino acid glycine) or as complex as a carbon skeleton (as in the amino acid arginine).
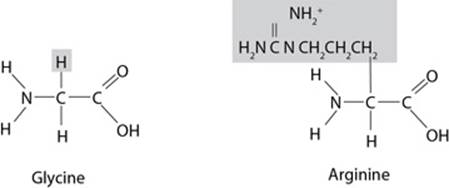
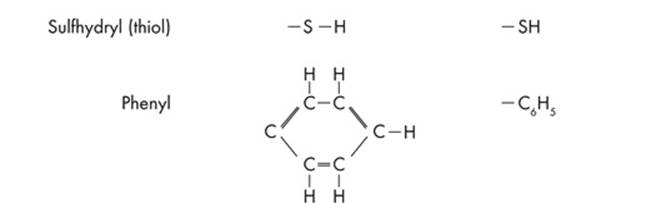
Since ETS will probably test you on chemical diagrams, it’s a good idea to identify the functional groups of the different structures. Functional groups are the distinctive groups of atoms that play a large role in determining the chemical behavior of the compound they are a part of. For example, an organic acid has a functional group, the carboxyl group (–COOH) that releases hydrogen ions in water. This makes the solution acidic. When it comes to spotting an amino acid, simply keep an eye out for the amino group (NH2), then look for the carboxyl molecule (COOH). The most common functional groups of organic compounds are listed on the next page.
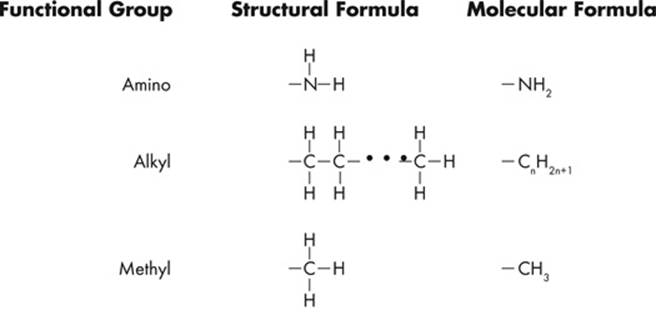
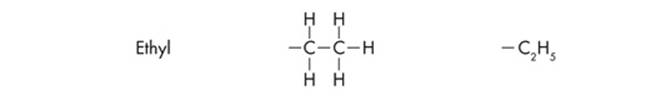
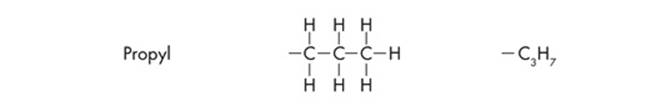
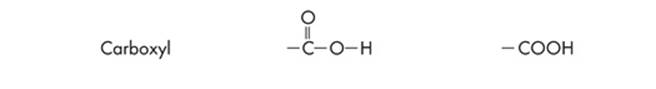
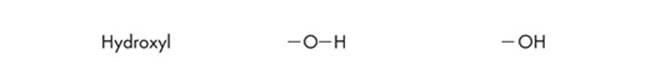
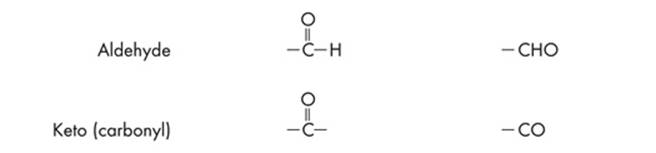
Polypeptides
When two amino acids join they form a dipeptide. The carboxyl group of one amino acid combines with the amino group of another amino acid. Here’s an example:
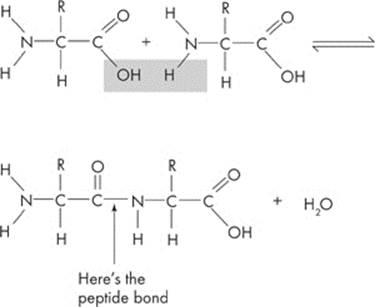
This is the same process we saw earlier: dehydration synthesis. Why? Because a water molecule is removed to form a bond. By the way, the bond between two amino acids has a special name—a peptide bond. If a group of amino acids are joined together in a “string,” the resulting organic compound is called a polypeptide. Once a polypeptide chain twists and folds on itself, it forms a three-dimensional structure called a protein.